UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
_________________________
FORM 20-F
_________________________
(Mark One)
| REGISTRATION STATEMENT PURSUANT TO SECTION 12(b) OR (g) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| ANNUAL REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
For the fiscal year ended December 31 , 2025
OR
| TRANSITION REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
OR
| SHELL COMPANY REPORT PURSUANT TO SECTION 13 OR 15(d) OF THE SECURITIES EXCHANGE ACT OF 1934 | |||||
Date of event requiring this shell company report
For the transition period from to
Commission file number: 001-41401
_________________________
(Exact name of Registrant as specified in its charter)
_________________________
N/A
(Translation of Registrant’s name into English)
(Jurisdiction of incorporation or organization)
(Address of principal executive offices)
Phone: +852 2210-9588
(Name, Telephone, and Address of Company Contact Person)
_________________________
Securities registered or to be registered pursuant to Section 12(b) of the Act:
| Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||||||||||||
Securities registered or to be registered pursuant to Section 12(g) of the Act:
None
(Title of Class)
Securities for which there is a reporting obligation pursuant to Section 15(d) of the Act:
None
(Title of Class)
_________________________
Indicate the number of outstanding shares of each of the issuer’s classes of capital or common stock as of the close of the period covered by the annual report:
As of December 31, 2025, there were 16,874,089 ordinary shares issued and outstanding, par value $0.0015 per share, being the sum of 15,293,117 Class A Ordinary Shares, 1,580,972 Class B Ordinary Shares, and 17,352,363 Warrants.
Indicate by check mark if the registrant is a well-known seasoned issuer, as defined in Rule 405 of the Securities Act.
Yes o No x
If this report is an annual or transition report, indicate by check mark if the registrant is not required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934.
Yes o No x
Note - Checking the box above will not relieve any registrant required to file reports pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934 from their obligations under those Sections.
Indicate by check mark whether the registrant: (1) has filed all reports required to be filed by Section 13 or 15(d) of the Securities Exchange Act of 1934 during the preceding 12 months (or for such shorter period that the registrant was required to file such reports), and (2) has been subject to such filing requirements for the past 90 days.
Indicate by check mark whether the registrant has submitted electronically every Interactive Data File required to be submitted pursuant to Rule 405 of Regulation S-T (§232.405 of this chapter) during the preceding 12 months (or for such shorter period that the registrant was required to submit such files).
Indicate by check mark whether the registrant is a large accelerated filer, an accelerated filer, a non-accelerated filer, or an emerging growth company. See the definitions of “large accelerated filer,” “accelerated filer,” and “emerging growth company” in Rule 12b-2 of the Exchange Act.
Large accelerated filer o | Accelerated filer o | Emerging Growth Company | |||||||||||||||||||||
If an emerging growth company that prepares its financial statements in accordance with U.S. GAAP, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards† provided pursuant to Section 13(a) of the Exchange Act. o
†The term “new or revised financial accounting standard” refers to any update issued by the Financial Accounting Standards Board to its Accounting Standards Codification after April 5, 2012.
Indicate by check mark whether the registrant has filed a report on and attestation to its management’s assessment of the effectiveness of its internal control over financial reporting under Section 404(b) of the Sarbanes-Oxley Act (15 U.S.C. 7262(b)) by the registered public accounting firm that prepared or issued its audit report.
Yes o No x
If securities are registered pursuant to Section 12(b) of the Act, indicate by check mark whether the financial statements of the registrant included in the filing reflect the correction of an error to previously issued financial statements. o
Indicate by check mark whether any of those error corrections are restatements that required a recovery analysis of incentive-based compensation received by any of the registrant’s executive officers during the relevant recovery period pursuant to §240.10D-1(b). o
Indicate by check mark which basis of accounting the registrant has used to prepare the financial statements included in this filing:
U.S. GAAP o | Other o | |||||||||||||
by the International Accounting Standards Board x | ||||||||||||||
If “Other” has been checked in response to the previous question, indicate by check mark which financial statement item the registrant has elected to follow.
o Item 17 o Item 18
If this is an annual report, indicate by check mark whether the registrant is a shell company (as defined in Rule 12b-2 of the Exchange Act).
Yes o No x
(APPLICABLE ONLY TO ISSUERS INVOLVED IN BANKRUPTCY PROCEEDINGS DURING THE PAST FIVE YEARS)
Indicate by check mark whether the registrant has filed all documents and reports required to be filed by Sections 12, 13 or 15(d) of the Securities Exchange Act of 1934 subsequent to the distribution of securities under a plan confirmed by a court.
Yes o No o
TABLE OF CONTENTS
| Page | |||||
CONVENTIONS AND FREQUENTLY USED TERMS
In this annual report, unless otherwise indicated or unless the context otherwise requires:
“ACT Genomics” means ACT Genomics Holdings Company Limited;
“ACT Acquisition” means the acquisition of 74.39% of the equity interest in ACT Genomics;
“ACT Sale and Purchase Agreements” means the Agreements for Sale and Purchase dated December 16, 2022 and January 3, 2023, respectively, by and among the Company, ACT Genomics, and certain other persons specified thereunder;
“Artisan” means Artisan Acquisition Corp., an exempted company limited by shares incorporated under the laws of the Cayman Islands;
“Business Combination” means the Initial Merger, the Acquisition Merger and the other transactions contemplated by the Business Combination Agreement;
"Business Combination Agreement" means the business combination agreement, dated September 15, 2021 (as amended by an Amendment to Business Combination Agreement dated as of March 30, 2022) by and among the Company, Artisan and other parties thereto;
“Cayman Islands Companies Act” means the Companies Act (As Revised) of the Cayman Islands;
“China” or “PRC,” in each case, means the People’s Republic of China, including Hong Kong and Macau and excluding, solely for the purpose of this annual report, Taiwan. The term “Chinese” has a correlative meaning for the purpose of this annual report;
“Class A Ordinary Share” means a Class A ordinary share, par value $0.0015 per share, of the Company;
“Class B Ordinary Share” means a convertible Class B ordinary share, par value $0.0015 per share, of the Company;
“Closing” means the closing of the Acquisition Merger;
“Closing Date” means May 18, 2022;
“Continental” means Continental Stock Transfer & Trust Company;
“ESOP” means the 2021 Share Incentive Plan of Prenetics adopted on June 16, 2021, as may be amended from time to time;
“Europa Group” means Europa Group Holdings Ltd, an exempted company limited by shares incorporated under the laws of the Cayman Islands;
“Hubmatrix” means Hubmatrix Partners, LLC; a limited liability company incorporated under the laws of the State of Delaware;
“IM8” means IM8, our flagship consumer health business;
“IM8 Group” means IM8 Group Holding Limited, an exempted company limited by shares incorporated under the laws of the Cayman Islands;
“Insighta” means Insighta Holdings Limited, an exempted company limited by shares incorporated under the laws of the Cayman Islands;
“mainland China” means the People’s Republic of China, excluding, solely for the purpose of this annual report, Hong Kong, Macau and Taiwan. The term “mainland Chinese” has a correlative meaning for the purpose of this annual report;
1
“Nasdaq” means the Nasdaq Stock Market;
“Prenetics HK” means Prenetics Limited, a limited liability company incorporated in Hong Kong;
"Reverse Stock Split" means the 1-for-15 reverse stock split effected by the Company on November 14, 2023. In this annual report, where we state historical share and per-share numbers, we have, where appropriate, reflected a retroactive adjustment due to the Reverse Stock Split in parentheses;
“SEC” means the U.S. Securities and Exchange Commission;
“securities” refer to our Class A Ordinary Shares and Warrants;
“shares” or “ordinary shares” refer to our Class A Ordinary Shares and Class B Ordinary Shares;
“U.S. Dollars,” “US$,” “USD” and “$” means United States dollars, the legal currency of the United States;
“Warrants” means warrants of the Company, each entitling its holder to purchase 1.29 Class A Ordinary Share at an exercise price of $133.65 per 1.29 shares (or an effective price of $103.60 per share), subject to adjustment pursuant to the terms of the Assignment, Assumption and Amendment Agreement and the warrant agreement, dated May 13, 2021, by and between Artisan and Continental.
“we,” “us,” “our,” “Prenetics,” “the Company” and “our company” refer to Prenetics Global Limited and its subsidiaries and consolidated affiliated entities, unless the context indicates otherwise.
References to “U.S. Dollars,” “USD,” “US$” and “$” in this annual report are to United States dollars, the legal currency of the United States. Discrepancies in any table between totals and sums of the amounts listed are due to rounding. Certain amounts and percentages have been rounded; consequently, certain figures may add up to be more or less than the total amount and certain percentages may add up to be more or less than 100% due to rounding. In particular and without limitation, amounts expressed in millions contained in this annual report have been rounded to a single decimal place for the convenience of readers.
2
FORWARD-LOOKING STATEMENTS
This annual report on Form 20-F includes statements that express our opinions, expectations, beliefs, plans, objectives, assumptions or projections regarding future events or future results of operations or financial condition and therefore are, or may be deemed to be, “forward-looking statements.” These forward-looking statements are made under the “safe harbor” provisions of the U.S. Private Securities Litigation Reform Act of 1995. These forward-looking statements can generally be identified by the use of forward-looking terminology, including the terms “believe,” “estimate,” “anticipate,” “expect,” “seek,” “project,” “intend,” “plan,” “may,” “will” or “should” or, in each case, their negative or other variations or comparable terminology. These forward-looking statements include all matters that are not historical facts. They appear in a number of places throughout this annual report and include statements regarding our intentions, beliefs or current expectations concerning, among other things, our results of operations, financial condition, liquidity, prospects, growth, strategies, future market conditions or economic performance and developments in the capital and credit markets and expected future financial performance, the markets in which we operate, as well as the possible or assumed future results of operations of our Company. Such forward-looking statements are based on available current market material and management’s expectations, beliefs and forecasts concerning future events impacting us. Factors that may impact such forward-looking statements include:
•The regulatory environment and changes in laws, regulations or policies in the jurisdictions in which we operate;
•Our ability to successfully compete in highly competitive industries and markets;
•Our ability to continue to adjust our offerings to meet market demand, attract customers to choose our products and services and grow our ecosystem;
•Political instability in the jurisdictions in which we operate;
•The overall economic environment and general market and economic conditions in the jurisdictions in which we operate;
•Our acquisition of digital assets in connection with our previous treasury strategy exposes us to financial and regulatory risks;
•Our ability to execute our strategies, manage growth and maintain our corporate culture as we grow;
•Our anticipated investments in new products, services, collaboration arrangements, technologies and strategic acquisitions, and the effect of these investments on our results of operations;
•Our ability to develop and protect intellectual property;
•Changes in the need for capital and the availability of financing and capital to fund these needs;
•Anticipated technology trends and developments and our ability to address those trends and developments with our products and services;
•The safety, affordability, convenience and breadth of our products and services;
•Man-made or natural disasters, health epidemics, and other outbreaks including war, acts of international or domestic terrorism, civil disturbances, occurrences of catastrophic events and acts of God such as floods, earthquakes, wildfires, typhoons and other adverse weather and natural conditions that may directly or indirectly affect our business or assets;
•The loss of key personnel and the inability to replace such personnel on a timely basis or on acceptable terms;
•Exchange rate fluctuations;
•Changes in interest rates or rates of inflation;
•Legal, regulatory and other proceedings;
•Our ability to maintain the listing of our securities on Nasdaq;
•The inability to increase the authorized capital stock;
•The results of any future financing efforts;
•Our ability to integrate our business successfully with newly acquired businesses and realize the anticipated synergies and related benefits, or to do so within the anticipated timeframe; and
•Other risks and uncertainties, including those described in “Item 3. Key Information — D. Risk Factors.”
3
The forward-looking statements contained in this annual report are based on our current expectations and beliefs concerning future developments and their potential effects on us. There can be no assurance that future developments affecting us will be those that we have anticipated. These forward-looking statements involve a number of risks, uncertainties (some of which are beyond our control) or other assumptions that may cause actual results or performance to be materially different from those expressed or implied by these forward-looking statements. These risks and uncertainties include, but are not limited to, those factors described under “Item 3. Key Information — D. Risk Factors.” Should one or more of these risks or uncertainties materialize, or should any of the assumptions prove incorrect, actual results may vary in material respects from those projected in these forward-looking statements. We do not undertake any obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise, except as may be required under applicable securities laws. In light of these risks and uncertainties, you should keep in mind that any event described in a forward-looking statement made in this annual report or elsewhere might not occur.
This annual report includes market data and industry statistics and forecasts that are based on independent industry publications and other publicly available information. Although we believe these sources are reliable, we do not guarantee the accuracy or completeness of this information and we have not independently verified this information. In addition, the market and industry data and forecasts included or incorporated by reference in this annual report may involve estimates, assumptions and other risks and uncertainties and are subject to change based on various factors, including those discussed under the heading “Item 3. Key Information — D. Risk Factors” contained in this annual report. Accordingly, you should not place undue reliance on such information.
4
PART I.
ITEM 1.IDENTITY OF DIRECTORS, SENIOR MANAGEMENT AND ADVISERS
Not applicable.
ITEM 2.OFFER STATISTICS AND EXPECTED TIMETABLE
Not applicable.
ITEM 3.KEY INFORMATION
Our Holding Company Structure
We are a Cayman Islands holding company with business operations primarily conducted by our subsidiaries. Investors purchasing our securities are purchasing equity interests in the Cayman Islands holding company.
Throughout this annual report, unless the context indicates otherwise, references to “we,” “us,” “our,” "Prenetics," “the Company” and “our company” refer to Prenetics Global Limited and its subsidiaries and consolidated affiliated entities.
Permissions Required from the PRC Authorities for Our Operations
We believe that, to the extent applicable, we and our subsidiaries have obtained all requisite permissions material to our operations as of the date of this annual report. Our operations are primarily conducted through subsidiaries in Hong Kong and other jurisdictions. For the years ended December 31, 2023, 2024 and 2025, we generated all of our revenue from businesses outside mainland China. We do not sell testing products, solicit customers, or manage customer personal data in mainland China, nor do we have access to such data. Based on advice from our PRC legal counsel, DaHui Lawyers, we believe we are not currently required to obtain any approvals from PRC governmental agencies to operate our business or to list our securities internationally.
However, if we incorrectly conclude that approvals are unnecessary or if regulatory requirements change, obtaining necessary approvals may require significant time and expense. Failure to obtain these approvals could subject us to penalties, sanctions, or restrictions on our business and listing status, materially and adversely affecting our operations and financial condition.
The Holding Foreign Companies Accountable Act
We are headquartered in Hong Kong. Given our operations in Hong Kong and our listing on Nasdaq, we are subject to the Holding Foreign Companies Accountable Act (HFCAA), which may impact investors in our securities. The HFCAA, enacted in December 2020 and amended by the Consolidated Appropriations Act of 2023, requires the U.S. Securities and Exchange Commission (the "SEC") to identify issuers whose audit reports are prepared by accounting firms that the Public Company Accounting Oversight Board (PCAOB) cannot inspect or investigate fully due to foreign jurisdictional restrictions. If an issuer is identified as a "Commission-Identified Issuer" for two consecutive years, the SEC must prohibit the trading of the issuer’s securities on U.S. exchanges, which could affect the liquidity and value of our shares.
For the fiscal year ended December 31, 2025, our financial statements were audited by Deloitte Touche Tohmatsu, a Hong Kong-based accounting firm registered with the PCAOB. In 2022, the PCAOB entered into a Statement of Protocol with the China Securities Regulatory Commission (CSRC) and the Ministry of Finance of the People’s Republic of China, allowing PCAOB inspections of audit firms in mainland China and Hong Kong. Since 2023, the PCAOB has successfully conducted inspections of Hong Kong-based audit firms, including those auditing U.S.-listed companies. As a result, the PCAOB vacated its prior determinations of non-compliance for Hong Kong firms in December 2023, and as of December 31, 2025, there are no PCAOB-identified jurisdictions posing inspection barriers. Consequently, we have not been identified as a Commission-Identified Issuer under the HFCAA for fiscal year 2025.
Risks Relating to Doing Business in Hong Kong
While we currently have no business operations in mainland China and do not utilize Variable Interest Entity (VIE) structures, our operations in Hong Kong as a Special Administrative Region of China expose us to certain legal and regulatory risks. The Chinese government maintains significant oversight authority over Hong Kong, and changes in PRC policies or their application to Hong Kong, particularly regarding data security, cybersecurity reviews, or foreign
5
investment restrictions, could materially impact our business. Although existing PRC regulations such as the Data Security Law and Personal Information Protection Law do not currently apply directly to our operations, any future extension of these or similar regulations to Hong Kong could subject us to new compliance obligations, potentially affecting our ability to operate efficiently, accept foreign investment, or maintain our U.S. listing. Furthermore, evolving U.S.-China relations and related policies may create additional regulatory complexities for Hong Kong-based companies like ours. We continue to monitor these developments and assess potential impacts on our operations and strategic plans.
Enforceability of Civil Liabilities
As a Cayman Islands exempted company with substantially all operations, assets, and management located outside the United States, you may face significant challenges in enforcing legal rights against us or our directors and officers. Most of our directors and executive officers reside in Hong Kong or other non-U.S. jurisdictions, making it difficult to effect service of process or bring actions in U.S. courts based on U.S. securities laws. Judgments obtained in U.S. courts may not be enforceable in the jurisdictions where we or our management are located.
Our corporate governance is subject to Cayman Islands law, including the Companies Act and common law precedents, which differ materially from U.S. corporate law. Notably: (1) Cayman Islands common law derives from limited local judicial precedent and non-binding English common law; (2) shareholder rights and director fiduciary duties differ from those under U.S. state laws like Delaware; and (3) Cayman Islands securities laws provide different protections than U.S. federal securities laws. Additionally, shareholders may lack standing to bring derivative actions in U.S. federal courts.
As a result of all of the above, you may face difficulties in protecting your interests, and your ability to protect your rights through U.S. courts may be limited. See “Item 3. Key Information — D. Risk Factors — Risks Relating to Our Securities — You may face difficulties in protecting your interests, and your ability to protect your rights through U.S. courts may be limited, because we are incorporated under the laws of the Cayman Islands, we conduct substantially all of our operations, and a majority of our directors and executive officers reside, outside of the United States."
A.[Reserved]
B. Capitalization and Indebtedness
Not applicable.
C. Reasons for the Offer and Use of Proceeds
Not applicable.
D. Risk Factors
Summary of Risk Factors
An investment in our Class A Ordinary Shares and Warrants involves significant risks. Below is a selected summary of certain material risks we face. These and other risks are more fully described under “ — D. Risk Factors.” You should carefully consider such risks before making an investment decision. Additional risks not presently known to us or that we currently deem immaterial may also impair our business operations. Our business, financial condition, results of operations or prospects could be materially and adversely affected by any of these risks.
Risks Relating to Our Business and Industry
•We are a relatively early-stage company with a limited operating history in rapidly developing markets, which may make it challenging to evaluate our business and predict our future performance.
•Failure to commercialize key products like CircleDNA and IM8 could materially affect our revenue and future prospects.
•If our products and services do not deliver reliable results as expected, our reputation, business and operating results will be adversely affected.
6
•Our business metrics and key performance indicators are calculated using internal data, methodologies and assumptions that are subject to inherent limitations, and real or perceived inaccuracies in such metrics may harm our reputation and negatively affect our business.
•We have incurred net losses since our inception, and we anticipate that we will continue to incur losses for the foreseeable future, which could harm our future business prospects.
•We have a limited history introducing new products and services to our customers. The future prospects of our business may be harmed if our efforts to attract new customers and engage existing customers by introducing new products are unsuccessful.
•Our CircleDNA and IM8 businesses depend on our ability to maintain a strong base of engaged customers and content creators, including the use of social media. We may not be able to maintain and enhance our brand if we experience negative publicity related to our marketing efforts or use of social media, fail to maintain and grow our network of content creators, or otherwise fail to meet our customers’ expectations.
•We have acquired digital assets in connection with our previous treasury strategy, which exposes us to financial and regulatory risks.
•We rely on a limited number of suppliers, manufacturers, distributors and other service providers, and may not be able to find replacements or immediately transition to alternatives, which could adversely affect our ability to meet customer demand.
•Our operating results may fluctuate significantly, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations.
•Our business significantly depends upon the strength of our brands, including Prenetics, CircleDNA and IM8, and any harm to our brands or reputation may materially and adversely affect our business and results of operations.
•If we cannot provide quality technical and customer and user support, we could lose customers, and our business and prospects may be adversely affected.
•If we are unable to successfully expand our sales and marketing infrastructure to match our growth, our business may be adversely affected.
•The launch of our new nutrient products under the IM8 brand name, may face challenges that could impact our results of operations and reputation.
•The consumer genetic testing market is highly competitive, and many of our competitors are more established and have stronger marketing capabilities and greater financial resources, which presents a continuous threat to the success of our consumer genetic testing business.
•We face risks associated with the acquisition and divestiture of businesses.
•We are highly dependent on our senior management team and key advisors and personnel, and our business and operating results could be harmed if we are unable to retain senior management and key personnel and to attract and retain qualified personnel necessary for our business.
•The sizes of the markets and forecasts of market growth for the demand of our current and pipeline products and services are based on a number of complex assumptions and estimates that are subject to change, and may be inaccurate.
•We may need to raise additional funds to develop our platform, commercialize new products or expand our operations, and we may be unable to raise capital when needed, or on acceptable terms, or at all.
•We may incur debt or assume contingent or other liabilities or dilute our shareholders in connection with acquisitions or strategic alliances.
•If we fail to implement and maintain an effective system of internal controls in the future, we may be unable to accurately report our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, the market price of the Class A Ordinary Shares and the Warrants.
•We depend on the information systems of our own and those of third parties for the effective service on our websites, mobile applications, and in our computer and logistics systems, and for the overall effective and efficient functioning of our business. Failure to maintain or protect our information systems and data integrity effectively could harm our business, financial condition and results of operations.
7
Risks Relating to Doing Business in Hong Kong
•We are subject to risks associated with our operations in Hong Kong, a Special Administrative Region of China.
•Since Hong Kong is a special administrative region of China, legal and operational risks associated with operating in China also apply to operations in Hong Kong and could materially and adversely affect our business and results of operations.
•Unfavorable economic and political conditions in Hong Kong and other parts of Asia could materially and adversely affect our business, financial condition, and results of operations.
•The mainland Chinese government has significant oversight, discretion and control over the manner in which companies incorporated under the laws of mainland China must conduct their business activities, but as we operate in Hong Kong and not mainland China, the mainland Chinese government currently does not exert direct oversight and discretion over the manner in which we conduct our business activities. However, there is no guarantee that the mainland Chinese government will not seek to intervene or influence our operations at any time. If we were to become subject to such oversight, discretion or control, including over overseas offerings of securities and/or foreign investments, it may result in a material adverse change in our operations, significantly limit or completely hinder our ability to offer or continue to offer securities to investors and cause the value of our securities to significantly decline, or be worthless, which would materially affect the interests of the investors.
•Our business, financial condition and results of operations, and/or the value of our securities or our ability to offer or continue to offer securities to investors may be materially and adversely affected to the extent the laws and regulations of mainland China become applicable to us. In that case, we may be subject to the risks and uncertainties associated with the evolving laws and regulations in mainland China, their interpretation and implementation, and the legal and regulatory system in mainland China more generally, including with respect to the enforcement of laws and the possibility of changes of rules and regulations with little or no advance notice.
•Tariffs and trade restrictions on non-U.S. materials could adversely affect our supply chain, costs, and financial performance.
Risks Relating to Government Regulation
•Our business collects and processes a large amount of data including personal information, and we will face legal, reputational, and financial risks if we fail to protect our customers’ data from security breaches or cyberattacks. We are also subject to various laws and regulations relating to privacy or the protection or transfer of data relating to individuals, and any change in such laws and regulations or any failure by us to comply with such laws and regulations could adversely affect our business.
•Our products and services are and will continue to be subject to extensive regulation, compliance of which could be costly and time-consuming or may cause unanticipated delays or prevent the receipt of the required approvals to offer our products and services.
•Our nutritional supplement products are affected by extensive regulations and our failure to comply with any regulations could lead to significant penalties or claims, which could materially harm our financial condition and operating results.
•We plan to expand our business and operations internationally to various jurisdictions in which we do not currently operate and where we have limited operating experience, all of which exposes us to business, regulatory, political, operational and financial risk.
Risks Relating to Intellectual Property and Legal Proceedings
•We may be subject to legal proceedings and litigation, which are costly to defend, and adverse publicity about any investigation, litigation, regulatory or legal action against us or our senior management could harm our reputation and business.
•We depend on intellectual property licensed from third parties for development and commercialization of certain products, and the termination of the licenses or other agreements permitting us to use such intellectual property or failure of such third parties to maintain or protect such intellectual property could result in the loss of significant rights by us, which would harm our business.
8
•We rely substantially on our trademarks and trade names. If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be harmed.
Risks Relating to Our Securities
•The trading prices of our Class A Ordinary Shares and Warrants may be volatile and a market for our Class A Ordinary Shares and Warrants may not develop, which would adversely affect the liquidity and price of our Class A Ordinary Shares.
•Sales of a substantial number of our securities in the public market could cause the price of our Class A Ordinary Shares and Warrants to fall.
•If securities or industry analysts do not publish research, publish inaccurate or unfavorable research or cease publishing research about us, our share price and trading volume could decline significantly.
•Future resales of our ordinary shares issued to our shareholders and other significant shareholders may cause the market price of our Class A Ordinary Shares to drop significantly, even if our business is doing well.
•Our dual-class voting structure may limit your ability to influence corporate matters and could discourage others from pursuing any change of control transactions that holders of our Class A Ordinary Shares may view as beneficial.
•The requirements of being a public company may strain our resources, divert our management’s attention and affect our ability to attract and retain qualified board members.
•We are an “emerging growth company,” and it cannot be certain if the reduced SEC reporting requirements applicable to emerging growth companies will make our Class A Ordinary Shares and Warrants less attractive to investors, which could have a material and adverse effect on us, including our growth prospects.
•We qualify as a foreign private issuer within the meaning of the rules under the Exchange Act, and as such we are exempt from certain provisions applicable to United States domestic public companies.
•We cannot guarantee that any share repurchase program will be fully consummated or that any share repurchase program will enhance long-term shareholder value, and share repurchases could increase the volatility of the price of our Class A Ordinary Shares and could diminish our cash reserves.
•As a company incorporated in the Cayman Islands and a “controlled company” within the meaning of the Nasdaq corporate governance rules, we are permitted to adopt certain home country practices in relation to corporate governance matters that differ significantly from Nasdaq corporate governance listing standards applicable to domestic U.S. companies or rely on exemptions that are available to a “controlled company”; these practices may afford less protection to shareholders than they would enjoy if we complied fully with Nasdaq corporate governance listing standards.
•You may face difficulties in protecting your interests, and your ability to protect your rights through U.S. courts may be limited, because we are incorporated under the laws of the Cayman Islands, and we conduct substantially all of our operations, and a majority of our directors and executive officers reside, outside of the United States.
•The PCAOB had historically been unable to inspect our auditor in relation to their audit work.
•Our securities may be prohibited from being traded in the United States under the Holding Foreign Companies Accountable Act in the future if the PCAOB is unable to inspect or investigate completely auditors located in China. The Holding Foreign Companies Accountable Act, as amended by the Consolidated Appropriations Act, 2023, decreased the number of “non-inspection years” from three years to two years, and thus, reduced the time before our securities may be prohibited from trading or delisted. The delisting of our securities, or the threat of them being delisted, may materially and adversely affect the value of your investment.
•We may issue additional securities without shareholder approval in certain circumstances, which would dilute existing ownership interests and may depress the market price of our shares.
•We have granted in the past, and we will also grant in the future, share incentives, which may result in increased share-based compensation expenses.
•The exercise or exchange of our outstanding warrants will dilute the ownership interest of existing shareholders and could adversely affect the market price of our Class A Ordinary Shares and Warrants.
9
•Our complex capital structure, including multiple classes of warrants with differing terms, may create uncertainty for investors and could adversely affect the trading price of our securities.
•The significant increase in our authorized share capital may result in additional dilution to existing shareholders.
•A provision in the Existing Warrant Agreement may result in additional dilution to our shareholders.
•Our securities may be delisted from Nasdaq as a result of our failure of meeting the Nasdaq continued listing requirements.
Risks Relating to Taxation
•We may be or become a passive foreign investment company (“PFIC”), which could result in adverse U.S. federal income tax consequences to U.S. Holders of our Class A Ordinary Shares or warrants.
Risks Relating to Our Business and Industry
We are a relatively early-stage company with a limited operating history in rapidly developing markets, which may make it challenging to evaluate our business and predict our future performance.
We have a limited operating history upon which you can evaluate our business and prospects. Our limited operating history may make it difficult to evaluate our current business and predict our future performance, prospects or viability. Operating in new markets like genetic testing and nutritional supplements creates uncertainties in forecasting revenue, scaling operations, and competing effectively. Any assessment of our prospects is subject to significant uncertainty and must be considered in light of the risks and difficulties frequently encountered by companies in their early stage of development, particularly those in new and rapidly evolving markets like us. These risks include, among others, an evolving and unpredictable business model and the management of growth. Failure to address these risks successfully, our revenue, results of operations and business could be materially and adversely affected.
Failure to commercialize key products like CircleDNA and IM8 could materially affect our revenue and future prospects.
The commercial success of our key products is dependent on gaining regulatory approvals, gaining acceptance from healthcare providers and consumers, maintaining competitive pricing, executing effective marketing, securing partnerships, and complying with regulations across jurisdictions. Rapid technological advances in diagnostics could render our products obsolete if we fail to innovate.
If our products are not successfully commercialized, our ability to generate revenue and achieve profitability could be materially impaired, limiting our capacity to develop new products.
Our recent expansion into the consumer health and wellness market through the launch of IM8, a premium supplements brand co-founded with David Beckham, introduces additional risks that could materially and adversely affect our business and future prospects. The success of IM8 is contingent upon factors such as: (i) the ability to develop and market products that meet consumer preferences and health trends; (ii) establishing and maintaining a strong brand reputation in a competitive market; (iii) effective collaboration with partners like David Beckham and institutions such as the San Francisco Research Institute; (iv) navigating regulatory requirements for health supplements across various jurisdictions; and (v) managing supply chain logistics, especially following the divestiture of our Europa 3PL business, to ensure timely distribution and product availability. Failure in any of these areas could impede the successful commercialization of IM8 products, thereby adversely impacting our revenue and profitability.
If our products and services do not deliver reliable results as expected, our reputation, business and operating results will be adversely affected.
The success of our products and services depends on the market’s confidence that we can provide safe and effective health supplement products through IM8, and reliable test kits that enable high-quality genomic testing with high accuracy, sensitivity and specificity and with short turnaround times through CircleDNA. There is no guarantee that the success and growth we have demonstrated to date will continue as our product deliveries increase and our product portfolio expands.
Our products and services use a number of complex and sophisticated biochemical and bioinformatics processes, many of which are highly sensitive to external factors, including human error. An operational, technological, user or other failure in one of these complex processes or fluctuations in external variables may result in performance outcomes that are
10
lower than we anticipate or result in customer dissatisfaction. As a result, the efficacy and commercial attractiveness of our products may be adversely affected, and our reputation may be harmed. If our products do not perform, or are perceived to not have performed, as expected or favorably in comparison to competitive products, our operating results, reputation, and business will suffer, and we may also be subject to legal claims arising from product limitations, errors, or inaccuracies.
Furthermore, there is no guarantee that customers will always use these products properly in the manner in which they are intended. Any intentional or unintentional misuse of these products by customers could lead to substantial civil and criminal monetary and non-monetary penalties, and could result in significant legal and investigatory fees.
Our business metrics and key performance indicators are calculated using internal data, methodologies and assumptions that are subject to inherent limitations, and real or perceived inaccuracies in such metrics may harm our reputation and negatively affect our business.
We track a number of operational and business metrics to evaluate the performance and growth of our business, including, among others, monthly revenue, annualized recurring revenue ("ARR"), average order value, new customer subscription rate, customer orders, and others. Our metrics and key performance indicators ("KPIs") are calculated using internal company data, definitions and methodologies that have not been independently verified by any third party and are not based on any standardized industry methodology. Other companies, including companies in the consumer health and direct-to-consumer industries, may calculate similarly titled metrics differently, which may reduce the value of such metrics as a basis for comparison.
We are a relatively early-stage company, and our business, particularly our IM8 brand, is growing rapidly across more than 30 countries, with significant operational complexity spanning product formulation, manufacturing, global distribution, subscription management, digital marketing and customer support. As our business scales, we are required to process and analyze increasingly large volumes of transactional, financial and operational data across multiple systems, platforms and geographies. While we have invested, and expect to continue to invest, in data infrastructure, internal controls and analytical tools designed to ensure the accuracy and reliability of our reported metrics, there can be no assurance that our current or future systems will be adequate to manage the growing scale and complexity of our operations or that our data will be free from errors, omissions or inaccuracies.
Our metrics may be affected by a number of factors, including but not limited to: limitations or errors in our data collection, processing or reporting systems; challenges in accurately tracking customer behavior across platforms and regions, including with respect to subscription renewals, cancellations and refund activity; the use of estimates or assumptions in the calculation of certain metrics; changes in our internal definitions or methodologies over time, which may limit period-to-period comparability; the impact of promotional activity, seasonal fluctuations or product launches on short-term metrics; and discrepancies between internal and externally reported data due to timing, rounding or classification differences. In addition, as we enter new markets and introduce new products, our existing systems and processes may not be sufficient to capture and report operational data with the same degree of accuracy or granularity as in our more established markets.
If our metrics are found to contain material inaccuracies, or if investors or analysts perceive them to be unreliable, our credibility and reputation could be harmed, which could in turn negatively affect the trading price of our Class A Ordinary Shares and Warrants. Furthermore, if we are unable to maintain accurate and reliable operational data, our management's ability to make informed strategic and operational decisions could be impaired, which could adversely affect our business, financial condition and results of operations. We may also need to expend significant additional resources to improve our data infrastructure and internal controls, and there can be no assurance that such efforts will be successful or timely.
We also expect that, as our business continues to evolve, we may revise or cease reporting certain metrics, or introduce new metrics, in order to better reflect the performance of our business. Any such changes could make it more difficult for investors to assess our historical or future performance on a consistent basis.
We have incurred net losses since our inception, and we anticipate that we will continue to incur losses for the foreseeable future, which could harm our future business prospects.
We have incurred substantial losses since our inception. For the years ended December 31, 2023, 2024 and 2025, our net losses were $64.8 million, $49.8 million and $38.7 million, respectively. We have historically financed our operations principally from the issuances of ordinary shares, preferred shares, convertible securities and warrants to third-party investors. We may continue to incur losses both in the near term and longer term as we continue to devote a significant portion of our resources to, among other things, expanding into consumer health, developing international markets, scaling up our business and operations, engaging in marketing activities to increase public awareness and acceptance of our
11
products, engaging in continued research and development, and other related business activities, and as we incur additional costs associated with operating as a public company.
While our revenue has increased over time, given the numerous risks and uncertainties associated with our research, development, and commercialization efforts, we expect to continue to incur significant losses as we develop and invest in our business, and we are unable to predict when we will become profitable on a sustained basis or at all. Our ability to achieve or sustain profitability is based on numerous factors, many of which are beyond our control, including, among other factors, market acceptance of our products, future product development, our market penetration, our margins and our ability to commercialize our pipeline of products. Losses have historically had an adverse effect on our working capital, total assets and shareholders’ equity, and expected future losses may continue to have an adverse effect on our working capital, shareholders’ equity, and the price of the Class A Ordinary Shares and the Warrants. Our failure to achieve and sustain profitability in the future would negatively affect our business, financial condition, results of operations and cash flows, and could cause the market price of the Class A Ordinary Shares and the Warrants to decline.
We have a limited history introducing new products and services to our customers. The future prospects of our business may be harmed if our efforts to attract new customers and engage existing customers by introducing new products are unsuccessful.
Our success depends on our ability to continuously attract new customers and retain and engage existing customers. If we are unable to introduce new and enhanced products and services, or if we introduce new products or services that are not favorably received by the market, we may not be able to attract or retain customers.
Our marketing efforts for CircleDNA and IM8 currently include various initiatives and consist primarily of digital marketing on a variety of social media channels, such as YouTube, Instagram, LinkedIn, Facebook, search engine optimization on websites, such as Google and Facebook Ads, various branding strategies, and email. During the fiscal year ended December 31, 2025, we spent $35.5 million on selling and marketing expenses, representing 38.5% of our revenue from continuing operations. We anticipate that sales and distribution expenses will continue to represent a significant percentage of our overall operating costs for the foreseeable future.
We have historically acquired a significant number of customers through digital advertising on platforms and websites owned by Google and Facebook, which may terminate their agreements with us at any time. Our investments in sales and marketing may not effectively reach potential customers and potential customers may decide not to buy our products or services, any of which could adversely affect our financial results.
If we are unable to attract new customers or engage existing customers either by introducing new products and services or through marketing efforts, our revenue and operating results may grow slower than expected or decline.
Our CircleDNA and IM8 businesses depend on our ability to maintain a strong base of engaged customers and content creators, including through the use of social media. We may not be able to maintain and enhance our brand if we experience negative publicity related to our marketing efforts or use of social media, fail to maintain and grow our network of content creators, or otherwise fail to meet our customers’ expectations.
We currently partner with content creators who help raise awareness of our brands and engage with our customers. Our ability to maintain relationships with our existing content creators and to identify new content creators is critical to expanding and maintaining our customer base. As our market becomes increasingly competitive or as we expand internationally, recruiting and maintaining content creators may become increasingly difficult and expensive. If we are not able to develop and maintain strong relationships with our network of content creators, our ability to promote and maintain awareness of our brands may be adversely affected.
Further, if we incur excessive expenses in this effort, our business, financial condition, and results of operations may be adversely affected. We and our content creators use third-party social media platforms to raise awareness of our brands and engage with our customers. As existing social media platforms evolve and new platforms develop, we and our content creators must continue to maintain a presence on these platforms and establish a presence on emerging popular social media platforms. If we are unable to cost-effectively use social media platforms as marketing tools, our ability to acquire new customers and our financial condition may suffer. Furthermore, as laws and regulations governing the use of these platforms evolve, any failure by us, our content creators, our sponsors, or other third parties acting at our direction, to abide by applicable laws and regulations in the use of these platforms could subject us to regulatory investigations, class action lawsuits, liability, fines, or other penalties and adversely affect our business, financial condition, and results of operations.
In addition, an increase in the use of social media for product promotion and marketing may cause an increase in the burden on us to monitor such materials, and increase the risk that such materials could contain problematic product or
12
marketing claims in violation of applicable regulations. For example, in some cases, the Federal Trade Commission (the “FTC”), has sought enforcement action against parties whose use of an endorsement has failed to clearly and conspicuously disclose a financial relationship or material connection between a social media content creator and an advertiser.
We also do not prescribe what content creators post on social media, and our content creators could engage in behavior or use their platforms in a manner that reflects poorly on our brands or is in violation of applicable regulations or platform terms of service, and all these actions may be attributed to us. Negative commentary regarding us, our products, our content creators, or other third parties, whether accurate or not, may be posted on social media platforms at any time and may adversely affect our reputation, brand, and business. The harm may be immediate, without affording us an opportunity for redress or correction and could adversely affect our business, financial condition, and results of operations.
In addition, customer complaints or negative publicity related to our website, products, product delivery times, customer data handling, marketing efforts, data privacy or security practices, or customer support, especially on blogs and social media websites, could diminish customer loyalty and customer engagement.
Further, laws and regulations, including associated enforcement priorities, rapidly evolve to govern social media platforms and other internet-based communications, and any failure by us, our ambassadors, or other third parties acting at our direction or on our behalf, to abide by applicable laws and regulations in the use of these platforms could subject us to regulatory investigations, class action lawsuits, liability, fines, or other penalties. Other risks associated with the use of social media and internet-based communication include improper disclosure of proprietary information, negative comments about our brand or products, exposure of confidential or personal information, fraud, hoaxes, or malicious dissemination of false information. Damage to our brand image and our reputation could have an adverse effect on our business, financial condition, and results of operations.
We have acquired digital assets in connection with our previous treasury strategy, which exposes us to financial and regulatory risks.
Between June and December 2025, we pursued a digital asset treasury strategy under which we acquired approximately 510 Bitcoin as of the date of this Annual Report. On December 4, 2025, we ceased all further digital asset acquisitions, and on December 30, 2025, the board of directors determined that the Company would not allocate any existing or new capital for the purpose of acquiring additional digital assets. We retain our existing digital asset holdings as a long-term treasury reserve asset. As of December 31, 2025, the fair value of our digital asset holdings represented a significant portion of our total assets.
The price of digital assets has historically been subject to dramatic fluctuations and is highly volatile. A significant decline in the market price of our digital assets could have a material adverse effect on the carrying value of our digital asset holdings, our financial condition, results of operations and the trading price of our Class A Ordinary Shares.
Under IFRS Accounting Standards, we account for our digital asset holdings as intangible assets using the revaluation model. Under this model, increases in fair value are generally recognized in other comprehensive income and accumulated in equity as a revaluation surplus, except to the extent they reverse previously recognized decreases in profit or loss. Decreases in fair value are generally recognized in profit or loss, except to the extent they offset any existing revaluation surplus for the same asset.
Accordingly, fluctuations in the price of Bitcoin may introduce significant volatility to our profit or loss, other comprehensive income and equity that are unrelated to the performance of our core consumer health business.
A material decline in the price of our digital assets in any reporting period would result in significant unrealized losses in profit or loss, which could adversely affect investor perception of our financial performance, even if our operating business is performing well. Conversely, any gains recognized in one period may not be sustained in subsequent periods.
Bitcoin is a relatively novel asset class, and our use of Bitcoin as a long-term treasury reserve asset is subject to a number of risks and uncertainties, including but not limited to the following:
•Regulatory uncertainty. The regulatory framework governing digital assets is evolving and varies across jurisdictions. Government authorities around the world, including in the United States, the Cayman Islands, Hong Kong and other jurisdictions in which we operate, may adopt laws, regulations or directives that adversely affect the use, transfer, exchange, value or holding of digital assets. Regulatory actions could restrict our ability to hold digital assets, require us to divest our holdings, impose additional reporting, tax or compliance obligations, or create an adverse environment that negatively impacts the value of our digital assets. There can be no assurance that future regulatory developments will not have a material adverse effect on the value of our digital asset holdings or our ability to retain them.
13
•Custody and security risks. Our digital assets are held in custody by a third-party custodian. Despite our use of institutional-grade custodial arrangements, there is a risk that our digital assets could be lost, stolen or destroyed through cyberattacks, security breaches, unauthorized access, fraud, technical failures or other events beyond our control. There may be limited or no recovery available in the event of such a loss. Unlike cash deposits at banking institutions, digital assets held in custodial accounts are generally not protected by deposit insurance or similar protections. Any security breach or loss of our digital assets could have a material adverse effect on our financial condition.
•Limited operating history and precedent. Holding digital assets as a corporate treasury reserve asset is a relatively recent practice, and there is limited historical precedent for the long-term performance of such strategies. Our decision to retain digital assets as a treasury reserve asset may not generate the returns or provide the inflation protection anticipated by our management. The performance of digital assets relative to other assets that we could hold in our treasury, such as cash, money market instruments or government securities, is uncertain, and past performance of digital assets is not indicative of future results.
•Liquidity and market disruption. Although digital assets are currently traded on a number of digital asset exchanges worldwide, there is no assurance that a liquid market for digital assets will be maintained. Digital asset exchanges and trading platforms have in the past experienced significant outages, delays, fraud, security breaches and closures. If the exchanges or platforms through which we may seek to sell our digital assets were to become unavailable or experience disruptions, or if market liquidity were to decline significantly, we may be unable to sell our digital assets at favorable prices or at all when we desire to do so.
•Taxation. The tax treatment of digital assets is uncertain and evolving. Changes in tax laws, regulations or interpretations in the Cayman Islands, Hong Kong, the United States or other jurisdictions in which we operate could adversely affect the tax consequences of holding, transferring or disposing of digital assets. In particular, the tax treatment of unrealized gains on our digital asset holdings may vary by jurisdiction and could result in additional tax liabilities. Any increase in our tax liabilities related to our digital asset holdings could adversely affect our financial condition and results of operations.
We rely on a limited number of suppliers, manufacturers, distributors and other service providers, and may not be able to find replacements or immediately transition to alternatives, which could adversely affect our ability to meet customer demand.
We rely on a limited number of suppliers for materials, manufacturers, distributors and genome sequencing service providers. We do not have long-term agreements with most of our suppliers, and our suppliers could cease supplying these materials and services at any time, or fail to provide us with sufficient quantities of materials or services that meet our specifications or that are satisfactory to us. Obtaining substitute materials and services could be difficult, time-consuming and costly and it could require us to redesign our products or revalidate our test kits. Our laboratory operations could be interrupted if we encounter delays or difficulties in securing reagents, sequencers or other equipment or materials, and if we cannot timely obtain acceptable substitutes such interruptions could significantly affect our ability to develop, distribute and commercialize our products and could adversely affect our ability to meet customer demand.
We rely entirely on third-party manufacturers, suppliers and distribution partners for the production, quality testing, fulfillment and delivery of our products, and do not maintain or plan to develop in-house manufacturing or distribution capability. Our IM8 products are manufactured by a limited number of nutraceutical manufacturers in FDA-registered facilities in the United States and undergo independent third-party testing through NSF Certified for Sport. While the principal raw materials used in our products are generally available from multiple suppliers and we have not historically experienced material difficulty in sourcing required quantities, any failure by our contract manufacturers or logistics partners to meet our quality specifications, production timelines or delivery requirements could disrupt our operations and adversely affect our ability to fulfill customer orders. Although we have adopted a diversification approach by engaging manufacturers and suppliers across different countries and regions, and we believe that alternative partners with comparable capabilities are available in our principal markets, any transition to new partners may require significant time and resources, during which our business, reputation and results of operations could be adversely affected.
Although we maintain relationships with suppliers with the objective of ensuring that we have adequate supply for the delivery of our products and services, increases in demand for our products and services can result in supply shortages and higher costs. Our suppliers may not be able to meet our delivery schedules or performance and quality specifications, and we may not be able to purchase such items at a competitive cost. Further, we may experience shortages in certain items as a result of limited availability, increased demand, pandemics or other outbreaks of contagious diseases, weather conditions and natural disasters, as well as other factors outside of our control. In addition, our freight costs may increase due to factors such as limited carrier availability, increased fuel costs, increased compliance costs associated with new or
14
changing government regulations, pandemics or other outbreaks of contagious diseases, and inflation. Furthermore, the prices charged for our products may not reflect changes in our packaging material, freight, tariff and energy costs at the time they occur, or at all. Any of the foregoing risks, if they occur, could have a material adverse effect on our business, financial condition and results of operations.
Our operating results may fluctuate significantly, which makes our future operating results difficult to predict and could cause our operating results to fall below expectations.
Our quarterly and annual operating results may fluctuate significantly, which makes it difficult for us to predict our future operating results. These fluctuations may occur due to a variety of factors, many of which are outside of our control, including, but not limited to: (i) the level of demand for our products; (ii) the timing and cost of, and level of investment in, research, development, manufacturing, regulatory approval and commercialization activities relating to our products, which may change from time to time; (iii) sales and marketing efforts and expenses; (iv) the rate at which we grow our sales force and the speed at which newly hired salespeople become effective; (v) changes in the productivity of our sales force; (vi) positive or negative coverage in the media or clinical publications of our products; (vii) the cost of manufacturing our products, which may vary depending on the quantity of production and the terms of our arrangements with our suppliers; (viii) introduction of new or enhanced products or technologies by us or others in our industries and markets; (ix) pricing pressures; (x) expenditures that we may incur to acquire, develop or commercialize products; (xi) the degree of competition in, and any change in the competitive landscape of, our industries and markets; (xii) changes in governmental regulations or in the status of our regulatory approvals or requirements; (xiii) future accounting pronouncements or changes in our accounting policies; and (xiv) general market conditions and other factors, including factors unrelated to our operating performance or the operating performance of our competitors.
The cumulative effects of factors discussed above and other factors could result in large fluctuations and unpredictability in our quarterly and annual operating results. As a result, comparing our operating results on a period-to-period basis may not be meaningful. Investors should not rely on our past results as an indication of our future performance. This variability and unpredictability could also result in our failure to meet the expectations of industry or financial analysts or investors for any period, which in turn could have a material adverse effect on our business and prospects, and the market price of the Class A Ordinary Shares and the Warrants.
Our business significantly depends upon the strength of our brands, including Prenetics, CircleDNA and IM8, and any harm to our brands or reputation may materially and adversely affect our business and results of operations.
We believe that the brand identity that we have developed has significantly contributed to the success of our business. It is critical that we continue to maintain and enhance the recognition and reputation of our brands.
Many factors, some of which are beyond our control, are important to maintaining and enhancing our brands and if not properly managed, may cause material harm to our brands. These factors include our ability to: (i) provide effective, safe, accurate and user-friendly health supplement products and testing services to customers; (ii) maintain the efficiency, reliability and quality of the health supplement products and testing services we provide to our consumers; (iii) maintain or improve consumer satisfaction with our after-sale services; (iv) increase brand awareness through marketing and brand promotion activities; and (v) preserve our reputation and goodwill in the event of any negative publicity on our services, product quality, price, data privacy and security, our industry and other players within the industry, or other issues affecting us or our peers.
If our products and services are perceived by the public to be of poor quality or if our test kits are perceived to provide inaccurate results or significantly delayed responses, such perception, even if factually incorrect or based on isolated incidents, could damage our reputation, diminish the value of our brand, undermine the trust and credibility we have established and have a negative impact on our ability to attract new clients and customers or retain our current clients and customers. If we fail to promote and maintain our brands including “Prenetics,” “CircleDNA,” “IM8,” or if we incur excessive expenses in this effort, our business, operating results and financial condition may be materially and adversely affected. We anticipate that, as the market becomes increasingly competitive, maintaining and enhancing our brands may become increasingly difficult and expensive.
In addition, we have entered into brand ambassadorship and partnership arrangements with a number of high-profile athletes and public figures, including our co-founding partner David Beckham, World No. 1 tennis player Aryna Sabalenka, Formula 1 driver Ollie Bearman and NBA champion Giannis Antetokounmpo, among others. These individuals
15
are closely associated with our IM8 brand and feature prominently in our marketing campaigns and public communications. The actions and conduct of these individuals are outside of our control. If any of our brand ambassadors or partners were to engage in conduct that is, or is perceived to be, illegal, unethical, socially objectionable or otherwise inconsistent with the values associated with our brands, or if they become the subject of allegations of misconduct, criminal proceedings, regulatory investigations, personal controversies or other reputational issues, whether or not such matters are ultimately substantiated, the negative publicity could adversely affect public perception of our brands. Furthermore, the termination or non-renewal of any such partnership, whether initiated by us in response to reputational concerns or by the ambassador for other reasons, could reduce the effectiveness of our marketing efforts, require us to incur costs to transition to alternative brand strategies, and result in the loss of the commercial benefits associated with such partnerships. Any of the foregoing could harm our brand image, reduce consumer trust, and materially and adversely affect our business, financial condition and results of operations.
If we cannot provide quality technical and customer and user support, we could lose customers, and our business and prospects may be adversely affected.
The provision of our products and services to our customers requires ongoing customer and user support and therefore recruitment, training and retention of technical, customer and user support teams. Hiring technical and customer and user support personnel is very competitive in the industry due to the limited number of people available with the necessary scientific and technical backgrounds and ability to understand our platform, products and services at a technical level. To effectively support potential new customers and ultimately users, we will need to substantially develop a technical and customer and user support staff. If we are unable to attract, train or retain the number of qualified technical and customer and user support personnel sufficient to meet our business needs, our business and prospects will suffer.
If we are unable to successfully expand our sales and marketing infrastructure to match our growth, our business may be adversely affected.
We currently have a limited sales and marketing organization and limited experience distributing preventive or consumer health products. Our future growth depends on scaling this infrastructure, including hiring, training, and retaining qualified personnel, which requires significant time and resources. New sales representatives may take time to become effective, and underperformance could delay revenue generation and diminish returns on our investment.
We face risks in both building in-house capabilities and outsourcing to third parties. Establishing a sales force internally is costly and time-consuming, and could result in premature commercialization expenses if product launches are delayed. Relying on third-party providers may limit our control over sales execution, and such partners may fail to adequately promote our products.
Our brand-building efforts, including celebrity endorsements and digital outreach, are also at an early stage. While we believe these channels are important to customer engagement, we have limited experience in implementing them, and there is no assurance they will lead to sustained revenue growth or justify associated marketing costs.
The launch of our new nutrient products under the IM8 brand may face challenges that could impact our results of operations and reputation.
The launch of our new nutrient products under the IM8 brand carries several risks that could negatively impact our results of operations and brand reputation. These include challenges in achieving market acceptance as consumer demand may fall short of expectations. Additionally, delays in obtaining regulatory approvals or meeting compliance requirements could hinder our products’ launch in key markets. Supply chain disruptions, production delays, or quality control issues could further delay market entry or lead to shortages and customer complaints, affecting both sales and brand perception.
The competitive nature of the nutrient product market also presents risks, as established competitors may introduce similar or superior products, making it difficult for IM8 to gain market traction. Moreover, the success of IM8 depends heavily on effective marketing and the public perception of our brand, and failure in these areas could harm consumer trust and loyalty. The development of our IM8 business may also strain our financial and operational resources, with significant upfront costs related to marketing, R&D, and distribution. If IM8 fails to meet revenue expectations, it could result in financial losses and hinder our long-term success.
16
The consumer genetic testing market is highly competitive, and many of our competitors are more established and have stronger marketing capabilities and greater financial resources, which presents a continuous threat to the success of our consumer genetic testing business.
We operate a consumer genetic testing business primarily through our CircleDNA product line. Consumer genetic testing is a rapidly growing market, and the number of companies with products and technologies similar to CircleDNA continues to increase.
We anticipate facing competition. Our ability to compete depends upon a number of factors both within and beyond our control, including, among others, the following: (i) the quality and reliability of our and others’ products; (ii) accessibility of results; (iii) turnaround time of testing results; (iv) price; (v) convenience and ease of use; (vi) selling and marketing efforts; (vii) additional value-added services and health informatics tools; (viii) customer service and support efforts; (ix) adaptability to evolving regulatory landscape; (x) the ability to execute strategies to protect data privacy and build customer trust; and (xi) our brand recognition relative to our competitors.
We also face competition from other companies attempting to enter the genetic testing market and capitalize on similar opportunities. Our competitors may develop products or services that are similar to or that achieve greater market acceptance than our offerings, and we may not be able to compete effectively against these organizations.
If we fail to compete successfully against our current and future competitors, we may be unable to increase sales revenue and market share, improve our results of operations, or achieve profitability.
We face risks associated with the acquisition and divestiture of businesses.
We have expanded our products and markets in part through strategic acquisitions, joint ventures and equity investments and may continue to do so in the future, depending on our ability to identify and successfully pursue suitable candidates and partners. In addition, we have made certain cash investments into investment funds.
Acquisitions involve numerous risks, including risks inherent in entering new markets in which we may not have prior experience; potential loss of significant customers or key personnel of the acquired business; not obtaining the expected benefits of the acquisition on a timely basis or at all; managing operations in new geographies; and potential diversion of management’s attention from other aspects of our business operations. Acquisitions may also cause us to incur debt or result in dilutive issuances of our equity securities, write-offs of goodwill and substantial amortization expenses associated with other intangible assets. We may not be able to obtain financing for future acquisitions on favorable terms, making any such acquisitions more expensive. Any such financing may have terms that restrict our operations. We may not be able to successfully integrate the operations of any acquired businesses into our operations and achieve the expected benefits of any acquisitions, and certain acquisitions or divestitures may not have the desired effect of enhancing our business and operations. Our acquisitions and divestitures could in the future result in exposure to contingent or unexpected liabilities, such as litigation.
Our acquisitions may not be accretive to our earnings per share. Our expectations regarding the timeframe in which a potential acquisition may become accretive to our earnings per share may not be realized. In addition, we could fail to realize all of the benefits anticipated in a potential acquisition or experience delays or inefficiencies in realizing such benefits. Such factors could, combined with the potential issuance of our ordinary shares in connection with a potential acquisition, result in such acquisition being dilutive to our earnings per share, which could negatively affect the market price of our ordinary share.
When we acquire businesses, products or technologies, our due diligence reviews are subject to inherent uncertainties and may not reveal all potential risks. Incomplete or inaccurate information could result in financial losses, and our auditor may face challenges in evaluating investment valuations due to limited transparency into underlying assets. We may fail to discover or may inaccurately assess undisclosed or contingent liabilities, including liabilities for which we may have responsibility as a successor to the seller or the target company. As a successor, we may be responsible for any past or continuing violations of law by the seller or the target company. Although we generally attempt to seek contractual protections, such as representations and warranties and indemnities, we cannot be sure that we will obtain such provisions in our acquisitions and divestitures or that such provisions will fully protect us from all unknown, contingent or other liabilities or costs. In addition, claims relating to any acquisition or divestiture may necessitate our seeking or defending claims for which the seller or the buyer may not indemnify us or that may exceed the scope, duration or amount of the seller’s or buyer’s indemnification obligations.
17
We may not consummate a potential acquisition for a variety of reasons, but still incur material costs in connection with an acquisition that we cannot recover. The failure to successfully integrate newly acquired businesses or achieve the expected benefits of strategic acquisitions in the future, or consummate a potential acquisition after incurring material costs, could have an adverse effect on our business, results of operations and financial position.
Joint ventures could also present potential additional risks to our business, including risks inherent in taking on a partner which limits our ability to make decisions in our strategy and operations unilaterally and differences in culture, experience and expectations could adversely affect our ability to work successfully with our partners. In growing our business, we also have made and may continue to make equity investments in other companies to provide greater opportunities for business collaboration, operational efficiency and financial gain. These equity investments may pose a risk to our business, if the companies we invest in have financial losses, suffer from market volatility, and become subject to new regional laws and regulations.
In addition, we have divested, and may divest in the future, businesses, brands and assets as part of ongoing efforts to refine our portfolio and redefine our strategic priorities. These divestitures may adversely affect our business, results of operations or financial condition if we are unable to offset the dilutive impacts from the loss of revenue associated with the divested businesses, brands or assets or otherwise achieve the anticipated benefits or cost savings from the divestitures. Furthermore, businesses, brands or assets under consideration for, or otherwise subject to, divestiture may be adversely impacted prior to completion of the divestiture, which could adversely affect our business, results of operations or financial condition.
We are highly dependent on our senior management team and key advisors and personnel, and our business and operating results could be harmed if we are unable to retain senior management and key personnel and to attract and retain qualified personnel necessary for our business.
We are highly dependent on our senior management team and key advisors and personnel. Our success will depend on our ability to retain senior management and to attract and retain qualified advisors and personnel in the future, including sales and marketing professionals and other highly skilled personnel and to integrate current and additional personnel in all departments. To induce valuable employees to remain at our Company, in addition to salary and cash incentives, we have issued, and will in the future issue, equity incentive awards that vest over time. The value to employees of such equity incentive awards that vest over time may be significantly affected by movements in our share price which is beyond our control, and may at any time be insufficient to counteract more lucrative offers from other companies. Despite our efforts to retain valuable employees, members of our management and development teams may terminate their employment with us on relatively short notice, even where we have employment agreements in place. The standard employment agreement of our employees provides that the employee can terminate the employment by giving at least one month’s notice or payment in lieu of notice, which means that any of our employees could leave their employment at any time on relatively short notice or without notice at all. We also do not maintain “key person” insurance policies on the lives of these people or the lives of any of our other employees. The loss of members of our senior management, sales and marketing professionals and scientists, as well as contract employees could result in delays in product development and harm our business. In particular, the loss of the services of Mr. Danny Yeung, our Director, Chairperson and Chief Executive Officer, could significantly delay or prevent the achievement of our strategic objectives and otherwise have a material adverse impact on our business. If we are not successful in attracting and retaining highly qualified personnel, our business, financial condition and results of operations will be negatively impacted.
Competition for skilled personnel across virtually all areas where we operate and need to attract additional talent is intense. If we are not successful in attracting and retaining highly qualified personnel, the rate and success at which we can develop and commercialize our products will be limited, and our business, financial condition and results of operations would be negatively impacted.
In addition, we rely on our scientific advisory board comprised of accomplished scholars and experts to offer invaluable insights on the latest scientific developments and provide guidelines on development of our pipeline products. If any of our scientific advisors leaves the advisory board, our research and development capabilities may be negatively affected.
18
The sizes of the markets and forecasts of market growth for the demand of our current and pipeline products and services are based on a number of complex assumptions and estimates that are subject to change, and may be inaccurate.
Our estimates of the total addressable markets for our products and services, including CircleDNA and IM8 are based on a number of internal and third-party estimates. Market opportunity estimates and growth forecasts included in this annual report have been derived from a variety of sources, including market research and our own internal estimates, and the conditions supporting our assumptions or estimates may change at any time.
Our market opportunity may also be limited by new services and products that enter the market. If any of our estimates prove to be inaccurate, the market opportunity for our existing and pipeline products and services could be significantly less than we estimate. If this turns out to be the case, it may impair our potential for growth and our business and future prospects may be materially and adversely affected.
We may need to raise additional funds to develop our platform, commercialize new products or expand our operations, and we may be unable to raise capital when needed, or on acceptable terms, or at all.
We may seek additional funding through equity or debt offerings, credit facilities, or other third-party arrangements to support regulatory approvals, product development, commercialization, and operational growth. Our funding needs may arise sooner or in greater amounts than anticipated, particularly if revenue falls short or regulatory timelines shift.
Our future capital requirements will depend on factors including the cost and timing of regulatory approvals, market adoption, clinical trial progress, intellectual property protection, and strategic collaborations. Raising capital through equity may dilute existing shareholders and involve securities with rights senior to ordinary shares. Debt financing may impose restrictive covenants, while strategic partnerships may require us to grant rights or revenue participation on unfavorable terms.
There is no guarantee additional funding will be available when needed or on acceptable terms, or at all. If we cannot secure sufficient capital, we may need to scale back or delay development, commercialization, or operational initiatives. Deterioration in global economic or financial conditions could further limit our financing options, which may materially impact our business, financial condition, and results of operations.
We may incur debt or assume contingent or other liabilities or dilute our shareholders in connection with acquisitions or strategic alliances.
We may issue equity securities to pay for future acquisitions or strategic alliances, which could be dilutive to existing shareholders. We may incur debt or assume contingent or other liabilities in connection with acquisitions and strategic alliances, which could impose restrictions on our business operations and harm our operating results. Further, any additional equity financing, debt financing, or credit facility used for such acquisitions may not be on favorable terms, and any such financing or facility may place restrictions on our business. In addition, to the extent that the economic benefits associated with any of our acquisitions diminish in the future, we may incur incremental operating losses, and may be required to record additional write downs of goodwill, intangible assets or other assets associated with such acquisitions, which would adversely affect our operating results.
If we fail to implement and maintain an effective system of internal controls in the future, we may be unable to accurately report our financial condition or results of operations, which may adversely affect investor confidence in us and, as a result, the market price of the Class A Ordinary Shares and the Warrants.
We are subject to Section 404 of the Sarbanes-Oxley Act of 2002, or Section 404, which require that we include a report from management on our internal control over financial reporting in our annual report on Form 20-F. In addition, once we cease to be an “emerging growth company” as such term is defined in the Jumpstart Our Business Startups Act of 2012, or JOBS Act, and if we are not a non-accelerated filer by then, our independent registered public accounting firm must attest to and report on the effectiveness of our internal control over financial reporting. Our management may conclude that our internal control over financial reporting is not effective. Moreover, even if our management concludes that our internal control over financial reporting is effective, our independent registered public accounting firm, after conducting its own independent testing, may issue a report that is qualified if it is not satisfied with our internal controls or
19
the level at which our controls are documented, designed, operated, or reviewed, or if it interprets the relevant requirements differently from us. We may be unable to timely complete the evaluation testing and any required remediation.
Our management conducted an evaluation of the effectiveness of our internal control over financial reporting and concluded that our internal control over financial reporting was effective as of December 31, 2025. However, there is no assurance that we will not have any material weakness or deficiencies in the future. Even effective internal control can provide only reasonable assurance with respect to the preparation and fair presentation of financial statements. Any failure to remediate the deficiencies, or the development of new deficiencies or material weaknesses in our internal control over financial reporting, could result in material misstatements in our financial statements, which in turn could have a material adverse effect on our financial condition.
Ineffective internal control over financial reporting could expose us to increased risk of fraud or misuse of corporate assets or inaccurate reporting of financial conditions and results of operations and subject us to potential delisting from the stock exchange on which we are listed, regulatory investigations and civil or criminal sanctions. We may also be required to restate our financial statements from prior periods. If we fail to achieve and maintain an effective internal control environment, we could suffer material misstatements in our financial statements and fail to meet our reporting obligations, which would likely cause investors to lose confidence in our reported financial information. This could in turn limit our access to capital markets, result in deterioration in our financial condition and results of operations, and lead to a decline in the market price of the Class A Ordinary Shares and the Warrants.
We depend on the information systems of our own and those of third parties for the effective service on our websites, mobile applications, and in our computer and logistics systems, and for the overall effective and efficient functioning of our business. Failure to maintain or protect our information systems and data integrity effectively could harm our business, financial condition and results of operations.
We rely on both internal and third-party information systems to operate our websites, manage logistics, manufacture and distribute products and services, and support critical functions such as accounting, inventory, and compliance. Any system failure—whether due to cyberattacks, software malfunctions, human error, or natural disasters—could disrupt operations, compromise data integrity, or expose confidential information.
Cybersecurity threats are becoming more sophisticated and prevalent. Unauthorized access, data breaches, or theft of intellectual property could lead to costly remediation efforts, reputational harm, and business disruption. Similar risks apply to our collaborators, suppliers, and service providers.
Our operations also depend on reliable transportation and logistics. Delays, losses, or damages—particularly during peak periods or due to external events like pandemics, geopolitical unrest, or natural disasters—could impact our ability to deliver or process products and services, resulting in lost revenue and customer dissatisfaction.
Our insurance coverage may be limited and not sufficient to cover all potential losses. Maintaining and protecting our systems requires ongoing investment, and any failure to do so could materially affect our business and financial performance.
Risks Relating to Doing Business in Hong Kong
We are subject to risks associated with our operations in Hong Kong, a Special Administrative Region of China.
•We currently have no business operations in mainland China and do not utilize Variable Interest Entity (“VIE”) structures. Our operations are based in Hong Kong as a Special Administrative Region of China, which subjects us to a different regulatory framework than mainland Chinese companies. However, our Hong Kong operations expose us to certain legal and regulatory risks related to China.
•Special Administrative Region Status: While Hong Kong operates under the "One Country, Two Systems" framework with its own legal and regulatory system, the Chinese government maintains significant oversight authority over Hong Kong. Changes in PRC policies or their application to Hong Kong could materially impact our business operations, regulatory compliance requirements, and ability to operate under the current legal framework.
•Potential Regulatory Extension: Although existing PRC regulations such as the Data Security Law, Cybersecurity Law, and Personal Information Protection Law do not currently apply directly to our Hong Kong operations, any future extension of these or similar regulations to Hong Kong could subject us to new
20
compliance obligations. This could potentially affect our ability to operate efficiently, accept foreign investment, or maintain our U.S. listing.
•Geopolitical Risks: Evolving U.S.-China relations and related policies may create additional regulatory complexities for Hong Kong-based companies like ours. Changes in trade policies, sanctions, or other geopolitical developments could affect our operations, market access, or ability to maintain our U.S. listing.
Since Hong Kong is a special administrative region of China, legal and operational risks associated with operating in China also apply to operations in Hong Kong and could materially and adversely affect our business and results of operations.
Prenetics, the holding company, does not conduct operations in China, though certain of the group’s subsidiaries conduct operations in Hong Kong, a special administrative region of the PRC. As a result, we face various legal and operational risks and uncertainties relating to our operations in Hong Kong, and from the evolving regulatory landscape in the PRC generally. As we presently do not have any business operations in mainland China, either directly or through VIE arrangements, we consider that the current laws and regulations of the PRC applicable in mainland China have no material impact on our business, financial condition or results of operations. However, since Hong Kong and Macau are special administrative regions of China, the legal and operational risks associated with operating in China also apply to operations in Hong Kong and Macau.
The PRC government has recently indicated an intent to exert more oversight and control over offerings conducted overseas (including those using entity VIE structures) and foreign investment in China-based companies. This has been evidenced by its probe into several U.S.-listed technology companies with operations in mainland China, focusing on anti-monopoly, financial technology regulation and more recently, with the passage of the PRC Data Security Law, how companies collect, store, process and transfer personal data. Currently, these laws and regulations are expected to apply to mainland China domestic businesses, rather than businesses in Hong Kong which operate under a different set of laws from mainland China. However, there can be no assurance that the PRC government will not, in the future, expand the application of such regulations, or adopt new laws, regulations, or policies, that could apply to our operations in Hong Kong, or that the government of Hong Kong may enact similar laws and regulations applicable to companies operating in Hong Kong.. Should the PRC government seek to affect operations of any company with any level of operations in Hong Kong, should certain PRC laws and regulations or these statements or regulatory actions become applicable to us in the future, or should the government of Hong Kong enact similar laws and regulations, it would likely have a material adverse impact on our business, financial condition and results of operations, ability to accept foreign investments and our ability to offer or continue to offer securities to investors in the United States or to list on a U.S. or other international securities exchange, any of which may cause the value of our securities to significantly decline or become worthless. For example, if the recent PRC regulatory actions on data and cyberspace security were to apply to us, including our operations in Hong Kong or Macau, we could become subject to certain cybersecurity and data privacy obligations, including the potential requirement to conduct a cybersecurity review for our listing or continued listing on a U.S. or a foreign stock exchange, and the failure to meet such obligations could result in penalties and other regulatory actions against us and may materially and adversely affect our business and results of operations. Regulatory actions related to data security or anti-monopoly concerns in Hong Kong or Macau may also impact our ability to conduct our business, accept foreign investments, or continue to be listed or list on a U.S. or foreign stock exchange.
Unfavorable economic and political conditions in Hong Kong and other parts of Asia could materially and adversely affect our business, financial condition, and results of operations.
Like many other companies that operate in Asia, our business will be materially affected by economic and political conditions in Asia, which could be negatively impacted by many factors beyond our control, such as inability to access capital markets, control of foreign exchange, changes in exchange rates, rising interest rates or inflation, slowing or negative growth rates, government involvement in allocation of resources, inability to meet financial commitments in a timely manner, terrorism, political uncertainty, epidemics or pandemics, civil unrest, fiscal or other economic policy of governments, and the timing and nature of any regulatory reform. Geopolitical uncertainties may also give rise to uncertainties in global economic conditions and adversely affect general investor confidence.
Political unrest such as protests or demonstrations could disrupt economic activities and adversely affect our business. There can be no assurance that these protests and other economic, social, or political unrest in the future will not have a material adverse effect on our financial conditions and results of operations.
21
The mainland Chinese government has significant oversight, discretion and control over the manner in which companies incorporated under the laws of mainland China must conduct their business activities, but as we operate in Hong Kong and not mainland China, the mainland Chinese government currently does not exert direct oversight and discretion over the manner in which we conduct our business activities. However, there is no guarantee that the mainland Chinese government will not seek to intervene or influence our operations at any time. If we were to become subject to such oversight, discretion or control, including over overseas offerings of securities and/or foreign investments, it may result in a material adverse change in our operations, significantly limit or completely hinder our ability to offer or continue to offer securities to investors and cause the value of our securities to significantly decline, or be worthless, which would materially affect the interests of the investors.
We currently do not have any business operations in mainland China or generate revenues from any businesses in mainland China. We believe that the laws and regulations of mainland China do not currently have any material impact on our business operations, and the mainland Chinese government does not currently exert direct influence or intervention over the manner in which we conduct our business. However, if we do decide to expand our operations into mainland China in the future, we could be subject to the significant oversight of the mainland Chinese government. In addition, because of our substantial operations in Hong Kong and given the mainland Chinese government’s significant oversight authority over the conduct of business in Hong Kong generally, there is no guarantee that we will not be subject to such direct influence or intervention in the future due to changes in laws or other unforeseeable reasons. There is always a risk that the mainland Chinese government may, in the future, seek to affect operations of any company with any level of operations in mainland China or Hong Kong, including its ability to offer securities to investors, list its securities on a U.S. or other foreign exchange, conduct its business or accept foreign investment. There also can be no assurance that the PRC government will not intervene or impose restrictions on our ability to transfer or distribute cash within our organization, which could result in an inability or prohibition on making transfers or distributions to entities outside of Hong Kong and adversely affect our business.
The PRC legal system is evolving rapidly and the PRC laws, regulations, and rules may change quickly with little or no advance notice. In particular, because these laws, rules and regulations are relatively new, and because of the limited number of published decisions and the non-precedential nature of these decisions, the interpretation of these laws, rules and regulations may contain inconsistencies, the enforcement of which involves uncertainties.
If we were to become subject to the direct intervention or influence of the mainland Chinese government at any time due to changes in laws or other unforeseeable reasons, it may require a material change in our operations and/ or result in increased costs necessary to comply with existing and newly adopted laws and regulations or penalties for any failure to comply. In addition, the market prices and value of our securities could be adversely affected as a result of anticipated negative impacts of any such government actions, as well as negative investor sentiment towards Hong Kong-based companies subject to direct mainland Chinese government oversight and regulation, regardless of our actual operating performance. There can be no assurance that the mainland Chinese government will not intervene in or influence our current or future operations at any time.
The PRC government has recently indicated an intent to exert more oversight and control over offerings that are conducted in the United States or in other international jurisdictions and/or foreign investment in China-based issuers. Based on the advice of our PRC legal counsel, DaHui Lawyers, we believe that we are currently not required to obtain any permission or approval from the CSRC, CAC or any other PRC governmental authority to operate our business or to list our securities on a U.S. securities exchange or issue securities to foreign investors.
With respect to the issuance of securities to foreign investors, the Regulations on Mergers and Acquisitions of Domestics Enterprises by Foreign Investors (“M&A Rules”) include, among other things, provisions that purport to require any offshore special purpose vehicle that is controlled by PRC companies or individuals and formed for the purpose of seeking a public listing on an overseas stock exchange through acquisition of PRC domestic companies to obtain the approval of the CSRC prior to the listing and trading of its securities on an overseas stock exchange. On September 21, 2006, the CSRC published on its official website procedures specifying documents and materials required to be submitted to it by any such special purpose vehicle seeking CSRC’s approval of overseas listings. However, substantial uncertainty remains regarding the scope and applicability of the M&A Rules and the CSRC approval requirement to offshore special purpose vehicles.
The revised Measures for Cybersecurity Review, or Review Measures, came into effect on February 15, 2022. The Review Measures stipulate that cybersecurity review is mandatory where a network platform operator that has personal information of more than one million users seeks to list overseas. As advised by our PRC legal counsel, DaHui Lawyers,
22
the offering of our securities is not subject to the foregoing cybersecurity review. That said, the Review Measures provide CAC and relevant authorities certain discretion to initiate cybersecurity review where any network product or service or any data handling activity is considered to affect or may affect national security, which may lead to uncertainties in relation to the impact of the Review Measures impact on our operations or the offering of our securities. As of the date of this annual report, there are no commensurate laws or regulations in Hong Kong which result in similar significant oversight over data security for companies seeking to offer securities on a foreign exchange. However, we cannot guarantee that, if, in the future, such laws or regulations were issued in Hong Kong, we would be compliant with such laws or regulations in a timely manner or at all. In addition, we may have to spend significant time and costs to become compliant. If we are unable to do so, on commercially reasonable terms, in a timely manner or otherwise, we may become subject to sanctions imposed by the relevant regulatory authorities, and our ability to conduct our business, or offer securities on a U.S. or other international securities exchange may be restricted. As a result of the foregoing, our business, reputation, financial condition, and results of operations may be materially and adversely affected.
Further, on July 6, 2021, the General Office of the Communist Party of China Central Committee and the General Office of the State Council jointly issued Opinions on Strictly Cracking Down on Illegal Securities Activities in accordance with the Law (“Opinions”). These Opinions have laid the groundwork for strengthening the Chinese government’s monitoring of illegal securities activities in China and the supervision of overseas listings by China-based companies. The Opinions generally provide that existing laws and regulations regarding data security, cross-border data transmission, and the protection of classified information should be further supplemented, and that the PRC government will seek to deepen its cross- border audit supervision cooperation with the regulatory bodies in other countries in law-based and reciprocal manner. On February 17, 2023, CSRC released the Trial Administrative Measures of Overseas Securities Offering and Listing by Domestic Companies and five supporting guidelines (collectively, “New Overseas Listing Rules”), which have come into effect on March 31, 2023. New Overseas Listing Rules stipulate filing requirements for foreign direct or indirect issuance and listing of securities by domestic companies. As advised by our PRC legal counsel, DaHui Lawyers, the offering of our securities is not subject to the New Overseas Listing Rules or filing requirements.
Based on their understanding of the current PRC laws and regulations, our PRC legal counsel, DaHui Lawyers, has advised that we are not required to obtain any prior permission under the M&A Rules or the Opinions from any PRC governmental authorities (including the CSRC) for the offering or listing of our securities in the United States, given that: (a) the CSRC currently has not issued any definitive rule or interpretation concerning whether offerings like ours are subject to the M&A Rules; and (b) we are not controlled by PRC companies or individuals nor formed for the purpose of seeking a public listing on an overseas stock exchange through acquisition of PRC domestic companies. In addition, our PRC legal counsel, DaHui Lawyers, has advised that the offering or listing of our securities is neither subject to the mandatory cybersecurity review under the Review Measures nor the filing requirements under New Overseas Listing Rules.
However, there is no guarantee that this will continue to be the case in relation to the continued listing of our securities on a securities exchange outside of China, or even if such permission is required and obtained, it will not be subsequently denied or rescinded. Any actions by the Chinese government to exert more oversight and control over offerings or listings that are conducted in the United States or in other international jurisdictions (including those by issuers whose primary operations are in Hong Kong) and/or foreign investments in Hong Kong-based issuers could significantly limit or completely hinder our ability to offer or continue to offer securities to investors and cause the value of our securities to significantly decline or be worthless.
Our business, financial condition and results of operations, and/or the value of our securities or our ability to offer or continue to offer securities to investors may be materially and adversely affected to the extent the laws and regulations of mainland China become applicable to us. In that case, we may be subject to the risks and uncertainties associated with the evolving laws and regulations in mainland China, their interpretation and implementation, and the legal and regulatory system in mainland China more generally, including with respect to the enforcement of laws and the possibility of changes of rules and regulations with little or no advance notice.
We conduct our operations primarily through our subsidiaries in Hong Kong and other jurisdictions. For the years ended December 31, 2021, December 31, 2022, December 31, 2023, December 31, 2024, and December 31, 2025 we generated all of our revenue from our businesses outside of mainland China. Moreover, we do not sell any products in mainland China or solicit any customer or collect, host or manage any customer’s personal data in mainland China. Nor do we have access to any personal data of any customer in mainland China that is collected, hosted or managed by our historical minority interest in a genomics business in mainland China. Accordingly, we believe that the laws and regulations of mainland China, including the developments in cybersecurity laws and regulations of mainland China, do
23
not currently have any material impact on our business, financial condition and results of operations or the listing of our securities, notwithstanding the fact that we have substantial operations in Hong Kong.
Pursuant to the Basic Law of the Hong Kong Special Administrative Region (the “Basic Law”), which is a national law of the PRC and the constitutional document for Hong Kong, national laws of the PRC shall not be applied in Hong Kong except for those listed in Annex III of the Basic Law and applied locally by promulgation or local legislation. The Basic Law expressly provides that the national laws of the PRC which may be listed in Annex III of the Basic Law shall be confined to those relating to defense and foreign affairs as well as other matters outside the autonomy of Hong Kong. While the National People’s Congress of the PRC has the power to amend the Basic Law, the Basic Law also expressly provides that no amendment to the Basic Law shall contravene the established basic policies of the PRC regarding Hong Kong. As a result, national laws of the PRC not listed in Annex III of the Basic Law, including the enacted version of PRC Data Security Law, the revised Measures for Cybersecurity Review (the “Review Measures”) issued by the CAC, and the PRC Personal Information Protection Law, do not apply in Hong Kong.
If certain PRC laws and regulations were to become applicable in Hong Kong in the future, the application of such laws and regulations may have a material adverse impact on our business, financial condition and results of operations and our ability to offer or continue to offer securities to investors, any of which may cause the value of our securities to significantly decline or become worthless. In addition, the laws and regulations in the PRC are evolving, and their enactment timetable, interpretation and implementation involve significant uncertainties. To the extent any PRC laws and regulations become applicable to our business, we may be subject to the risks and uncertainties associated with the legal system in the PRC including with respect to the enforcement of laws and the possibility of changes of rules and regulations with little or no advance notice.
Tariffs and trade restrictions on non-U.S. materials could adversely affect our supply chain, costs, and financial performance
We currently source certain packaging materials, ingredients, components, and equipment used in our business and products from suppliers outside the United States, including in China. Recent and proposed U.S. tariffs, including those announced in 2025 targeting imports from China and Hong Kong, could significantly increase the cost of these non-U.S. materials or disrupt our supply chain. These tariffs may increase our operational costs, particularly for materials not manufactured in the United States.
Higher tariffs could lead to increased prices from our suppliers, reduced availability of critical components, or delays in production and delivery, adversely affecting our ability to meet customer demand. While we are exploring mitigation strategies, such as diversifying our supplier base or seeking alternative materials, there is no assurance that these efforts will fully offset the impact. Additionally, retaliatory tariffs from other countries, as seen in prior trade disputes, could further complicate our global supply chain, particularly given our operations in Hong Kong, which is subject to evolving U.S. and Chinese regulatory oversight.
Risks Relating to Government Regulation
Our business collects and processes a large amount of data including personal information, and we will face legal, reputational, and financial risks if we fail to protect our customers’ data from security breaches or cyberattacks. We are also subject to various laws and regulations relating to privacy or the protection or transfer of data relating to individuals, and any change in such laws and regulations or any failure by us to comply with such laws and regulations could adversely affect our business.
We collect, store, and process sensitive information, including personally identifiable data, genetic and health data, payment details, intellectual property, and other proprietary information. This data relates not only to our customers, but also to third parties and internal operations. We manage such data through cloud-based platforms and maintain safeguards, including logical segregation of sensitive customer data from other business systems. However, these measures may not fully eliminate risks such as unauthorized access, data loss, misuse or modification, and failure to protect this information could materially harm our business.
Our systems and those of our third-party vendors and business partners remain vulnerable to cybersecurity threats, including malware, ransomware, denial-of-service attacks, software failures, and human error. Outages, cyberattacks, or technical malfunctions—whether intentional or accidental—could interrupt our operations, result in the unauthorized
24
disclosure of sensitive information, and expose us to liabilities and compliance issues. We have experienced occasional disruptions in the past and expect continued targeting as cybersecurity threats increase in frequency and sophistication. Service outages by cloud or logistics providers, including during peak demand or holidays, could affect our ability to process, deliver, or receive products and services, damage our reputation, and negatively impact customer satisfaction.
Our operations depend on the secure and uninterrupted performance of both internal and external information systems. We use third-party vendors for logistics and transport services, and disruptions to these partners—such as performance failures, data breaches, or supply chain interruptions—could materially impact our business continuity. External events such as pandemics, natural disasters, political unrest, and other unforeseen circumstances may also disrupt operations or reduce our ability to serve customers effectively.
Beyond operational risks, we face substantial and growing legal exposure relating to data privacy and protection. We are subject to a patchwork of laws and regulations across jurisdictions, including the Personal Data (Privacy) Ordinance (PDPO) in Hong Kong, the U.K. General Data Protection Regulation (U.K. GDPR), the U.K. Data Protection Act (U.K. DPA) and the EU GDPR . These frameworks impose detailed obligations on the handling of personal data, including requirements for consent, data security, breach notification, and enhanced protections for “special category” data such as genetic and health information.
Although the PDPO does not specifically regulate genetic data, we remain subject to general requirements regarding the secure handling of personal data, and noncompliance can lead to civil or criminal penalties, including fines and imprisonment. In the U.K. and EU, GDPR enforcement has resulted in substantial fines—up to €20 million or 4% of global revenue—for data controllers and processors. As a company processing special categories of data, we are subject to heightened scrutiny and must maintain rigorous data governance protocols across our services, platforms, and laboratories.
Despite our investment in compliance programs and cybersecurity infrastructure, legal interpretations of privacy laws continue to evolve and vary across regions. Any failure—or perceived failure—to comply with applicable regulations may result in government investigations, enforcement actions, class action lawsuits, or reputational damage. The costs of responding to data breaches, updating systems, and maintaining ongoing compliance may be significant and may divert resources from strategic initiatives.
We may also be required to modify or limit certain operations in response to changing privacy laws, increasing regulatory oversight, or enforcement activity. There is no guarantee we will remain in full compliance with all applicable data protection requirements in each of the jurisdictions where we operate. Any compromise of data integrity, breach of security, or regulatory violation could materially and adversely affect our reputation, customer relationships, financial condition, and business operations.
Our products and services are and will continue to be subject to extensive regulation, compliance of which could be costly and time-consuming or may cause unanticipated delays or prevent the receipt of the required approvals to offer our products and services.
Our CircleDNA products are classified as medical devices, and in addition to our dietary supplement products are subject to rigorous regulatory oversight in the jurisdictions where we operate. These regulations cover a broad range of activities, including product design, testing, labeling, manufacturing, clinical validation, marketing, distribution, post-market surveillance, recalls, and adverse event reporting.
While regulatory approval is not currently required for our products in Hong Kong, expansion into other jurisdictions may necessitate additional clearances. For example, voluntary registration with Hong Kong’s Medical Device Administrative Control System requires detailed evidence of safety and performance. In the U.K. and European Union, in vitro diagnostic (IVD) devices must meet requirements under the EU In Vitro Diagnostic Directive (IVDD), or under U.K. MHRA rules effective since January 2021, which mandate device registration and conformity with safety and performance standards. In the United States, the formulation, manufacturing, packaging, holding, labeling, promotion, advertising, distribution, and sale of our IM8 products are subject to regulation by various federal governmental agencies, including the U.S Food and Drug Administration (the “FDA”). For more information on regulations that our business is or may become subject to, see “Item 4. Information on the Company — B. Business Overview — Government Regulations.”
Failure to obtain required approvals, or to comply with applicable regulations, could result in fines, product recalls, or restrictions on sales. Any quality deficiencies or safety issues could damage our reputation, result in significant financial costs, and materially harm our business and operations.
25
Our nutritional supplement products are affected by extensive regulations and our failure to comply with any regulations could lead to significant penalties or claims, which could materially harm our financial condition and operating results.
Our IM8 products are classified as dietary supplements. In both domestic and foreign markets, the formulation, manufacturing, packaging, labeling, distribution, advertising, importation, exportation, licensing, sale, and storage of our IM8 products are subject to extensive government regulation. These regulations govern, among other things, product formulation, manufacturing practices, labeling, advertising, and safety. Changes in existing laws or regulations, or the introduction of new requirements, could impose additional compliance costs, restrict our ability to market our products, or subject us to enforcement actions, penalties, or product recalls if we fail to comply.
In the United States, the Dietary Supplement Health and Education Act of 1994 (DSHEA) permits dietary supplement manufacturers to make certain structure/function claims but requires that these claims be substantiated and accompanied by disclaimers. Regulatory authorities may challenge our product claims, and any failure to meet substantiation requirements could lead to enforcement actions, including fines, product relabeling, or removal from the market. Furthermore, our operations and those of our manufacturing partners must comply with current Good Manufacturing Practices (cGMPs), which are subject to regular inspections and audits. Non-compliance with cGMPs could lead to production delays, increased costs, or enforcement actions.
In addition to U.S. regulations, our international operations may be subject to varying regulatory frameworks that govern the import, export, marketing, and sale of nutritional supplements. Compliance with these requirements can be complex and costly, and any failure to meet local standards may restrict our ability to expand into new markets or maintain existing ones. If we fail to adequately address these regulatory risks, our ability to commercialize our products, maintain our reputation, and achieve our growth objectives could be materially and adversely affected.
We plan to expand our business and operations internationally to various jurisdictions in which we do not currently operate and where we have limited operating experience, all of which exposes us to business, regulatory, political, operational and financial risk.
One of our key business strategies is to pursue international expansion of our business operations and market our products in multiple jurisdictions.
As a result, we expect that our business will be subject to a variety of risks associated with doing business internationally, including an increase in our expenses and diversion of the management’s attention from other aspects of our business. Accordingly, our business and financial results in the future could be adversely affected due to a variety of factors, including: (i) political, social and/or economic instability; (ii) risks related to governmental regulations in foreign jurisdictions and unexpected changes in regulatory requirements and enforcement; (iii) fluctuations in currency exchange rates; (iv) higher levels of credit risk and payment fraud; (v) burdens of complying with a variety of foreign laws; (vi) complexities and difficulties in obtaining intellectual property protection and reduced protection for intellectual property rights in some countries; (vii) difficulties in staffing and managing global operations and the increased travel, infrastructure and legal compliance costs associated with multiple international locations and subsidiaries; (viii) management of tax consequences and compliance; and (ix) other challenges caused by distance, language, and cultural differences, making it harder to do business in certain international jurisdictions.
In addition, we may be subject to increased regulatory risks and local competition in various jurisdictions where we plan to expand operations but have limited operating experience. Such increased regulatory burden and competition may limit the available market for our products and services and increase the costs associated with marketing the products and services where we are able to offer our products. If we are unable to manage the complexity of global operations successfully, or fail to comply with any of the regulations in other jurisdictions, our financial performance and operating results could suffer.
26
Risks Relating to Intellectual Property and Legal Proceedings
We may be subject to legal proceedings and litigation, which are costly to defend, and adverse publicity about any investigation, litigation, regulatory or legal action against us or our senior management could harm our reputation and business.
We and our management are, and may in the future become, subject to claims and lawsuits relating to intellectual property, consumer protection, privacy, labor and employment, marketing and communications practices, commercial disputes, and other matters. The number and significance of our legal disputes and inquiries have increased as we have grown larger, as our business has expanded in scope and geographic reach, and as our services have increased in complexity.
Moreover, becoming a public company may have raised our public profile, which may result in increased litigation as well as increased public awareness of any such litigation. There is substantial uncertainty regarding the scope and application of many of the laws and regulations to which we are subject, which increases the risk that we will be subject to claims alleging violations of those laws and regulations. In the future, we may also be accused of having, or be found to have, infringed, misappropriated or otherwise violated third-party intellectual property rights.
Regardless of the outcome, legal proceedings can have a material and adverse impact on us due to their costs, diversion of our resources, and other factors. Litigation and other legal proceedings are inherently unpredictable and can result in excessive or unanticipated verdicts and/or injunctive relief that affect how we operate our business. We could incur judgments or enter into settlements of claims for monetary damages or for agreements to change the way we operate our business, or both. We may decide to settle legal disputes on terms that are unfavorable to us. Furthermore, if any litigation to which we are a party is resolved adversely, we may be subject to an unfavorable judgment that we may not choose to appeal or that may not be reversed upon appeal. In addition, the terms of any settlement or judgment in connection with any legal claims, lawsuits, or proceedings may require us to cease some or all of our operations, or pay substantial amounts to the other party and could materially and adversely affect our business, reputation, financial condition and results of operations.
In addition, adverse publicity about regulatory or legal action against us could damage our reputation and brand image, undermine our customers’ confidence and reduce long-term demand for our products and services, even if the regulatory or legal action is unfounded or not material to our operations.
We depend on intellectual property licensed from third parties for development and commercialization of certain products, and the termination of the licenses or other agreements permitting us to use such intellectual property or failure of such third parties to maintain or protect such intellectual property could result in the loss of significant rights by us, which would harm our business.
We depend on intellectual property licensed from third parties for the development and commercialization of our products. The inability to license such intellectual property on favorable terms, including obtaining exclusive rights in relevant jurisdictions, and the termination of such licenses or other agreements permitting us to use such intellectual property, could adversely affect our business.
We rely substantially on our trademarks and trade names. If our trademarks and trade names are not adequately protected, then we may not be able to build name recognition in our markets of interest and our business may be harmed.
We rely substantially upon trademarks and trade names to build and maintain the integrity of our brands. Our registered and unregistered trademarks or trade names may be challenged, infringed, circumvented, declared unenforceable or determined to be violating or infringing on other intellectual property rights. We may not be able to protect or enforce our rights to these trademarks and trade names, which we rely upon to build name recognition among potential partners and customers. Furthermore, our trademark applications may not be approved by the applicable trademark authority in the markets in which we operate. Our trademarks, including our registered trademarks, could also be the subject of challenges by third parties. In the event that our trademarks are successfully challenged, we could be forced to rebrand our products, which could result in loss of brand recognition, and could require us to devote resources to advertising and marketing new brands. In addition, there could be potential trade name or trademark infringement or dilution claims brought by owners of other trademarks against us. Further, at times, competitors or other third parties may adopt trade names or trademarks
27
similar to those of us, thereby impeding our ability to build brand identity and possibly leading to market confusion. Asserting claims against such third parties may be prohibitively expensive. Over the long term, if we are unable to establish name recognition based on our trademarks and trade names, we may not be able to compete effectively and our business may be adversely affected. Any of our efforts to enforce or protect our proprietary rights related to trademarks, trade secrets, domain names or other intellectual property may be ineffective, could result in substantial costs and diversion of resources, and could harm our business, financial condition and results of operations.
Risks Relating to Our Securities
The trading prices of our Class A Ordinary Shares and Warrants may be volatile and a market for our Class A Ordinary Shares and Warrants may not develop, which would adversely affect the liquidity and price of our Class A Ordinary Shares.
The trading prices of Class A Ordinary Shares and Warrants may be volatile and may fluctuate due to a variety of factors, some of which are beyond our control, including, but not limited to: (i) changes in the sectors in which we operate; (ii) changes in its projected operating and financial results; (iii) changes in laws and regulations affecting our business; (iv) ability to continue to innovate and bring products to market in a timely manner; (v) changes in our senior management team, our board of directors or key personnel; (vi) our involvement in litigation or investigations; (vii) the anticipation of releases of remaining lock-up restrictions; (viii) negative publicity about us or our products; (ix) the volume of Class A Ordinary Shares and Warrants available for public sale; (x) announcements of significant business developments, acquisitions, or new offerings; (xi) general economic, political, regulatory, industry, and market conditions; and (xii) natural disasters or major catastrophic events.
In addition, an active trading market for our Class A Ordinary Shares and Warrants may never develop or, if developed, may not be sustained. You may be unable to sell your Class A Ordinary Shares unless a market can be established and sustained.
These and other factors may cause the market price and demand for our Class A Ordinary Shares and Warrants to fluctuate substantially, which may limit or prevent investors from readily selling their shares and may otherwise negatively affect the liquidity of Class A Ordinary Shares or Warrants. Following periods of such volatility in the market price of a company’s securities, securities class action litigation has often been brought against that company. Because of the potential volatility of Class A Ordinary Shares or Warrants, we may become the target of securities litigation in the future. Securities litigation could result in substantial costs and divert management’s attention and resources from our business.
Sales of a substantial number of our securities in the public market could cause the price of our Class A Ordinary Shares and Warrants to fall.
Sales of a substantial number of Class A Ordinary Shares and/or Warrants, or the perception that those sales might occur, could result in a significant decline in the public trading price of our Class A Ordinary Shares and Warrants and could impair our ability to raise capital through the sale of additional equity securities. We are unable to predict the effect that such sales may have on the prevailing market price of our Class A Ordinary Shares and Warrants.
A certain number of our Warrants have become exercisable for our Class A Ordinary Shares, which would increase the number of shares eligible for future resale in the public market and result in dilution to our shareholders.
Our Warrants to purchase up to 1,492,306 Class A Ordinary Shares have become exercisable on June 17, 2022 in accordance with the terms of the Assignment, Assumption and Amendment Agreement and the Existing Warrant Agreement governing those securities. The exercise price of the Warrants is $133.65 per 1.29 shares (or an effective price of $103.60 per share), subject to adjustment pursuant to the terms of the Assignment, Assumption and Amendment Agreement and the Existing Warrant Agreement. To the extent such Warrants are exercised, additional Class A Ordinary Shares will be issued, which will result in dilution to the existing holders of Class A Ordinary Shares and increase the number of shares eligible for resale in the public market. Sales of substantial numbers of such shares in the public market or the fact that such Warrants may be exercised could adversely affect the market price of Class A Ordinary Shares. Assuming the exercise of all outstanding Warrants for cash, we would receive aggregate proceeds of approximately $154.6 million. However, we will only receive such proceeds if all the Warrant holders exercise all of their Warrants. We believe that the likelihood that warrant holders determine to exercise their warrants, and therefore the amount of cash proceeds that we would receive, is dependent upon the market price of our Class A Ordinary Shares. If the market price for our Class A
28
Ordinary Shares is less than the exercise price of the warrants (on a per share basis), we believe that warrant holders will be very unlikely to exercise any of their warrants, and accordingly, we will not receive any such proceeds. There is no guarantee that the Warrants will ever be “in the money” prior to their expiration, and as such, the Warrants may expire worthless.
If securities or industry analysts do not publish research, publish inaccurate or unfavorable research or cease publishing research about us, our share price and trading volume could decline significantly.
The trading market for our Class A Ordinary Shares will depend, in part, on the research and reports that securities or industry analysts publish about us or our business. We may be unable to sustain coverage by well-regarded securities and industry analysts. If either none or only a limited number of securities or industry analysts maintain coverage of us, or if these securities or industry analysts are not widely respected within the general investment community, the demand for our Class A Ordinary Shares could decrease, which might cause our share price and trading volume to decline significantly. In the event that we obtain securities or industry analyst coverage, if one or more of the analysts who cover us downgrade their assessment or publish inaccurate or unfavorable research about our business, the market price and liquidity for our Class A Ordinary Shares and Warrants could be negatively impacted.
Future resales of our ordinary shares issued to our shareholders and other significant shareholders may cause the market price of our Class A Ordinary Shares to drop significantly, even if our business is doing well.
Certain of our shareholders are subject to contractual lock-ups. Upon expiration or waiver of the applicable lock-up periods, certain of our shareholders and certain other significant shareholders may sell large amounts of our ordinary shares in the open market or in privately negotiated transactions, which could have the effect of increasing the volatility in our share price or putting significant downward pressure on the price of our Class A Ordinary Shares.
Our dual-class voting structure may limit your ability to influence corporate matters and could discourage others from pursuing any change of control transactions that holders of our Class A Ordinary Shares may view as beneficial.
Our authorized and issued ordinary shares are divided into Class A Ordinary Shares and Class B Ordinary Shares. Each Class A Ordinary Share is entitled to one (1) vote, while each Class B Ordinary Share is entitled to twenty (20) votes with all ordinary shares voting together as a single class on most matters. Each Class B Ordinary Share is convertible into one Class A Ordinary Share at any time by the holder thereof, while Class A Ordinary Shares are not convertible into Class B Ordinary Shares under any circumstances. Only Class A Ordinary Shares are listed and traded on Nasdaq, and we intend to maintain the dual-class voting structure.
Mr. Yeung beneficially owns all of the issued Class B Ordinary Shares with disparate voting powers associated with our dual-class share structure. As a result of the dual-class share structure and the concentration of control, holders of Class B Ordinary Shares have considerable influence over matters such as decisions regarding election of directors and other significant corporate actions. This concentration of control may discourage, delay, or prevent a change in control of us, which could have the effect of depriving our other shareholders of the opportunity to receive a premium for their shares as part of a sale of us and may reduce our share price. This concentrated control will limit the ability of holders of Class A Ordinary Shares to influence corporate matters and could discourage others from pursuing any potential merger, takeover, or other change of control transactions that holders of Class A Ordinary Shares may view as beneficial.
The requirements of being a public company may strain our resources, divert our management’s attention and affect our ability to attract and retain qualified board members.
As a publicly traded company, we are subject to the reporting requirements of the Securities Exchange Act of 1934, the Sarbanes-Oxley Act, the Dodd-Frank Act, Nasdaq listing standards, and other applicable regulations. These laws require us to file periodic reports, maintain effective internal controls, and adhere to corporate governance standards, resulting in increased legal, accounting, and compliance expenses. These costs may rise further if we lose our status as an “emerging growth company.”
Ongoing changes in disclosure and governance requirements introduce compliance uncertainty and may require significant management attention and external advisory support. Many of our executives have limited experience managing a public company, and adapting to these demands may divert focus from executing our growth strategy. We may also face
29
challenges attracting qualified directors and officers, particularly for audit committee roles, and may incur higher costs to maintain adequate director and officer liability insurance.
Public company status increases the visibility of our operations and financial condition, which may lead to greater scrutiny and potential litigation from third parties, including competitors. Even if claims are ultimately dismissed or resolved in our favor, the cost and distraction of defending them could materially impact our business, financial condition, results of operations, and reputation.
We are an “emerging growth company,” and it cannot be certain if the reduced SEC reporting requirements applicable to emerging growth companies will make our Class A Ordinary Shares and Warrants less attractive to investors, which could have a material and adverse effect on us, including our growth prospects.
We are an “emerging growth company” as defined in the JOBS Act. We will remain an “emerging growth company” until the earliest to occur of (i) the last day of the fiscal year (a) following the fifth anniversary of the closing of the Business Combination, (b) in which we have total annual gross revenue of at least $1.235 billion, or (c) in which we are deemed to be a large accelerated filer, which means the market value of our shares held by non-affiliates exceeds $700 million as of the last business day of the most recently completed second fiscal quarter, and (ii) the date on which we issued more than $1.0 billion in non-convertible debt during the prior three-year period. We intend to take advantage of exemptions from various reporting requirements that are applicable to most other public companies, whether or not they are classified as “emerging growth companies,” including, but not limited to, an exemption from the provisions of Section 404(b) of the Sarbanes-Oxley Act requiring that our independent registered public accounting firm provide an attestation report on the effectiveness of our internal control over financial reporting and reduced disclosure obligations regarding executive compensation.
In addition, Section 102(b)(1) of the JOBS Act exempts “emerging growth companies” from being required to comply with new or revised financial accounting standards until private companies (that is, those that have not had a Securities Act registration statement declared effective or do not have a class of securities registered under the Exchange Act) are required to comply with the new or revised financial accounting standards. The JOBS Act provides that a company can elect to opt out of the extended transition period and comply with the requirements that apply to non-emerging growth companies, but any such election to opt out is irrevocable. We have elected not to opt out of such extended transition period, which means that when a standard is issued or revised and we have different application dates for public or private companies, we, as an emerging growth company, can adopt the new or revised standard at the time private companies adopt the new or revised standard. This may make comparison of our financial statements with certain other public companies difficult or impossible because of the potential differences in accounting standards used.
Furthermore, even after we no longer qualify as an “emerging growth company,” as long as we continue to qualify as a foreign private issuer under the Exchange Act, we will be exempt from certain provisions of the Exchange Act that are applicable to U.S. domestic public companies, including, but not limited to, the sections of the Exchange Act regulating the solicitation of proxies, consents or authorizations in respect of a security registered under the Exchange Act; certain sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time; and the rules under the Exchange Act requiring the filing with the SEC of quarterly reports on Form 10-Q containing unaudited financial and other specified information, or current reports on Form 8-K, upon the occurrence of specified significant events. Section 8103 of the National Defense Authorization Act for Fiscal Year 2026, named the “Holding Foreign Insiders Accountable Act”, which was signed into law on December 18, 2025, will require directors and officers of foreign private issuers to make insider reports under Section 16(a) of the Exchange Act, effective March 18, 2026. Our principal shareholders continue to remain exempt from the reporting under Section 16(a) of the Exchange Act and our directors, officers and principal shareholders continue to remain exempt from the short-swing profit recovery provisions contained in Section 16(b) of the Exchange Act. In addition, we will not be required to file annual reports and financial statements with the SEC as promptly as U.S. domestic companies whose securities are registered under the Exchange Act, and are not required to comply with Regulation FD, which restricts the selective disclosure of material information.
As a result, our shareholders may not have access to certain information they deem important or at the same time if we were not a foreign private issuer. We cannot predict if investors will find our Class A Ordinary Shares and Warrants less attractive because we rely on these exemptions. If some investors find our Class A Ordinary Shares and Warrants less attractive as a result, there may be a less active trading market and share price for our Class A Ordinary Shares and Warrants may be more volatile.
30
We qualify as a foreign private issuer within the meaning of the rules under the Exchange Act, and as such we are exempt from certain provisions applicable to United States domestic public companies.
Because we qualify as a foreign private issuer under the Exchange Act, we are exempt from certain provisions of the securities rules and regulations in the United States that are applicable to U.S. domestic issuers, including: (i) the rules under the Exchange Act requiring the filing of quarterly reports on Form 10-Q or current reports on Form 8-K with the SEC; (ii) the sections of the Exchange Act regulating the solicitation of proxies, consents, or authorizations in respect of a security registered under the Exchange Act; (iii) certain sections of the Exchange Act requiring insiders to file public reports of their share ownership and trading activities and liability for insiders who profit from trades made in a short period of time; and (iv) the selective disclosure rules by issuers of material nonpublic information under Regulation FD.
Section 8103 of the National Defense Authorization Act for Fiscal Year 2026, named the “Holding Foreign Insiders Accountable Act”, which was signed into law on December 18, 2025, will require directors and officers of foreign private issuers to make insider reports under Section 16(a) of the Exchange Act, effective March 18, 2026. Our principal shareholders continue to remain exempt from the reporting under Section 16(a) of the Exchange Act and our directors, officers and principal shareholders continue to remain exempt from the short-swing profit recovery provisions contained in Section 16(b) of the Exchange Act.
We will be required to file an annual report on Form 20-F within four months of the end of each fiscal year. In addition, we intend to publish our results on a quarterly basis through press releases, distributed pursuant to the rules and regulations of Nasdaq. Press releases relating to financial results and material events will also be furnished to the SEC on Form 6-K. However, the information we are required to file with or furnish to the SEC will be less extensive and less timely compared to that required to be filed with the SEC by U.S. domestic issuers. Accordingly, you may receive less or different information about us than you would receive about a U.S. domestic public company.
We could lose our status as a foreign private issuer under current SEC rules and regulations if more than 50% of our outstanding voting securities become directly or indirectly held of record by U.S. holders and any one of the following is true: (i) the majority of our directors or executive officers are U.S. citizens or residents; (ii) more than 50% of our assets are located in the United States; or (iii) our business is administered principally in the United States. If we lose our status as a foreign private issuer in the future, we will no longer be exempt from the rules described above and, among other things, will be required to file periodic reports and annual and quarterly financial statements as if we were a company incorporated in the United States. If this were to happen, we would likely incur substantial costs in fulfilling these additional regulatory requirements, and members of our management would likely have to divert time and resources from other responsibilities to ensuring these additional regulatory requirements are fulfilled.
We cannot guarantee that any share repurchase program will be fully consummated or that any share repurchase program will enhance long-term shareholder value, and share repurchases could increase the volatility of the price of our Class A Ordinary Shares and could diminish our cash reserves.
On November 30, 2022, our board of directors authorized a share repurchase program, under which we may repurchase up to US$20 million of our Class A Ordinary Shares in the open market over the following 24 months. As of the date of this annual report, we had repurchased 251,390 Class A Ordinary Shares for approximately US$2.45 million under this share repurchase program.
On March 6, 2026, our board of directors authorized a share repurchase program, under which we may repurchase up to US$40 million of our Class A Ordinary Shares through open market purchases, privately negotiated transactions, block purchases, or otherwise in accordance with applicable U.S. federal securities laws, over the following 12 months. As of April 29, 2026, we had repurchased 860,401 Class A Ordinary Shares for approximately US$17.2 million under this share repurchase program.
The share repurchase program, authorized by our board of directors, does not obligate us to repurchase any specific dollar amount or to acquire any specific number of Class A Ordinary Shares. The share repurchase program could affect the price of our Class A Ordinary Shares and increase volatility and may be suspended or terminated at any time, which may result in a decrease in the trading price of our Class A Ordinary Shares.
As a company incorporated in the Cayman Islands and a “controlled company” within the meaning of the Nasdaq corporate governance rules, we are permitted to adopt certain home country practices in relation to corporate
31
governance matters that differ significantly from Nasdaq corporate governance listing standards applicable to domestic U.S. companies or rely on exemptions that are available to a “controlled company”; these practices may afford less protection to shareholders than they would enjoy if we complied fully with Nasdaq corporate governance listing standards.
We are a company incorporated in the Cayman Islands and are listed on Nasdaq as a foreign private issuer. Nasdaq rules permit a foreign private issuer like us to follow the corporate governance practices of our home country. Certain corporate governance practices in the Cayman Islands, which is our home country, may differ significantly from Nasdaq corporate governance listing standards applicable to domestic U.S. companies.
We are a “controlled company” as defined under the Nasdaq rules because Mr. Yeung, chairman of our board of directors and our chief executive officer, owns more than 50% of the total voting power of all of our issued and outstanding ordinary shares. For so long as we remain a controlled company under that definition, we are permitted to elect to rely, and may rely, on certain exemptions from Nasdaq corporate governance rules.
As a foreign private issuer and a “controlled company,” we are permitted to elect to rely, and may rely, on certain exemptions from corporate governance rules, including (i) an exemption from the rule that a majority of our board of directors must be independent directors; (ii) an exemption from the rule that director nominees must be selected or recommended solely by independent directors; (iii) an exemption from the rule that the compensation committee must be comprised solely of independent directors; (iv) an exemption from the requirement that an audit committee be comprised of at least three members; (v) an exemption from the requirement that an annual general meeting must be held; (vi) an exemption from the requirement that we must obtain shareholder approval prior to a plan or other equity compensation arrangement is established or materially amended; and (vii) an exemption from the requirement to obtain shareholder approval for issuing additional securities exceeding 20% of our outstanding ordinary shares. We intend to rely on some or all of the foregoing exemptions available to foreign private issuers and “controlled company.”
As a result, you may not be provided with the benefits of certain corporate governance requirements of Nasdaq applicable to companies that are subject to these corporate governance requirements.
You may face difficulties in protecting your interests, and your ability to protect your rights through U.S. courts may be limited, because we are incorporated under the laws of the Cayman Islands, and we conduct substantially all of our operations, and a majority of our directors and executive officers reside, outside of the United States.
We are an exempted company limited by shares incorporated under the laws of the Cayman Islands, and we conduct the majority of our operations through subsidiaries outside the United States. Substantially all of our assets, and the assets of our directors and executive officers, are located outside the United States. A majority of our directors and officers reside in Hong Kong, and none reside in mainland China. As a result, it may be difficult for U.S. investors to effect service of process on us or these individuals, to bring actions in foreign courts under U.S. securities laws, or to enforce U.S. court judgments abroad.
Our corporate affairs are governed by the Cayman Islands Companies Act, our amended and restated memorandum and articles of association, and Cayman Islands common law. The rights of shareholders under Cayman law are more limited than those under U.S. law and are based in part on English common law precedent, which is persuasive but not binding in the Cayman Islands. For example, shareholders of Cayman Islands companies have no general right to inspect corporate records beyond those required by law, such as the memorandum and articles of association, a list of directors, and any shareholder-approved resolutions. Our amended and restated memorandum and articles of association allow directors to restrict access to financial records and other company documents, further limiting shareholder visibility.
Shareholders also have more limited ability to pursue derivative actions in Cayman courts compared to U.S. jurisdictions. Cayman law may restrict actions for breaches of fiduciary duties, and shareholders may not be able to bring such claims in U.S. courts. In addition, Cayman courts may be unwilling to recognize or enforce U.S. judgments predicated on securities law violations, particularly where such judgments are deemed penal in nature. Although a final, conclusive foreign judgment may be recognized in the Cayman Islands as a debt claim, enforcement is subject to several conditions, including jurisdictional adequacy, procedural fairness, and alignment with local public policy.
As a foreign private issuer listed on the Nasdaq Global Market, we are permitted to follow certain home country corporate governance practices in lieu of Nasdaq’s domestic issuer requirements. To the extent we elect to rely on these exemptions under Nasdaq Rule 5615(a)(3), shareholders may not receive the same level of protection as would be provided
32
under U.S. corporate governance standards. For example, our board composition, committee structure, and shareholder approval requirements may differ from those applicable to U.S.-incorporated companies.
These jurisdictional and legal differences may make it more difficult for shareholders to assert claims, protect their interests, obtain corporate information, or influence management or board decisions. As a result, investors in our securities may face greater challenges in protecting their rights than shareholders of companies incorporated and operating primarily in the United States.
The PCAOB had historically been unable to inspect our auditor in relation to their audit work.
Our auditor, the independent registered public accounting firm that issues the audit report included elsewhere in this annual report, as an auditor of companies that are traded publicly in the United States and a firm registered with the PCAOB, is subject to laws in the United States pursuant to which the PCAOB conducts regular inspections to assess its compliance with the applicable professional standards. The auditor is located in Hong Kong, a jurisdiction where the PCAOB was historically unable to conduct inspections and investigations completely before 2022. The inability of the PCAOB to conduct inspections of auditors in China in the past has made it more difficult to evaluate the effectiveness of our independent registered public accounting firm’s audit procedures or quality control procedures as compared to auditors outside of China that are subject to the PCAOB inspections. On December 15, 2022, the PCAOB issued a report that vacated its December 16, 2021 determination and removed mainland China and Hong Kong from the list of jurisdictions where it is unable to inspect or investigate completely registered public accounting firms. However, if the PCAOB determines in the future that it no longer has full access to inspect and investigate completely accounting firms in mainland China or Hong Kong, and we use an accounting firm headquartered in one of these jurisdictions to issue an audit report on our financial statements filed with the SEC, we and investors in our securities would be deprived of the benefits of such PCAOB inspections again, which could cause investors and potential investors in our securities to lose confidence in our audit procedures and reported financial information and the quality of our financial statements. Any of the foregoing could have a material adverse effect on the market value of our securities.
Our securities may be prohibited from being traded in the United States under the Holding Foreign Companies Accountable Act in the future if the PCAOB is unable to inspect or investigate completely auditors located in China. The Holding Foreign Companies Accountable Act, as amended by the Consolidated Appropriations Act, 2023, decreased the number of “non-inspection years” from three years to two years, and thus, reduced the time before our securities may be prohibited from trading or delisted. The delisting of our securities, or the threat of them being delisted, may materially and adversely affect the value of your investment.
Pursuant to the Holding Foreign Companies Accountable Act, as amended by the Consolidated Appropriations Act, 2023, or the HFCAA, if the SEC determines that an issuer has filed audit reports issued by a registered public accounting firm located in a jurisdiction that has not been subject to inspections by the PCAOB for two consecutive years, the SEC will prohibit the securities of the issuer from being traded on a national securities exchange or in the over-the-counter trading market in the United States.
On June 22, 2021, the U.S. Senate passed the Accelerating Holding Foreign Companies Accountable Act and on December 29, 2022, the Consolidated Appropriations Act was signed into law by President Biden, which, among other things, reduced the number of consecutive non-inspection years required for triggering the prohibitions under the Holding Foreign Companies Accountable Act from three years to two years. The decrease in non-inspection years would reduce the time period before our securities may be prohibited from trading or delisted if the PCAOB determines that it is unable to inspect or investigate completely registered public accounting firms located in Hong Kong, China, under the HFCAA. The location of our auditor is in Hong Kong, China.
On December 16, 2021, the PCAOB issued a report to notify the SEC of its determination that the PCAOB was unable to inspect or investigate completely registered public accounting firms headquartered in mainland China or Hong Kong. On December 15, 2022, the PCAOB removed mainland China and Hong Kong from the list of jurisdictions where it is unable to inspect or investigate completely registered public accounting firms.
Each year, the PCAOB will determine whether it can inspect and investigate completely audit firms in mainland China and Hong Kong, among other jurisdictions. If the PCAOB determines in the future that it no longer has full access to inspect and investigate completely accounting firms in mainland China or Hong Kong and we use an accounting firm headquartered in one of these jurisdictions to issue an audit report on our financial statements filed with the SEC, we would
33
be identified as a Commission-Identified Issuer following the filing of the annual report on Form 20-F for the relevant fiscal year. In accordance with the HFCAA, our securities would be prohibited from being traded on a national securities exchange or in the over-the-counter trading market in the United States if we are identified as a Commission-Identified Issuer for two consecutive years in the future. If our securities are prohibited from trading in the United States, there is no certainty that we will be able to list on a non-U.S. exchange or that a market for our shares will develop outside of the United States. A prohibition of being able to trade in the United States would substantially impair your ability to sell or purchase our securities when you wish to do so, and the risk and uncertainty associated with delisting would have a negative impact on the price of our securities. Also, such a prohibition would significantly affect our ability to raise capital on terms acceptable to us, or at all, which would have a material adverse impact on our business, financial condition, and prospects.
We may issue additional securities without shareholder approval in certain circumstances, which would dilute existing ownership interests and may depress the market price of our shares.
We may issue additional Class A Ordinary Shares, Class B Ordinary Shares convertible into Class A Ordinary Shares or other equity or convertible debt securities of equal or senior rank in the future without approval of the holders of the Class A Ordinary Shares in certain circumstances, including as consideration for strategic acquisitions such as we did with a portion of the consideration for the acquisition of ACT Genomics, or in connection with compensation paid to strategic partners or service providers such as our brand ambassadors. Our issuance of additional Class A Ordinary Shares, Class B Ordinary Shares, or other equity or convertible debt securities of equal or senior rank would have the following effects: (i) our existing shareholders’ proportionate ownership interest in us may decrease; (ii) the relative voting power of each previously outstanding Class A Ordinary Share may be diminished; and (iii) the market price of Class A Ordinary Shares may decline.
We have granted in the past, and we will also grant in the future, share incentives, which may result in increased share-based compensation expenses.
We approved and adopted the 2022 Share Incentive Plan, or the 2022 Plan. Initially, the maximum number of ordinary shares that may be issued under the 2022 Plan is (i) 10% of the total number of our ordinary shares that were outstanding (on a fully diluted basis) as of the date of consummation of the Business Combination (inclusive of the award pool that remains authorized but unissued prior to the consummation of the Business Combination), plus (ii) the number of shares reserved for issuance in accordance with our employee share purchase program, the maximum number being 2% of the total number of our ordinary shares that were outstanding (on a fully diluted basis) as of the date of consummation of the Business Combination. In addition, the number or ordinary shares that may be issued under the 2022 Plan will be increased on the first day of each calendar year, in an amount equal to the lesser of (A) three percent (3%) of the total number of shares issued and outstanding on an as-converted fully-diluted basis on the last day of the immediately preceding fiscal year and (B) such number of ordinary shares determined by the Board. For more information on our 2022 Plan, see “Item 6. Directors, Senior Management and Employees — B. Compensation — Share Incentive Plans — The 2022 Plan.”
We believe the granting of share-based compensation is of significant importance to our ability to attract and retain key personnel, employees and service providers, and as such, we will also grant share-based compensation and incur share-based compensation expenses in the future. As a result, expenses associated with share-based compensation may increase, which may have an adverse effect on our financial condition and results of operations.
The exercise or exchange of our outstanding warrants will dilute the ownership interest of existing shareholders and could adversely affect the market price of our Class A Ordinary Shares and Warrants.
As of the date of this Annual Report, we have multiple classes of outstanding warrants to purchase Class A Ordinary Shares, each with different exercise prices, expiration dates and other terms:
•Warrants. In connection with the Business Combination completed in May 2022, we assumed Warrants originally issued by Artisan Acquisition Corp. These Warrants are publicly traded on the Nasdaq Capital Market under the symbol “PRENW.” The exercise price of the Warrants was adjusted to reflect the 1-for-15 reverse stock split effected in November 2023. Given the significant difference between the current exercise price and recent trading prices of our Class A Ordinary Shares, these Warrants are substantially out of the money as of the date of this annual report. However, if our share price were to increase significantly and the
34
Warrants were exercised by the Warrant holders, the exercise of these Warrants would result in the issuance of a substantial number of additional Class A Ordinary Shares.
•Class A Warrants, Class B Warrants and Placement Agent Warrants. In October 2025, we completed a best-efforts public offering of Class A Ordinary Shares together with accompanying Class A Warrants and Class B Warrants. In connection with the offering, we also issued Placement Agent Warrants to Dominari Securities LLC as partial compensation for placement agent services. For further details regarding the October 2025 offering, the terms of the Class A Warrants, Class B Warrants and Placement Agent Warrants, and the number of such warrants issued, see the Company’s report on Form 6-K filed with the Securities and Exchange Commission on October 28, 2025.
•Class C Warrants. In December 2025, we announced a voluntary warrant exchange program pursuant to which holders of Class A Warrants and Class B Warrants could exchange their warrants for new Class C Warrants with a lower strike price on a two-for-one basis. The warrant exchange program resulted in a significant reduction in the total number of outstanding warrants issued in the October 2025 offering. For further details regarding the warrant exchange program and the terms of the Class C Warrants, see the Company’s reports on Form 6-K filed with the Securities and Exchange Commission on December 23, 2025 and January 5, 2026.
The exercise of any of the foregoing warrants would increase the number of Class A Ordinary Shares outstanding and dilute the ownership percentage and voting power of existing shareholders. Sales of Class A Ordinary Shares issued upon exercise of the warrants in the public market, or the perception that such sales could occur, could depress the market price of our Class A Ordinary Shares. The existence of outstanding warrants may also impair our ability to raise additional capital through the sale of equity or equity-linked securities at a time and price that we deem appropriate.
Our complex capital structure, including multiple classes of warrants with differing terms, may create uncertainty for investors and could adversely affect the trading price of our securities.
Our capital structure includes Class A Ordinary Shares, Class B Ordinary Shares with super-voting rights, Warrants listed on Nasdaq, and unlisted Class A Warrants, Class B Warrants, Class C Warrants and Placement Agent Warrants. These instruments have different exercise prices, expiration dates, and terms. In January 2026, we completed a voluntary warrant exchange program in which holders of approximately 86.7% of the Class A Warrants and Class B Warrants exchanged their warrants for new Class C Warrants with a lower exercise price and shorter term. The complexity of our capital structure may make it more difficult for investors to understand the potential dilutive impact of outstanding securities, which could negatively affect the trading price of our Class A Ordinary Shares and Warrants.
The significant increase in our authorized share capital may result in additional dilution to existing shareholders.
On August 1, 2025, our shareholders approved a special resolution to increase our authorized share capital from US$50,000 (divided into 33,333,334 shares of US$0.0015 par value each) to US$320,000 (divided into 213,333,334 shares of US$0.0015 par value each), representing an approximately 6.4-fold increase. This increase was approved in connection with our plan to file a shelf registration statement with the SEC for the offering of securities with an aggregate offering price of up to US$1 billion to provide flexibility for future capital raising. While we have subsequently filed such shelf registration statement on Form F-3, the significant number of authorized but unissued shares available for issuance in connection therewith gives our board of directors the ability to issue additional shares without further shareholder approval, which could substantially dilute existing shareholders’ ownership interests. Future issuances in connection with the shelf registration statement or otherwise could depress the market price of our Class A Ordinary Shares and may be on terms unfavorable to existing shareholders.
A provision in the Existing Warrant Agreement may result in additional dilution to our shareholders.
Because we issued additional Class A Ordinary Shares for capital raising purposes in connection with the Business Combination at an effective issue price of US$7.75 per Class A Ordinary Share (the “Newly Issued Price”), equivalent to US$116.25 per share after giving effect to the 15-to-1 reverse stock split on November 14, 2023, and the aggregate gross proceeds from such issuances represented more than 60% of the total equity proceeds, and interest thereon, available for the funding of the Business Combination on the date of its completion (net of redemptions), the terms of our Warrants were adjusted pursuant to the Existing Warrant Agreement. Specifically, if the volume weighted average trading price of our Class A Ordinary Shares during the 20-trading day period starting on the trading day prior to the day on which we consummated the Business Combination (such price, the “Market Value”) is below US$9.20 per share (US$138.00 post-
35
split), then: (i) the exercise price of the Warrants will be adjusted (to the nearest cent) to 115% of the higher of the Market Value and the Newly Issued Price; (ii) the US$18.00 per share redemption trigger price (US$270.00 post-split) applicable to our Warrants will be adjusted (to the nearest cent) to 180% of the higher of the Market Value and the Newly Issued Price; and (iii) the US$10.00 per share redemption trigger price (US$150.00 post-split) applicable to our Warrants will be adjusted (to the nearest cent) to the higher of the Market Value and the Newly Issued Price.
As of June 14, 2022, the Market Value was determined to be US$5.41 per share (US$81.15 post-split). As a result, effective after the close of trading on June 14, 2022, and further adjusted to reflect the 15-to-1 reverse stock split on November 14, 2023: (i) the exercise price of the Warrants was adjusted from US$172.50 per 0.086 shares (originally US$11.50 per 1.29 shares) to US$133.65 per 0.086 shares (representing 115% of the post-split Newly Issued Price of US$116.25); (ii) the US$270.00 per share redemption trigger price (originally US$18.00 per share) applicable to the Warrants was adjusted to US$209.25 per share (representing 180% of the post-split Newly Issued Price); and (iii) the US$150.00 per share redemption trigger price (originally US$10.00 per share) applicable to the Warrants was adjusted to US$116.25 per share (representing the post-split Newly Issued Price). These adjustments under the Existing Warrant Agreement may result in additional dilution to our shareholders following the reverse stock split.
Our securities may be delisted from Nasdaq as a result of our failure of meeting the Nasdaq continued listing requirements.
Our securities are currently listed on Nasdaq under the symbol “PRE.” On June 29, 2023, we received a written notice from Nasdaq, notifying us that the Company was not in compliance with the minimum bid price requirement set forth under the Nasdaq Listing Rule 5450(a)(1) (the “Minimum Bid Price Requirement”) as the bid price of the Company’s securities closed below US$1.00 per share for 30 consecutive business days. Pursuant to the Nasdaq Listing Rules, the applicable grace period to regain compliance is 180 days. We had until December 29, 2023 to regain compliance with the Minimum Bid Price Requirement. On November 1, 2023, our shareholders approved a 1-for-15 reverse stock split of our issued and unissued Class A Ordinary Shares and Class B Ordinary Shares, which was effected on November 14, 2023. The effect of the reverse stock split was to consolidate every 15 issued and unissued Class A Ordinary Share and Class B Ordinary Share of US$0.0001 par value each into one Class A Ordinary Share or Class B Ordinary Share, as applicable, of US$0.0015 par value each.
On November 29, 2023, we received a notification letter from Nasdaq, indicating that the closing bid price of the Company’s securities had been at US$1.00 per share or greater for 10 consecutive business days from November 14, 2023 through November 28, 2023, and the Company had regained compliance with the Minimum Bid Price Requirement, and the matter is closed.
However, there can be no assurance that our securities will remain in compliance with the Nasdaq Global Market continued listing requirements going forward. If Nasdaq determines to delist our securities, or if we fail to list our securities on other stock exchanges or find alternative trading venue for our securities, the market liquidity and the value of an investment in our securities will be materially and adversely affected.
Risks Relating to Taxation
We may be or become a passive foreign investment company (“PFIC”), which could result in adverse U.S. federal income tax consequences to U.S. Holders of our Class A Ordinary Shares or warrants.
Depending on the value of our assets, which is determined based, in part, on the market value of our Class A Ordinary Shares, and the nature of our assets and income over time, we could be or become classified as a passive foreign investment company (“PFIC”) for U.S. federal income tax purposes. A non-U.S. corporation, such as us, will be classified as a PFIC for U.S. federal income tax purposes for any taxable year if either (i) at least 75% of its gross income for such year consists of certain types of “passive” income; or (ii) at least 50% of the value of its assets (generally determined on the basis of a quarterly average) during such year is attributable to assets that produce passive income or are held for the production of passive income (the “asset test”). Due to the lack of authority and guidance, the application of such rules with respect to digital assets, or transactions involving digital assets, is subject to uncertainty, although digital assets are likely generally treated as passive assets.
Based on our income and assets and the market value of our Class A Ordinary Shares and treating our digital assets as passive assets for such purposes, we believe that we were not a PFIC for the taxable year ended December 31, 2025. There
36
can be no assurance regarding our PFIC status for the current taxable year or foreseeable future taxable years, however, because our PFIC status is a factual determination made annually that will depend, in part, upon the composition of our income and assets, and includes the uncertainty under the PFIC rules with respect to digital assets and transactions involving digital assets. The value of our assets for purposes of the asset test, including the value of our goodwill and unbooked intangibles, may be determined in part by reference to the market price of our ordinary shares from time to time (which may be volatile). Because we will generally take into account our current market capitalization in estimating the value of our goodwill and other unbooked intangibles, our PFIC status for the current taxable year and foreseeable future taxable years may be affected by our market capitalization, and therefore, fluctuations in our market capitalization create a material risk that we may be classified as a PFIC for the current taxable year and foreseeable future taxable years. In addition, the composition of our income and our assets will be affected by how, and how quickly, we spend our liquid assets. Under circumstances where our revenue from activities that produce passive income significantly increases relative to our revenue from activities that produce non-passive income, or where we determine not to deploy significant amounts of cash for active purposes or our holdings of digital assets increase relative to our other assets, our risk of becoming a PFIC may substantially increase.
Because there are uncertainties in the application of the relevant rules, it is possible that the Internal Revenue Service may challenge our classification of certain income or assets as non-passive, or our valuation of our goodwill and other unbooked intangibles, each of which could cause us to become classified as a PFIC for the current or subsequent taxable years.
If we are a PFIC for any taxable year during which a U.S. Holder (as defined in Item 10. Additional Information — E. Taxation — U.S. Federal Income Tax Considerations to U.S. Holders) holds our Class A Ordinary Shares, or warrants, the U.S. Holder may be subject to certain adverse U.S. federal income tax consequences. Additionally, if we are a PFIC for any taxable year during which U.S. Holders hold our Class A Ordinary Shares or warrants, we would generally continue to be treated as a PFIC with respect to such U.S. Holders even if we do not satisfy either of the above tests to be classified as a PFIC in a subsequent taxable year unless such U.S. Holders were to make certain elections, although such elections could be unavailable with respect to the warrants. Please see “Item 10. Additional Information — E. Taxation — U.S. Federal Income Tax Considerations to U.S. Holders — Passive Foreign Investment Company Status.”
ITEM 4. INFORMATION ON THE COMPANY
A.History and Development of the Company
IM8 is the flagship brand of Prenetics. Prenetics was founded in 2014 in Hong Kong by Danny Yeung and has evolved—through a series of deliberate strategic pivots—from a genomic testing company into a consumer health and longevity platform. IM8 was co-founded with David Beckham and launched in December 2024. Its performance in FY2025 accelerated a strategic conclusion: Prenetics would become a consumer health company, and IM8 would be its engine.
In January 2025, IM8 generated $1 million in monthly revenue. By December 2025, that figure was $10 million. In twelve months, a newly launched brand produced $60.1 million in revenue and reached $120 million in annualized recurring revenue1 ("ARR")—a trajectory that, to our knowledge, is without precedent in the global supplement industry.
From Genomics to Consumer Health
From 2014 to 2019, we built scientific infrastructure in genomic testing and diagnostic services in Hong Kong. In late 2019, we launched CircleDNA, a next generation sequencing consumer genetic testing product that today serves customers in more than 30 countries.
Between 2020 and 2024, we developed the operational capabilities that would underpin IM8. During the COVID-19 pandemic, we became a leading testing services provider for the Hong Kong government and the English Premier League, rapidly scaling laboratory operations and logistics infrastructure. In December 2022, we acquired a majority stake in ACT Genomics, an Asia-based precision oncology company. In June 2023, we invested in Insighta, an early cancer detection
1 The Company uses annualized recurring revenue (“ARR”) as a key operating metric and is calculated by multiplying the monthly revenue from the last month of a given period by 12.
37
company co-founded by Professor Dennis Lo. In August 2024, we acquired Europa Sports Partners, a U.S.-based sports distribution company.
The Launch of IM8
Leveraging the scientific expertise, supply chain infrastructure, and consumer insights accumulated over a decade, we launched IM8 in December 2024 alongside David Beckham as co-founding partner. The thesis: the $150+ billion global supplement industry lacked a brand that combined clinical-grade science, world-class brand partnerships, and a direct-to-consumer subscription model built for scale.
IM8 generated its first $1 million revenue month in January 2025. Monthly revenue surpassed $4 million by June 2025, $6.6 million by September 2025, and exceeded $10 million in December 2025—reaching $120 million in ARR after just twelve months of launch.
Strategic Transformation: Capital Allocation
IM8’s performance made the strategic path clear: concentrate resources on the highest-returning asset. During 2025 and early 2026, we executed three divestitures to transform Prenetics into a focused consumer health company:
Transaction | Value | Rationale | ||||||||||||
ACT Genomics (October 2025) | Up to ~$72M cash (~$46M gross to Prenetics) | Eliminated non-core operational complexity and cash burn; generated non-dilutive capital. | ||||||||||||
Europa 3PL Business (January 2026) | Up to $13M (all-stock) | ~1% gross margin profile fundamentally misaligned with IM8’s premium economics. | ||||||||||||
Insighta (February 2026) | $70M cash | Largest non-dilutive capital event in Company history; 35% equity stake sold to Tencent. | ||||||||||||
The cumulative result: a materially simpler operating structure and a strategic mandate centered entirely on consumer health through IM8.
Capital Markets Initiatives
In October 2025, the Company completed an equity offering raising approximately $44 million in gross proceeds. In December 2025, we completed a voluntary warrant exchange program achieving 86.7% participation, consolidating two series of outstanding warrants into a single new series and materially reducing potential dilution.
In March 2026, the Company’s board of directors authorized a 12-month share repurchase program of up to $40 million, reflecting the board’s view that the Company’s shares were trading below intrinsic value. This followed approximately $2.75 million in open market purchases by the Company’s executive team in Q4 2025 and Q1 2026.
38
Global Ambassador Partnerships
To amplify IM8’s global reach and reinforce its positioning at the intersection of elite performance and science-backed nutrition, we entered into strategic partnerships with world-class athletes throughout 2025 and 2026. Each partnership originated organically—the athlete discovered and used IM8 products before any commercial relationship was discussed—and each ambassador is a Prenetics shareholder, directly aligning financial interests with long-term Company performance.
Ambassador | Sport | Credential | Structure | |||||||||||||||||
David Beckham | Football (Soccer) | Co-founding partner; 85M+ Instagram followers; global cultural icon | Co-founder and equity holder | |||||||||||||||||
Aryna Sabalenka | Tennis | World No. 1; four-time Grand Slam champion | Global ambassador and shareholder (June 2025) | |||||||||||||||||
Ollie Bearman | Formula 1 | Haas F1 Team driver; youngest British F1 starter | Global ambassador and shareholder (February 2026) | |||||||||||||||||
Giannis Antetokounmpo | Basketball (NBA) | Two-time NBA MVP; NBA Champion | All-equity deal; global ambassador and shareholder (April 2026) | |||||||||||||||||

39


40

This roster spans four major global sports, providing year-round brand visibility across the world’s most-watched competitions. To our knowledge, no other consumer health company has assembled a comparable roster of athlete-shareholders across multiple sports.
We are a company incorporated in the Cayman Islands. Our registered office is at Unit 703-706, K11 Atelier King’s Road, 728 King’s Road, Quarry Bay, Hong Kong and our telephone number is +852-2210-9588. Our website is https://www.prenetics.com. The information contained in, or accessible through, our website does not constitute a part of this annual report. The SEC maintains an internet site that contains reports, proxy and information statements, and other information regarding issuers, such as we, that file electronically, with the SEC at www.sec.gov. Our agent for service of process in the United States is Cogency Global Inc., 122 East 42nd Street, 18th Floor New York, N.Y. 10168.
B.Business Overview
We are a consumer health company advancing human health and longevity through science-backed products, global brand partnerships, and AI-driven operations. Our flagship brand, IM8—co-founded with David Beckham—generated $60.1 million in revenue in its first full fiscal year, achieved $120 million in ARR as of December 2025, and ships to more than 30 countries.
Metric | FY2025 | FY2024 | YoY | FY2026E Guidance | ||||||||||||||||||||||
Total Revenue2 | $92.4M | $15.9M | +480% | — | ||||||||||||||||||||||
IM8 Revenue | $60.1M | $0.4M | +16,156% | $180M–$200M | ||||||||||||||||||||||
Gross Profit / Margin | $48.8M / 52.8% | $9.3M / 58.2% | +426% | — | ||||||||||||||||||||||
IM8 Gross Margin | ~63% | — | — | ~60% target | ||||||||||||||||||||||
Loss for the year | $(40.0M) | $(49.8M) | (19.7)% | — | ||||||||||||||||||||||
2 Represents revenue from continuing operations only.
41
Adjusted EBITDA Loss3 | $(13.9M) | $(17.7M) | +21.5% | $(16M)–$(20M); adjusted EBITDA profitability by Q4 2027 | ||||||||||||||||||||||
Loss for the period (Q4 only) | $(7.5M) | $(17.5M) | (57.0)% | — | ||||||||||||||||||||||
Adjusted EBITDA Loss (Q4 only) | $(3.2M) | $(7.6M) | +58.3% | — | ||||||||||||||||||||||
Following our strategic transformation throughout 2025 and into 2026, our business is organized around two brands:
Business Unit4 | FY2025 Revenue | Gross Margin | Status | |||||||||||||||||
IM8 | $60.1M | ~63% | Core growth engine | |||||||||||||||||
CircleDNA | $12.9M | ~85% | Retained; complementary asset | |||||||||||||||||
Europa (3PL business divested in January 2026) | $19.3M | ~1% | Divested | |||||||||||||||||
IM8: Flagship Consumer Health Brand
IM8 was developed in collaboration with our Scientific Advisory Board that includes experts from Cedars-Sinai and leading academic institutions. The brand was built to address a specific market gap: the supplement industry’s chronic deficit of products combining clinical-grade formulation with consumer-grade brand experience. David Beckham, as co-founding partner, was instrumental in shaping the brand’s vision and provides IM8 with organic global demand generation through his 85+ million Instagram following and cultural relevance across markets.
Operating Metrics
IM8 Operating Metric | Q4 2025 | Q3 2025 | QoQ Growth | |||||||||||||||||
Monthly Revenue (End of Period) | $10.0M | $6.6M | +51% | |||||||||||||||||
Quarterly Revenue | $27.4M | $17.2M | +59% | |||||||||||||||||
Total Customer Orders | 230,000+ | 170,000+ | +35% | |||||||||||||||||
Average Order Value (Last Month of Period) | $133 | $102 | +31% | |||||||||||||||||
New Customer Subscription Rate | ~80% | ~80% | Maintained | |||||||||||||||||
Gross Margin | ~60% | ~62% | (2)% | |||||||||||||||||
Our Company’s total revenue in Q4 2025 reached $36.6 million, a 457% year-over-year increase and 55% quarter-over-quarter increase. Q4 2025 gross margin improved to approximately 59%, up from approximately 37% from the same period in the prior year—a nearly 2,300 basis point improvement driven by the increasing dominance of IM8’s premium unit economics in the revenue mix. Based on current cohort performance and modeled retention trends, IM8 is projected to achieve LTV:CAC ratios above 3x across its product portfolio, including premium offerings such as the Beckham Stack.
3 Adjusted EBITDA is a non-IFRS financial measure used by us to measure the strength of our core financial and operating performance. Adjusted EBITDA excludes (1) depreciation and amortization, (2) interest income, (3) other finance costs, (4) income tax expense/(credit), (5) amortization of deferred expenses, (6) equity-settled share-based payment expenses, (7) non-recurring expenses related to acquisition, disposal and fundraising, (8) strategic realignment and discontinued products impact, (9) exchange gain or loss, net, (10) fair value loss on financial assets at fair value through profit or loss, (11) gain on warrant exchange, (12) fair value loss/(gain) on warrant liabilities, (13) unrealized fair value loss on digital asset, (14) gain on partial disposal of an equity-accounted investee, (15) share of loss of equity-accounted investees, net of tax, (16) impairment loss of goodwill, and (17) (profit)/loss from discontinued operations, net of tax. These adjustments are made for items that may not be indicative of our business performance, including non-cash and/or non-recurring items. For more information regarding this non-IFRS financial measure, see "Item 5. Operating and Financial Review and Prospects — A. Operating Results — Non-IFRS Financial Measures."
4 IM8 and Europa form our Consumer Health segment and focus on health and wellness solutions and sports distribution, respectively. CircleDNA forms our Prevention segment and focuses on genetic testing. For more information, see Notes 5 and 6 to our audited consolidated financial statements included elsewhere in this annual report.
42
Product Portfolio
IM8’s product architecture is organized around two pillars: daily performance ("For Today") and longevity ("For Tomorrow"), with a premium bundle combining both.
Product | Description | Key Metric | Certifications | |||||||||||||||||
Daily Ultimate Essentials Pro | All-in-one daily supplement replacing 16+ individual products. 90+ ingredients including vitamins, minerals, adaptogens, pre/pro/postbiotics. Multiple flavors. | Best-selling SKU; flagship product | NSF Certified for Sport; non-GMO; vegan; allergen-free | |||||||||||||||||
Daily Ultimate Longevity | Targets all 12 hallmarks of aging via a five-complex system. Therapeutic doses of NMN (300mg), triple senolytic complex, Spermidine (3mg), Glycine (3g), Taurine (2g), Dihydroberberine (100mg). | Premium longevity SKU | NSF Certified for Sport; non-GMO; vegan; allergen-free | |||||||||||||||||
Beckham Stack | Bundled Essentials Pro + Longevity. Full-spectrum daily nutrition and aging support. Highest average order value SKU in portfolio. | ~50% quarterly plan adoption (March 2026) | NSF Certified for Sport; non-GMO; vegan; allergen-free | |||||||||||||||||

43


All IM8 products are manufactured in FDA-registered facilities in the United States and undergo independent third-party testing through NSF Certified for Sport, verifying products are free from over 280 contaminants and confirming ingredient dosages match labeling.
Product Pipeline and Category Expansion
44
We are actively expanding the IM8 product portfolio. We expect to launch a minimum of two new SKUs by the end of 2026, each targeting multi-billion dollar addressable categories. We have a further product pipeline extending into 2027 with additional SKU launches planned. These new products are designed to complement our existing Daily Ultimate range, increase average order value, broaden our addressable market, and deepen customer engagement within the IM8 ecosystem. We believe this cadence of innovation—entering new categories while deepening our existing product lines—is critical to sustaining growth and reinforcing IM8’s competitive position.
Subscription-First Business Model
Approximately 80% of new IM8 customers opted into a subscription plan at initial purchase throughout FY2025—a rate that held consistent from Q1 through Q4.
In December 2025, we introduced quarterly subscription plans in the U.S. market, expanding globally across our international markets in January 2026. The impact was substantial. Approximately half of Beckham Stack customers and approximately one-third of Daily Ultimate Essentials Pro customers are opting for quarterly subscriptions, with adoption accelerating across all SKUs. This shift produced a step-change in unit economics:
Period | Average Order Value | |||||||
FY2025 | ~$110 | |||||||
Q4 2025 (Last Month of Period) | ~$133 | |||||||
January–February 2026 (New Customer) | ~$233 | |||||||
This step-change reflects both the shift toward quarterly prepayments and increased adoption of higher-value product bundles. The quarterly model improves cash flow through full upfront prepayment, reduces per-month fulfillment costs, and creates more predictable renewal cadences. The resulting increase in customer acquisition cost reflects the deliberate acquisition of higher-quality, longer-duration subscribers with materially higher lifetime value.
Global Revenue Distribution
Over 60% of IM8’s FY2025 revenue was generated outside the United States.
Market | IM8 Revenue5 | % of IM8 Revenue | ||||||||||||
United States | $23.8M | 39.7% | ||||||||||||
Canada | $8.8M | 14.7% | ||||||||||||
United Kingdom | $7.7M | 12.8% | ||||||||||||
Australia | $3.2M | 5.3% | ||||||||||||
Singapore | $2.4M | 4.0% | ||||||||||||
In 2026, we are executing market-specific localization—including localized websites, native-language advertising, and culturally tailored content—across Japan, Germany, France, Spain, Hong Kong, and Australia, while entering new high-growth geographies. IM8 products are sold exclusively through our direct-to-consumer online store at www.im8health.com and through affiliated social media channels.
CircleDNA
CircleDNA is our consumer genetic testing brand leveraging next-generation sequencing technology to deliver over 500 personalized reports across 20+ categories. Since its global launch in November 2019, CircleDNA has delivered more than 500,000 test kits across 30+ countries. For FY2025, CircleDNA generated $12.9 million in revenue at approximately 85% gross margins. We offer four product tiers—Vital, Family Planning, Health, and Premium—sold primarily through www.circledna.com.
5 Revenue breakdown as shown in this table is based on the market to which the IM8 products are delivered. For a breakdown of the Company’s revenues from continuing operations from external customers based on the location of the relevant Group entity’s domicile, see Note 6 to our audited consolidated financial statements included elsewhere in this annual report.
45
We view IM8 and CircleDNA as complementary assets. CircleDNA’s genetic insights into nutrition, fitness, and health risk create a natural cross-sell pathway into IM8’s product portfolio. We intend to deepen integration between the platforms over time.
Research and Development
Our investment in clinical validation is a structural competitive advantage. Unlike the majority of nutraceutical companies—which rely on ingredient-level literature to support marketing claims—we invest in randomized, controlled clinical trials conducted by independent research institutions to substantiate product-level efficacy. To our knowledge, very few companies in the Vitamins, Minerals and Supplements (“VMS”) category conduct product-level randomized controlled trials at all; fewer still run multiple concurrent trials across different product lines.
Scientific Advisory Board
Our R&D strategy is guided by a nine-member Scientific Advisory Board drawn from leading institutions:
Advisor | Institutional Affiliation | Domain | ||||||||||||
Prof. Suzanne Devkota | Cedars-Sinai Medical Center | Microbiome research | ||||||||||||
Dr. James L. Green | Former Chief Scientist, NASA (42 years) | Scientific rigor and evidence standards | ||||||||||||
Dr. Dawn Mussallem | CMO, Fountain Life; former Mayo Clinic physician (20+ years) | Longevity and integrative medicine | ||||||||||||
Dr. James DiNicolantonio | Cardiovascular research scientist and Doctor of Pharmacy; 300+ published papers | Cardiovascular nutrition; dosing protocols | ||||||||||||
Dr. Pamela Mehta | Board-certified orthopedic surgeon; Co-founder of Learn and Pinnacle | Regenerative and longevity medicine | ||||||||||||
Dr. Amy Shah | Double board-certified physician | Integrative medicine, allergy/immunology | ||||||||||||
Dr. Ara Suppiah | Head of Medical Services, LIV Golf | Sports medicine and performance | ||||||||||||
Dr. Darshan Shah | CEO, Next Health; board-certified surgeon | Longevity medicine | ||||||||||||
Simon Hill | Physiotherapist; nutrition scientist; The Proof podcast (40M+ listens) | Longevity and nutrition | ||||||||||||
Clinical Research and Product Validation
In October 2024, we commenced a 12-week randomized, controlled clinical trial evaluating Daily Ultimate Essentials at the San Francisco Research Institute with 60 healthy adults aged 25–60. In the trial, 95% of participants reported improved daily energy levels, 85% reported improved digestion and reduced bloating, 80% reported better sleep, and 75% reported improved focus and mental clarity.
2026 Clinical Trial Program
In 2026, we intend to launch three additional randomized, controlled clinical trials—a scale of clinical investment that, to our knowledge, is unprecedented in the VMS category. Critically, these trials are designed to generate biomarker-level data, moving beyond self-reported outcomes to objective, measurable endpoints that strengthen both our scientific credibility and marketing claims.
Trial | Scope | Key Endpoints | ||||||||||||
Daily Ultimate Essentials Pro (2nd Gen) | Larger cohort, advanced biomarker endpoints; deepening evidence base for flagship SKU | Blood biomarkers, energy, immunity, digestion | ||||||||||||
46
Daily Ultimate Longevity Biomarker Trial | Impact on biological aging markers, cellular health, systemic inflammation; includes wearable biometric monitoring | Biological age, heart rate variability, sleep quality, inflammatory markers | ||||||||||||
Gut Health / Microbiome | Designed with Prof. Devkota (Cedars-Sinai); evaluating gut microbiome composition, digestive symptom severity, gut barrier function | Microbiome composition, gut barrier integrity | ||||||||||||
The output of these trials—published, peer-reviewable clinical data generated by independent research institutions—is designed to provide a compounding competitive advantage: each result strengthens subsequent product launches, marketing claims, and partnership credibility.
AI-Driven Operations: Technology as a Structural Advantage
Artificial intelligence is not a peripheral initiative at Prenetics—it is embedded in our operational infrastructure. We believe our AI capabilities represent a durable competitive advantage that legacy supplement brands cannot easily replicate, and we expect it to be a primary driver of operating leverage as we scale toward profitability.
AI-Powered Creative Production. We utilize generative AI for advertising production—video, imagery, and copy—at scale, speed, and cost levels that would be impossible with traditional production methods. As of April 1, 2026, we maintain over 2,000 active ad creatives running simultaneously in our Meta Ads Manager—a 20x increase from approximately 100 active ads one year prior. The cost of producing static and video creative assets has decreased by approximately 80% year-over-year as a result of AI-driven production workflows. A single AI-generated video campaign featuring brand ambassador Aryna Sabalenka garnered over 233 million views on Instagram and was the number one social video advertisement on the platform for that year—demonstrating that AI-powered creative production can match or exceed the engagement of conventionally produced content at a fraction of the cost. This capability enables us to produce hundreds of unique ad variations per week, test them against real-time performance data, and iterate rapidly—a creative velocity that fundamentally changes the economics of digital customer acquisition.
AI-Optimized Customer Acquisition. Our marketing engine leverages machine learning for audience segmentation, bid optimization, and predictive modeling of customer lifetime value. AI models analyze behavioral signals across our customer base to identify high-propensity prospects, dynamically allocate marketing spend across channels and geographies, and optimize landing page experiences in real-time based on visitor attributes and predicted conversion probability. Each landing page experience is dynamically assembled using AI to match messaging, social proof, and product recommendations to the individual visitor’s profile, maximizing conversion rates and improving customer acquisition cost efficiency.
AI Use Across the Organization. We promote AI proficiency and use across the entire organization. Every employee has access to advanced AI tools including Claude and Claude Code for their daily workflows. We use AI across product development (clinical research review, ingredient interaction analysis, dosing optimization), supply chain management (demand forecasting, inventory optimization across 30+ countries), and internal operations. We believe this organization-wide AI adoption is critical to sustaining our lean operating model. Every new hire must be justified against the potential for AI to perform the function, and all candidates are required to complete a structured AI competency challenge as part of the hiring process—a standard we believe is unique in the consumer health industry.
Company-Wide AI Agent Initiative. In March 2026, we launched a company-wide initiative requiring every team member to build and present a functional AI agent designed to improve their specific area of the business. This program ensures that AI adoption is distributed across every function of the organization, not confined to the technology team, and is designed to compound productivity gains over time.
AI in Search and Discovery. We are actively implementing AI-optimized structured data, metadata, and content strategies across our digital properties to ensure IM8 is discoverable and accurately represented by AI-powered search engines and recommendation systems. As consumer search behavior migrates from traditional keyword queries to conversational AI interfaces, we believe early investment in AI search optimization will provide a meaningful organic traffic advantage relative to competitors who have not made similar investments.
Manufacture, Distribution and Supply
We rely on third-party manufacturers and distributors for production and distribution. We do not maintain in-house manufacturing or distribution capability and do not plan to develop such capacity in the foreseeable future.
47
Our IM8 products are manufactured in collaboration with leading nutraceutical manufacturers in FDA-registered facilities in the United States. All products undergo NSF Certified for Sport testing. We source ingredients from multiple suppliers and are not dependent on any single-source supplier. We have not historically experienced material difficulty in obtaining raw materials in required quantities and do not believe that prices of our principal raw materials are subject to significant volatility.
We rely on a limited number of third-party logistics partners for fulfillment and delivery in our key markets. While the loss of, or a significant disruption in our relationship with, any of these partners could temporarily affect our ability to fulfill orders in a timely manner, we believe alternative partners with comparable capabilities are available in each of our principal markets and that we could transition to such alternatives within a reasonable timeframe if required. To mitigate supply chain and distribution risk, we have diversified manufacturers and suppliers across countries and regions. For risks related to third-party suppliers, see "Item 3. Key Information — D. Risk Factors — Risks Relating to Our Business and Industry — We rely on a limited number of suppliers, manufacturers, distributors and other service providers, and may not be able to find replacements or immediately transition to alternatives, which could adversely affect our ability to meet customer demand."
Sales and Marketing
IM8. Our marketing architecture combines three elements: science-led credibility (Scientific Advisory Board endorsements, clinical trial results), global sports partnerships (David Beckham, Aryna Sabalenka, Ollie Bearman, Giannis Antetokounmpo—all equity-aligned), and AI-powered performance marketing (generative creative production, machine learning-based targeting and optimization). As of March 31, 2026, approximately 80% of paid spend is deployed on Meta platforms, with 15% on Google. In 2026, we are diversifying into YouTube, TikTok, and AppLovin to reduce platform concentration risk and access new demographics. Selling and marketing expense represented 38.5% of total revenue for FY2025 and we expect this ratio to be in the range of 45% to 50% of revenue in 2026 as we invest in IM8’s next phase of international expansion, and will continue to closely monitor this figure as a key indicator of marketing efficiency.
We supplement paid acquisition with content marketing (expert interviews, educational content), community events in key markets (Miami, New York, London), segmented email campaigns targeting active, at-risk, and churned subscribers, and affiliate marketing through micro-influencer networks.
CircleDNA. We utilize digital advertising (Google, Meta), influencer marketing, affiliate partnerships, and collaborations with healthcare providers and research institutions.
Competition
We operate in highly competitive markets across our business units. Our ability to innovate, maintain operational efficiency, build brand loyalty, and scale our AI-driven operating model will be critical to sustaining our market position and achieving long-term growth.
IM8. IM8 operates in the global consumer health and wellness market, which exceeds hundreds of billions of dollars annually. We compete with established supplement brands with significant distribution and brand recognition, direct-to-consumer all-in-one brands such as Athletic Greens (AG1), Gruns, and longevity-focused brands such as Elysium Health. Barriers to entry are relatively low, leading to continuous competitive pressure from smaller brands that compete primarily on price.
IM8’s competitive differentiation rests on a combination of factors that we believe are difficult to replicate in aggregate: our co-founding partnership with David Beckham, a roster of equity-aligned athlete ambassadors spanning four major global sports, clinical validation through independent randomized controlled trials, a world-class Scientific Advisory Board, comprehensive formulations replacing 16+ individual supplements, NSF Certified for Sport certification, an AI-powered creative and customer acquisition engine operating at 2,000+ simultaneous ad creatives, a subscription-first model generating predictable recurring revenue, and a demonstrated product pipeline entering new multi-billion dollar categories. Failure to maintain product quality, innovation, or consumer trust could adversely affect our competitive position.
CircleDNA. CircleDNA competes with established players including Ancestry.com LLC and MyHeritage Ltd. Our competitive strength lies in comprehensive next generation sequencing methodology and focus on actionable health insights. Intense competition, price pressure, and the need for continuous innovation could challenge growth.
48
Intellectual Property
We regard our patents, trademarks, copyrights, domain names, proprietary formulations, know-how, trade secrets, and similar intellectual property as critical to our success. We rely on patent, trademark, and copyright law, employment agreements with IP assignment clauses, and confidentiality and non-compete agreements to protect our rights. We have implemented measures to protect and preserve our trade secrets and proprietary rights through confidentiality agreements with employees, manufacturers, suppliers, and R&D collaborators. Our ability to compete effectively is also dependent on intellectual property licensed from brand partners and proprietary formulations developed with third-party manufacturers.
We may from time to time engage in litigation to protect trade secrets or know-how, defend against infringement claims, or determine the scope and validity of others’ proprietary rights. See "Item 3. Key Information — D. Risk Factors — Risks Relating to Intellectual Property and Legal Proceedings" for additional information.
Government Regulations
Regulation of Consumer Genetic Testing and IVD devices
In Hong Kong, there are no specific laws or regulations that directly regulate the sales of consumer genetic testing and IVD devices, such as our CircleDNA products.
In Hong Kong, there are certain laws and regulations relating to consumer protection, advertisements, data protection, codes of practice and standards, which may apply to our business.
Regulations relating to Consumer Protection and Advertising in Hong Kong
We make certain representations with respect to our products on various media, including the product itself, our website, social media (including using social media influencers), advertising billboards, advertising vehicles and broadcast media. The Trade Descriptions Ordinance (Cap. 362), as amended by the Trade Descriptions (Unfair Trade Practices) (Amendment) Ordinance 2012, (“TDO”), provides the overriding principle that all product descriptions must be true and not misleading and prohibits the application of a false trade description to any goods or to supply or offer to supply any goods to which a false trade description is applied. The TDO broadly applies to all goods, including our consumer genetic testing kits and IVD device. “Trade description” is broadly defined to cover indications, direct or indirect, and given by whatever means, of various matters with respect to goods or parts of goods, including quantity, composition and fitness for purpose, strength, performance, behavior and accuracy. The Customs and Excise Department is the principal enforcement agency of the TDO. The maximum penalty for non-compliance with the TDO on conviction is a fine of HK$500,000 and imprisonment for five years. The TDO also provides for a civil compliance-based mechanism as an alternative to initiating prosecution under which the Customs and Excise Department may, with the consent of the Secretary for Justice, accept a written undertaking from a trader believed to have engaged, be engaging, or be likely to engage in conduct that constitutes any of the prohibited practices to the discontinuation of the relevant conduct.
Advertisements on television or radio must comply with the Generic Code of Practice on Television Advertising Standards (“TV Code”) and the Radio Code of Practice on Advertising Standards (“Radio Code”). The general standard provided for by the TV Code and Radio Code is that advertising should be legal, clean, decent, honest and truthful. The TV Code also strictly controls the design and content of medical product advertisements, and prohibits impression of professional advice and support from medical professionals, appeals to fear or exploitation of credulity, encouragement of excess, and exaggerated claims using superlative or comparative adjectives such as “the most successful” or “quickest.” Complaints regarding advertisements in broadcasting should be made to the Communications Authority. Penalties for breach of the TV Code or the Radio Code are typically applied to broadcasters, rather than the product owner and include fines up to HK$200,000 for the first occasion a penalty is imposed, up to HK$500,000 for the second occasion, and up to HK$1,000,000 for any subsequent occasion. If we are at fault for these breaches, we may be required to assume the relevant liabilities by our contract with the broadcaster.
Regulations relating to Dietary Supplement Products in the United States
In the United States, the formulation, manufacture, packaging, holding, labeling, promotion, advertising, distribution, and sale of our dietary supplements are regulated by multiple federal and state authorities, including: (1) the FDA; (2) the Federal Trade Commission (“FTC”); (3) the Consumer Product Safety Commission (“CPSC”); (4) the U.S. Environmental Protection Agency (“EPA”); (5) U.S. Customs and Border Protection (“CBP”); and (6) state attorneys general. Our activities also are regulated by various agencies of the states, localities and foreign countries in which our products are
49
manufactured, distributed, or sold. The FDA, in particular, regulates dietary supplements primarily as food, including the formulation, manufacture, and labeling of dietary supplements. The IM8 products marketed by us in the United States are classified as dietary supplements under the FFDCA.
FDA regulates dietary supplements through current good manufacturing practice (“CGMP”) requirements in 21 CFR Part 111 (“Part 111”). Among other things, Part 111 requires manufacturers to establish specifications and to conduct 100% identity testing of each dietary ingredient before use, unless FDA grants a petitioned exemption, to ensure products are not adulterated and are properly labeled. We have implemented a comprehensive quality assurance program that is designed to maintain compliance with the CGMPs for products manufactured on our behalf for distribution in the United States.
Facilities that manufacture, process, pack, or hold our dietary supplements for U.S. consumption must be registered with FDA and renew such registration every even-numbered year. Imported shipments are subject to prior notice to FDA before arrival and are subject to inspection and refusal if non-compliant.
Where we import finished supplements or components from foreign suppliers, we or our U.S. importer must comply with FDA’s Foreign Supplier Verification Programs (“FSVP”) rule (21 CFR Part 1, Subpart L). The FSVP rule generally requires risk-based verification activities and importer identification at entry, with modified requirements for dietary supplements and for importers that establish and verify specifications under Part 111.
Product labeling must include a Supplement Facts panel and otherwise comply with FDA’s food labeling rules (21 CFR 101.36). Dietary supplement labeling may include structure/function claims if the company has substantiation that the statements are truthful and not misleading, the product bears the required FDA disclaimer, and FDA is notified within 30 days of first marketing. Disease claims are not permitted. Apart from structure and function claims, FDA permits companies to use FDA-approved full and qualified health claims for food and supplement products containing specific ingredients that meet stated requirements.
If a product contains a new dietary ingredient (i.e. not marketed in the United States before October 15, 1994), a new dietary ingredient notification is generally required to be submitted to FDA at least 75 days before marketing, demonstrating that the ingredient, under labeled conditions of use, is reasonably expected to be safe.
We are required to report to FDA any serious adverse events associated with our dietary supplements sold in the United States within 15 business days of receipt of a consumer report of an adverse event, and to maintain related records for six years. As a result of reported adverse events, we may from time to time elect, or be required, to remove a product from a market, either temporarily or permanently.
In the United States, the FTC exercises jurisdiction over the advertising of our products. The FTC’s Health Products Compliance Guidance emphasizes that health-related advertising claims must be truthful, not misleading, and supported by competent and reliable scientific evidence, which are typically well-controlled human clinical trials for efficacy claims. These principles extend to endorsements, testimonials, and influencer marketing. We evaluate our advertising to align with this guidance. The FTC has in the past instituted enforcement actions against dietary supplement and food companies generally for false and misleading advertising of some of their products.
In addition to federal oversight, some state laws impose specific requirements relevant to dietary supplements. For example, California’s Proposition 65 may require warnings for exposure to listed chemicals (such as lead) above established “safe harbor” levels. We monitor state developments and evaluate our products for compliance when sold in such jurisdictions.
In foreign markets, prior to making or permitting sales of our products in the market, we may be required to obtain an approval, license or certification from the relevant country’s ministry of health or comparable agency. The approval process generally requires us to present each product and product ingredient to appropriate regulators and, in some instances, arrange for testing of products by local technicians for ingredient analysis. The approvals may be conditioned on reformulation of our products, or may be unavailable with respect to some products or some ingredients.
Regulations relating to Health Supplement Products in Canada
In Canada, our IM8 products are classified as natural health products ("NHPs") and are regulated by Health Canada under the Natural Health Products Regulations (SOR/2003-196) (the "NHP Regulations"), made pursuant to the Food and Drugs Act (R.S.C. 1985, c. F-27). NHPs include vitamins, minerals, herbal remedies, probiotics, and other supplement products sold for health-related purposes.
50
Before an NHP may be sold in Canada, it must hold a product licence issued by Health Canada's Natural and Non-prescription Health Products Directorate. To obtain a product licence, an applicant must submit a product licence application demonstrating that the product is safe and efficacious for its recommended conditions of use, including evidence supporting any health claims. Health Canada may rely on compendial references, such as its licensed Natural Health Products Database and monograph system, as well as traditional use evidence and published clinical data, in evaluating applications. Each licensed NHP is assigned a Natural Product Number ("NPN") or, for homeopathic products, a Drug Identification Number – Homeopathic Medicine ("DIN-HM"), which must appear on the product label.
Any person who manufactures, packages, labels, or imports an NHP for sale in Canada must hold a site licence issued by Health Canada. Site licence holders must comply with the good manufacturing practices ("GMP") set out in the NHP Regulations, which address premises, equipment, sanitation, quality assurance, quality control, stability testing, and recordkeeping. Our IM8 products are manufactured in the United States and shipped directly to consumers in Canada. NHP shipments entering Canada are subject to inspection by Health Canada and the Canada Border Services Agency, and may be refused entry if the products are unlicensed or non-compliant with applicable requirements.
NHP labeling must comply with the requirements of the NHP Regulations and the Food and Drugs Act, including the display of the NPN or DIN-HM, recommended use or purpose, recommended dose, route of administration, risk information (including cautions, warnings, contraindications, and known adverse reactions), medicinal and non-medicinal ingredients, and proper storage conditions. Health claims appearing on NHP labeling must be consistent with the claims approved in the applicable product licence. The Food and Drugs Act prohibits the labeling, packaging, sale, or advertising of an NHP in a manner that is false, misleading, or deceptive, or that is likely to create an erroneous impression regarding its character, value, composition, merit, or safety.
Licence holders are required to report serious adverse reactions associated with their NHPs to Health Canada within 15 days of becoming aware of such reactions. Health Canada may suspend or revoke a product licence if it determines that an NHP poses a serious or imminent risk to health, or if the licence holder fails to comply with the terms of the licence or applicable regulatory requirements. Non-compliance with the Food and Drugs Act or the NHP Regulations may result in enforcement actions, including product recalls, seizures, injunctions, and criminal prosecution.
Advertising of NHPs in Canada is subject to the Food and Drugs Act, which prohibits false, misleading, or deceptive advertising, as well as advertising a product as a treatment, preventative, or cure for the diseases, disorders, or abnormal physical states set out in Schedule A to the Food and Drugs Act. Advertising must also be consistent with the terms of the product's licence. The Competition Act (R.S.C. 1985, c. C-34), enforced by the Competition Bureau, further prohibits materially false or misleading representations in the promotion of a product.
Regulations relating to Food Supplement Products in the United Kingdom
In the United Kingdom, our IM8 products are classified as food supplements and are regulated primarily under the Food Supplements (England) Regulations 2003 (S.I. 2003/1387) and equivalent regulations in Scotland, Wales, and Northern Ireland (collectively, the "Food Supplements Regulations"), which implement retained EU Directive 2002/46/EC. Food supplements are also subject to the general requirements of the Food Safety Act 1990, the General Food Regulations 2004, and retained Regulation (EC) No 178/2002 (the "General Food Law").
Food supplements placed on the UK market must be safe for human consumption. Under the General Food Law, the food business operator responsible for placing the product on the UK market bears primary responsibility for ensuring the safety of the products and must not market food that is unsafe or injurious to health. Our IM8 products are manufactured in the United States and sold directly to consumers in the United Kingdom through our website. We maintain a UK-based responsible person who acts as the food business operator for purposes of compliance with applicable UK food law. Food supplements may only contain vitamins, minerals, and other substances that are permitted under applicable legislation. Operators placing food supplements on the UK market must notify the relevant competent authority in each constituent nation of the United Kingdom.
The manufacture, processing, and distribution of food supplements must comply with food hygiene requirements, including retained Regulation (EC) No 852/2004 on the hygiene of foodstuffs, and applicable food safety management procedures based on Hazard Analysis and Critical Control Points ("HACCP") principles. Food businesses must be registered with their local authority and are subject to inspection by local authority environmental health officers and trading standards officers.
Food supplement labeling must comply with the Food Supplements Regulations and retained Regulation (EU) No 1169/2011 on the provision of food information to consumers ("FIC"). For products sold to consumers via our website or
51
other distance means, the FIC requires that mandatory food information be available to the consumer before the purchase is concluded and at the time of delivery. Required information includes the names and amounts of the categories of nutrients or substances that characterize the product, the recommended daily portion, a warning not to exceed the stated recommended daily dose, a statement that food supplements should not be used as a substitute for a varied diet, and a statement that the product should be stored out of reach of young children. Labeling must not attribute to food supplements the property of preventing, treating, or curing a human disease, nor make any medicinal claims, as such claims would cause the product to be classified as a medicinal product subject to the requirements of the Human Medicines Regulations 2012.
Nutrition and health claims made in respect of food supplements must comply with retained Regulation (EC) No 1924/2006 on nutrition and health claims made on foods (the "NHCR"). Under the NHCR, only health claims that have been authorized and included on the GB Register of Nutrition and Health Claims may be used. Claims must be based on generally accepted scientific evidence, must not be false, ambiguous, or misleading, and must not encourage or condone excess consumption. Disease risk reduction claims and claims referring to children's development and health are subject to additional authorization requirements. The use of unauthorized health claims may result in enforcement action.
The Food Standards Agency ("FSA") is the central competent authority responsible for food safety policy in England, Wales, and Northern Ireland, with Food Standards Scotland performing this role in Scotland. Enforcement of food law, including the Food Supplements Regulations and labeling requirements, is carried out at the local level by trading standards officers and environmental health officers. The Advertising Standards Authority ("ASA"), through the Committee of Advertising Practice ("CAP") Code and the Broadcast Committee of Advertising Practice ("BCAP") Code, regulates the advertising of food supplements and requires that advertising claims are substantiated, truthful, and not misleading.
Non-compliance with UK food law may result in enforcement actions including improvement notices, prohibition orders, product withdrawal or recall, and criminal prosecution. Offenses under the Food Safety Act 1990 may result in fines and, for certain offenses, imprisonment for a term of up to two years.
Regulation of Clinical Trials
We conduct clinical trials to validate the efficacy and safety of certain products. Although dietary supplements are generally not subject to the same premarket approval requirements as pharmaceutical drugs, the Company elects to conduct clinical trials to substantiate the structure/function claims we make about our products and to provide scientific validation of product efficacy. These clinical trials are designed to generate evidence supporting our marketing claims and to differentiate our products in the marketplace through rigorous science-backed validation.
Clinical trials conducted in the United States involving human subjects are subject to FDA regulations (21 CFR Parts 50, 56, and 312) to the extent they involve investigational drugs or devices. For dietary supplement studies, the FDA’s regulations may apply if the study design involves an Investigational New Drug (“IND”) application or if the supplement is being studied for use as a drug. We assess each study to determine the applicable regulatory requirements and engage with Institutional Review Boards (“IRBs”) to ensure appropriate oversight and protection of study participants.
Regulations relating to Privacy and Data Protection
We collect, process and use personal data for our products and services and are subject to laws, rules and regulations relating to the privacy and security of directly or indirectly identifiable personal information (collectively, “Data Protection Laws”). Such Data Protection Laws address the collection, storage, sharing, use, disclosure, and protection of certain types of personal information, including genetic information, and frequently evolve in scope and enforcement. There can also be uncertainty, differing interpretations and contradictory requirements across the legal and regulatory landscape regarding privacy and security.
Data Protection in Hong Kong
In Hong Kong, the main data protection law is Personal Data (Privacy) Ordinance (Cap. 486) (“PDPO”). The PDPO is enforced by the Office of the Privacy Commissioner for Personal Data (“PCPD”). Under the PDPO, personal data means any data “relating directly or indirectly to a living individual, from which it is possible and practical to ascertain the identity of the individual from the said data, in a form in which access to or processing of the data is practicable”. According to the PCPD, genetic data possesses the characteristics of being unique to the individual and a particular individual could be identified when the data is linked with personal data in another database.
52
The PDPO does not have extraterritorial effect and applies to data users that control the collection, holding, processing or use of personal data in or from Hong Kong. Notably, PCPD has noted that even if any genetic companies are registered outside Hong Kong and the PDPO has no extraterritorial jurisdiction, the PCPD may liaise with overseas personal data protection authorities for follow-up actions pursuant to international enforcement agreements and cooperation arrangements in appropriate cases where Hong Kong residents’ personal data is involved.
While there is no concept of “sensitive personal data” under the PDPO, the PCPD has published guidance note on how data users should collect and use biometric data (including DNA samples) in compliance with the PDPO. PCPD has emphasized the need for caution in handling sensitive biometric data as any wrongful disclosure of biometric data could lead to grave consequences. Such guidance note does not have the force at law, but any non-compliance can be a proof of contravention of the relevant requirements under PDPO. We are also subject to the general requirements under the PDPO including obligations that are set out under the following Data Protection Principles (DPPs):
•DPP 1 – Purpose and manner of collection of personal data: personal data shall only be collected for a lawful purpose directly related to a function or activity of the data user and the data collected should be necessary and adequate but not excessive in relation to that purpose. DPP 1 also provides for the steps required and information a data user must give to a data subject when personal data is collected.
•DPP 2 – Accuracy and duration of retention of personal data: data users shall take all practicable steps to ensure that personal data is accurate and is not kept longer than is necessary for the fulfilment of the purpose for which the data is used.
•DPP 3 – Use of personal data: personal data should only be used for the purposes for which they were collected or a directly related purpose. A data user is required to obtain the prescribed consent of the data subject if the data user intends to use the personal data for a new purpose.
•DPP 4 – Security of personal data: data users shall take all practicable steps to protect the personal data they hold against unauthorized or accidental access, processing, erasure, loss or use.
•DPP 5 – Information to be generally available: data users shall take all practicable steps to ensure that a person can ascertain a data user’s policies and practices on personal data and be informed of the kind of personal data and the main purposes for which personal data are held.
•DPP 6 – Access to personal data: data subjects have the rights to request access to and correction of their own personal data, and be given reasons when any such request is refused.
We obtain informed consent from our customers prior to obtaining their samples. In some situations, we may be required to share health data with authorities for public health purposes. Under section 60B of the PDPO, there is an exemption from the requirement to obtain prescribed consent (i.e. DPP 3) to use the personal data collected, including health data, for purposes other than the original purpose if the use of the data is required or authorized by or under any laws or court order in Hong Kong. This may include requests properly made by the legal authorities under laws such as the Prevention and Control of Diseases Ordinance (Cap. 599). Section 59 of the PDPO also provides an exemption for disclosing health data if the data user can show that obtaining express consent from the individual would likely cause serious harm to the health of the individual or others. Under Section 59(1) of the PDPO, in circumstances where the application of the restrictions on the use of data would be likely to cause serious harm to the physical or mental health of the data subject or any other individual, the data user may disclose personal data relating to the physical or mental health of the data subject to a third party without the consent of the data subject (exemption for DPP 3). Section 59(2) provides that under the above circumstances, the data user can also disclose the identity or location of a data subject to a third party without the consent of the data subject.
When the PCPD receives a complaint or has reasonable grounds to believe that there may be a contravention of PDPO, the PCPD may conduct an investigation of the suspected contravention and publish a report setting out the investigation results and recommendations if it is in the public interest to do so. Contravention of a DPP is not an offence. However, contravention of certain provisions of PDPO (e.g. breach of an enforcement notice issued by PCPD) may amount to an offence. Breaches of the PDPO may lead to a variety of civil and criminal sanctions including fines and imprisonment. In the event of a breach, the PCPD may issue an enforcement notice requiring the data user to take remedial action and/or preventive steps. Failure to comply with an enforcement notice constitutes an offense, resulting in a maximum fine of HK$50,000 and up to two years’ imprisonment (plus a daily fine of HK$1,000 in the event the offense continues). Subsequent convictions can result in a maximum fine of HK$100,000 and imprisonment for up to two years, with a daily penalty of HK$2,000. There are certain offenses under the PDPO that carry more onerous penalties (e.g. a person committing an offence of disclosing personal data without consent from data users with intent to gain or to cause loss to the data subject may be liable on conviction to a fine of up to HK$1 million and imprisonment for up to five years (section 64(1) and (3) of PDPO)). In addition, data subjects have a right to bring proceedings in court to seek compensation
53
for damage. The PCPD may also grant legal assistance to the aggrieved individual who intends to institute proceedings to seek compensation.
Data Protection in the United States
There is no U.S. federal law applicable to all industry sectors governing the collection, use and disclosure of personal data. Comprehensive data protection laws are regularly introduced in the United States Congress, but none have been adopted.
At the U.S. federal level, providers of healthcare and medical services (and their processors) that engage in certain transactions related to government or commercial insurance programs are subject to the Health Insurance Portability and Accountability Act of 1996, as amended (“HIPAA”), and its implementing rules and regulations, which broadly regulate the collection, use, and disclosure of genetic information and personal information relating to health. In addition, federal law prohibits the use of genetic information in making employment-related decisions or for insurance underwriting purposes. Because they are generally outside of the healthcare provider environment, providers of DTC supplements and genetic and other health-related or medical tests are generally not subject to these federal requirements.
In addition, the U.S. Department of Justice has promulgated regulations implementing Executive Order 14117 on Preventing Access to Americans’ Bulk Sensitive Personal Data and United States Government-Related Data by Countries of Concern. These regulations, which became effective in April 2025, prohibit transactions involving certain sensitive personal data categories, including health data, genetic data, and biospecimens, to countries of concern, including China. The regulations also restrict investment agreements, employment agreements and vendor agreements involving such data and countries of concern. Should these regulations apply to any aspect of our business, we may be required to implement additional due diligence, risk assessment, notification, and recordkeeping measures, particularly with respect to cross-border data transfers and transactions involving bulk sensitive data. Noncompliance may result in civil or criminal penalties.
State laws regulating the collection, use and disclosure of personal data by providers of DTC supplements and genetic and other health-related or medical tests are not uniform and they vary in significant ways, resulting in a “patchwork” of different compliance obligations, enforcement mechanisms, and penalties for violations.
Several states have adopted laws to protect genetic information collected by direct-to-consumer testing services. These laws, which vary by state, generally require full disclosure of the company’s security protections, purposes for collection, and marketing and retention practices. They also require express consent to perform the test and disclose the results to third parties, and a process to withdraw consent. Violations may lead to civil fines and even criminal penalties and some states enable consumers to bring a private lawsuit to enforce these protections.
All states require notification to affected individuals of a breach of the specific types of personal information set out in each state’s law. However, many of these laws do not cover a breach of genetic or any other type of health-related information. Some states, but not all, also require notification of a data breach to the state’s attorney general. State breach notification laws are enforced by the states’ attorneys general and, in some states, consumers have a private right of action.
A number of states require a private company to maintain reasonable safeguards to protect unencrypted, computerized personal information of state residents, including health-related information, against access or acquisition by an unauthorized person. However, only a few states provide guidance as to what security measures are needed to meet the standard of reasonableness.
At least 19 states have adopted data protection laws that have much broader protection and cover all types of personal data that can identify or reasonably be linked to a natural person. Similar laws are under active consideration in other states. These laws have some features that are similar to the protection of personal data in the U.K. GDPR. These privacy laws generally treat health and genetic data as “sensitive” information subject to additional restrictions including, for example, (i) collection only with informed consent, (ii) use only for specified and limited purposes, and (iii) transparency about disclosure to third parties and retention. Additional states (including Washington and Nevada) have enacted laws that regulate the collection, use, and disclosure of “consumer health data,” including with respect to analytics, website, and marketing activities. Consumers in Washington have a private right of action.
Even where the federal and state laws and regulations described above do not apply, the Federal Trade Commission and state attorneys general have asserted that failures to abide by public statements regarding consumer privacy or to implement appropriate data security measures may constitute unfair acts or practices in or affecting commerce in violation of Section 5(a) of the Federal Trade Commission Act and state consumer protection laws. In addition, plaintiffs’ lawyers
54
are also increasingly using privacy-related statutes at both the state and federal level to bring lawsuits against companies for their data-related practices. In particular, there have been a significant number of cases filed against companies for their use of pixels and other web trackers. These cases often allege violations of the California Invasion of Privacy Act and other state laws regulating wiretapping, as well as the federal Video Privacy Protection Act.
Several states, including Colorado and California, have passed laws regulating various uses of artificial intelligence (“AI”). In addition, various federal regulators have issued guidance on the use of AI in regulated sectors. If we develop or use AI systems governed by these laws or regulations, including as informed by regulatory guidance, we will need to meet high standards of data quality, transparency, monitoring, and human oversight, and we would need to adhere to specific and potentially burdensome and costly ethical, accountability, and administrative requirements, with the potential for significant enforcement or litigation in the event of any perceived non-compliance.
Concern is high and increasing among U.S. federal and state lawmakers and regulators about protecting the security of personal data and prohibiting its undisclosed commercialization or other uses not known to or approved by the individual. We anticipate that government regulation and public expectations for personal data protection, particularly for sensitive genetic and health-related data, will become more demanding over time and require us to stay abreast of new legal developments. In addition to meeting our compliance obligations, we recognize that the perception of personal data concerns, whether or not valid, may harm our reputation and brand and adversely affect our business, financial condition, and results of operations.
C.Organizational Structure
The following diagram depicts a simplified organizational structure of the Company as of the date of this annual report.
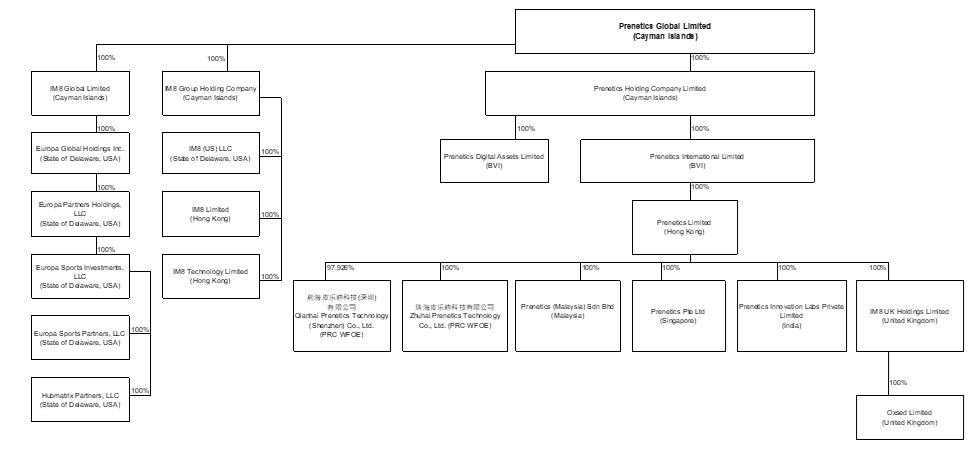
D.Property, Plants and Equipment
We are headquartered in Hong Kong and have employees across our international markets, including in the United States. We lease office space in Hong Kong and Malaysia. Our headquarters serves as the center for management, sales and marketing, R&D coordination, technology support, and general administration. We believe our existing facilities are sufficient for our current needs, and we will obtain additional facilities, principally through leasing, to accommodate future expansion plans as needed.
ITEM 4A. UNRESOLVED STAFF COMMENTS
None.
55
ITEM 5. OPERATING AND FINANCIAL REVIEW AND PROSPECTS
The following discussion and analysis of our financial condition and results of operations should be read in conjunction with our consolidated financial statements and the related notes thereto included elsewhere in this annual report. In addition to historical consolidated financial information, the following discussion contains forward-looking statements that reflect our plans, estimates, and beliefs that involve risks and uncertainties. Our actual results could differ materially from those discussed in the forward-looking statements as a result of many factors, including those factors set forth in the sections titled “Item 3. Key Information – D. Risk Factors” and “Forward-Looking Statements.” We caution you that our businesses and financial performance are subject to substantial risks and uncertainties. For discussion of year-over-year comparisons between 2024 and 2023 that are not included in this annual report on Form 20-F, refer to “Item 5. Operating and Financial Review and Prospects” found in our Form 20-F for the year ended December 31, 2024, that was filed with the Securities and Exchange Commission on April 30, 2025.
A.Operating Results
Overview
We are a consumer health company advancing human health and longevity through science-backed products, global brand partnerships, and AI-driven operations. Our flagship consumer health brand, IM8—co-founded with David Beckham—generated $60.1 million in revenue in its first full fiscal year, achieved $120 million in ARR as of December 2025, and ships to more than 30 countries. We also operate a consumer genetic testing brand in CircleDNA leveraging next-generation sequencing technology to deliver over 500 personalized reports across 20+ categories. Since its global launch in November 2019, CircleDNA has delivered more than 500,000 test kits across 30+ countries. We view IM8 and CircleDNA as complementary assets. CircleDNA’s genetic insights into nutrition, fitness, and health risk create a natural cross-sell pathway into IM8’s product portfolio.
During 2025 and early 2026, we executed divestitures of ACT Genomics, our Europa 3PL business, and Insighta to transform Prenetics into a focused consumer health company.
Key Factors Affecting Results of Operations
We believe that our performance and future success depend on several factors that present significant opportunities for us but also pose risks and challenges. The following are key factors that we believe will affect our results of operations going forward.
Ability to convert customers into paying subscribers and to retain customers
Our ability to convert new customers into paying subscribers and to retain existing subscribers is a key factor in our ability to grow revenue and achieve profitability. The substantial majority of our IM8 revenue is generated through subscription-based plans offered on our direct-to-consumer platform, under which subscribers are billed and shipped products on a recurring basis. As of December 31, 2025, approximately 80% of all new IM8 orders were placed by subscribers through a monthly or quarterly subscription plan. This subscription-driven revenue model provides a degree of predictability and visibility into future revenues.
In late 2025 and early 2026, we began transitioning a growing proportion of new IM8 customers from monthly to quarterly subscription plans, with the aim of increasing upfront cash collection, improving logistics efficiency, and enhancing customer lifetime value. The shift has resulted in a meaningful increase in average order value, from approximately US$110 for full year 2025 to approximately US$233 for new customers in January and February 2026. We expect to continue optimizing our subscription mix and pricing to improve unit economics and strengthen long-term revenue visibility.
Subscriber retention is critical to the sustainability of our growth trajectory. Subscriber renewal rates, average order frequency, and the relationship between customer lifetime value and cost of acquiring new customers are important to the long-term health of our business. If subscriber retention declines, or if we are unable to convert a sufficient proportion of new customers into recurring subscribers, our revenue and operating results could be adversely affected. Consumer preferences in the health and wellness market may shift, competitive alternatives may emerge, or macroeconomic conditions may reduce consumer willingness to maintain discretionary health-related subscriptions.
Ability to grow and strengthen the IM8 brand and expand into new markets
Our future growth is closely tied to our ability to build, maintain, and strengthen the IM8 brand as a premium, science-backed health and longevity brand with global appeal. IM8 was launched in December 2024 as a premium-positioned consumer health brand co-founded with David Beckham, and after just twelve months achieved approximately
56
US$120 million in annualized recurring revenue, shipping to over 30 countries. Our ability to sustain this growth trajectory depends on continued consumer perception of IM8 as a trusted, differentiated brand within the increasingly competitive health and wellness market.
Brand strength is driven by several factors, including the quality and efficacy of our product formulations, the credibility of our Scientific Advisory Board, the visibility and relevance of our global ambassador partnerships, and the consistency of our customer experience across all touchpoints. Any adverse publicity, product quality issues, negative consumer sentiment, or failure to maintain our premium positioning could diminish the value of the IM8 brand and negatively affect our ability to attract and retain customers. Additionally, as the global health and wellness supplement market is large and highly fragmented, we face competition from both established supplement brands and new market entrants, including private-label offerings. Our ability to differentiate the IM8 brand through product innovation, marketing excellence, and scientific credibility will be important to maintaining and expanding our market share.
Geographic expansion is a significant component of our growth strategy. In fiscal year 2025, IM8 shipped products to more than 30 countries, with over 60% of IM8 revenue generated outside the United States. Our top markets by revenue include the United States, Canada, the United Kingdom, Australia, and Singapore. We intend to deepen our market penetration in these existing markets while selectively expanding into new geographies where we believe there is strong demand for premium health and longevity products. Consolidating our position in existing markets will require continued investment in localized marketing, customer support infrastructure, and logistics optimization to deliver a consistent brand experience across regions.
Expanding into new markets involves additional risks and challenges, including the need to comply with varying regulatory frameworks governing the formulation, labeling, importation, and sale of dietary supplements in each jurisdiction, as well as adapting our marketing strategies and product offerings to local consumer preferences and competitive dynamics. We may also need to establish new fulfillment and distribution capabilities, enter into local partnerships, and navigate foreign currency exposures. If we are unable to successfully consolidate our position in existing markets or expand into new geographies, our revenue growth and long-term market opportunity could be limited.
Ability to manage costs, including freight, marketing and logistics optimization
Our ability to effectively manage our cost structure is an important factor that will affect our profitability and cash flow. As a direct-to-consumer health and wellness brand that ships products globally across more than 30 countries, we incur significant costs related to freight, warehousing, packaging, last-mile delivery, and fulfillment operations. Changes in global shipping rates, carrier availability, fuel costs, tariffs, and customs duties can materially impact our direct costs and gross margins.
In fiscal year 2025, our gross margin from continuing operations was approximately 53%, reflecting the blended contribution of our higher-margin IM8 consumer health business and the lower-margin Europa distribution business. With the divestiture of our Europa 3PL business completed in January 2026 and our strategic focus now centered on IM8, we expect our gross margin profile to improve to over 60% in 2026, subject to our ability to manage input costs, optimize our supply chain, and negotiate favorable terms with contract manufacturers and logistics providers.
We are actively pursuing supply chain optimization initiatives, including vendor consolidation, freight route optimization, strategic inventory management, and the pursuit of volume-based procurement efficiencies, to reduce per-unit costs as we scale. Our transition toward quarterly subscription plans is also expected to yield logistics efficiencies by reducing the frequency of individual shipments per subscriber per year. If we are unable to manage these costs effectively, or if we experience unexpected increases in input costs, freight rates, or other supply chain disruptions, our gross margins and overall profitability could be negatively impacted.
Ability to grow and enhance our product portfolio, including through research and development
Our growth strategy depends in part on our ability to develop and introduce new science-backed health and wellness products that resonate with consumers, drive incremental revenue, and increase customer lifetime value. Since IM8’s commercial launch in December 2024, we have introduced multiple product offerings, including Daily Ultimate Essentials Pro, Daily Ultimate Longevity, and the Beckham Stack bundle, each of which has contributed to meaningful increases in average order value and subscriber engagement.
We intend to continue investing in research and development to expand our product portfolio across complementary categories within health, wellness, and longevity. Our R&D efforts are supported by our in-house scientific capabilities, our Scientific Advisory Board, and our partnerships with academic and industry experts. New product introductions, such as the launch of Daily Ultimate Longevity in October 2025, have historically driven step-changes in our business performance, including in average order value and subscriber acquisition rates.
57
Our ability to successfully develop, formulate, and commercialize new products in a timely manner is subject to various risks, including regulatory requirements in multiple jurisdictions, the complexity of product formulation and testing, supply chain readiness, and consumer acceptance. If we are unable to maintain the pace and quality of product innovation, or if new products fail to achieve market acceptance, our revenue growth and competitive position could be adversely affected.
Investment in sales and marketing, and management of customer acquisition costs
We expect to make continued significant investments in sales and marketing to drive customer acquisition, build brand awareness, and expand our global footprint. Marketing is an important driver of our growth, and our selling and marketing expenses represented approximately 38% of revenue from continuing operations for the year ended December 31, 2025, compared to 34% in 2024. We anticipate that selling and marketing expenses will continue to increase in absolute terms as we scale our operations and enter new markets, although we expect these expenses to stabilize or decline as a percentage of revenue over time as we achieve greater operating leverage.
A central element of our marketing strategy is our portfolio of high-profile global ambassador and influencer partnerships. IM8 was co-founded with David Beckham, whose involvement has been instrumental in establishing the brand’s premium positioning and generating consumer trust. In 2025 and early 2026, we expanded our ambassador ecosystem to include Aryna Sabalenka, World No. 1 tennis player and four-time Grand Slam champion, Ollie Bearman, a Formula 1 driver and one of the sport’s most exciting stars, and Giannis Antetokounmpo, two-time NBA MVP and NBA champion, each of whom also became shareholders of Prenetics. These partnerships serve to reinforce IM8’s association with elite athletic performance and wellness, and have contributed to significant brand awareness and viral marketing campaigns, including content that generated hundreds of millions of social media impressions. We may, from time to time, enter into additional ambassador or influencer partnerships across multiple sports and wellness verticals, and deploy mass media campaigns to further promote our products and brand.
Our ability to manage customer acquisition costs efficiently is critical to the long-term sustainability of our business model. We acquire new customers through a variety of channels, including paid social media, influencer-driven content, organic search, partnerships, and physical advertising. As we have transitioned toward acquiring longer-duration quarterly subscribers, our per-customer acquisition costs have increased on an absolute basis, reflecting our deliberate investment in higher-quality subscribers with greater projected lifetime value. However, changes in digital advertising rates, platform algorithms, privacy regulations, or the effectiveness of our marketing content could result in increases in customer acquisition costs that are not offset by corresponding improvements in customer quality or retention. Our results of operations will be materially affected by our ability to balance growth-oriented marketing investments with disciplined management of the costs incurred to acquire and onboard new subscribers.
Key Components of Results of Operations
Revenue from continuing operations
We manage our businesses by divisions organized based on the nature of the products and services offered.
During the fiscal year ended December 31, 2024, we identified three operating segments: the Prevention segment, Diagnostics segment and Consumer Health segment.
On October 1, 2025, we disposed of our Diagnostics business, which is presented as discontinued operations in accordance with IFRS 5 in our consolidated financial statements for the year ended December 31, 2025. As a result of this disposal, we currently operate through two reportable segments: (i) the Prevention segment and (ii) the Consumer Health segment.
The table below sets forth our revenue from continuing operations by business segment for the periods presented.
| Year Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
(in thousands of U.S. dollars) | |||||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Prevention | $ | 12,945 | $ | 10,367 | $ | 6,155 | |||||||||||
| Consumer Health | 79,445 | 5,569 | n/a | ||||||||||||||
| Total revenue from continuing operations | $ | 92,390 | $ | 15,936 | $ | 6,155 | |||||||||||
58
For the year ended December 31, 2025, the Prevention segment from continuing operations and the Consumer Health segment from continuing operations contributed approximately 14% and 86% of total revenue from continuing operations, respectively.
For the year ended December 31, 2024, the Prevention segment from continuing operations and the Consumer Health segment from continuing operations contributed approximately 65% and 35% of our total revenue from continuing operations, respectively.
For the year ended December 31, 2023, the Prevention segment from continuing operations contributed 100% of our total revenue from continuing operations.
The table below presents our revenue from continuing operations by region for the years indicated. For more information on how we present our revenue from continuing operations by region, see Note 6 to our audited consolidated financial statements included elsewhere in this annual report.
| Year Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
(in thousands of U.S. dollars) | |||||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Hong Kong | $ | 23,070 | $ | 10,425 | $ | 6,155 | |||||||||||
| The United States | 69,320 | 5,511 | — | ||||||||||||||
| $ | 92,390 | $ | 15,936 | $ | 6,155 | ||||||||||||
Revenue in our Prevention segment is derived from the provision of genetic testing services. Revenue is recognized at a point in time upon when the Group satisfies its performance obligation by delivering the test results or reports to the customer.
Revenue in our Consumer Health segment is derived from (i) the sale of consumer health and wellness products and (ii) the provision of fulfillment and distribution services for sports nutrition products. Revenue from the sale of consumer health products is recognized at a point in time when control of the goods transfers to the customer, which occurs upon delivery. For fulfillment and distribution services, revenue is recognized at a point in time when the customer's goods are delivered to the named destination. Revenue related to storage, packaging, receiving, and in-bound freight management services is recognized over time as the services are rendered.
Direct Costs, Gross Profit, and Gross Margin from continuing operations
Direct costs from continuing operations primarily comprise costs of inventories, staff costs, depreciation of lab equipment, freight and delivery expenses, and service-related charges, including next generation sequencing cost. As we continue to focus on our Consumer Health business, we are implementing disciplined cost management across procurement, manufacturing and logistics. We expect our cost structure to remain well-controlled and aligned with revenue growth, supported by supply chain optimization, vendor negotiations and operational efficiency initiatives. Over time, we believe scale efficiencies within our platform and tighter inventory and fulfillment management will support stable and sustainable gross margins.
Gross profit from continuing operations represents the difference between total revenue from continuing operations and total direct costs from continuing operations, and gross margin from continuing operations represents gross profit from continuing operations as a percentage of total revenue from continuing operations. Over the long term, we expect gross profit to benefit from revenue growth and disciplined cost management. As we continue to scale our Consumer Health business, we anticipate that operating leverage, supply chain optimization and procurement efficiencies will support stable and sustainable gross margins.
Other Income and Other Net Gains from continuing operations
Other income and other net gains from continuing operations primarily consist of government subsidies, bank interest income, dividend income. net foreign exchange (loss)/gain and sundry income.
59
Selling and Marketing Expenses from continuing operations
Selling and marketing expenses from continuing operations primarily comprise advertising and marketing expenses, staff costs, commission expenses, assembly and fulfillment expenses and other marketing and distribution expenses. These costs are incurred to support brand-building activities, marketing campaigns, customer acquisition efforts, packaging and fulfillment activities and sales channel development.
We expect selling and marketing expenses to increase in absolute terms as we continue to invest in customer acquisition, brand awareness and market expansion. Given our direct-to-consumer growth strategy, we anticipate marketing expenses will remain relatively stable as a percentage of revenue, reflecting sustained investment to drive revenue growth and market penetration.
Research and Development Expenses from continuing operations
Research and development expenses from continuing operations primarily comprise staff costs, production-related expenses, product infrastructure expenses and depreciation of right-of-use assets. These costs support product innovation, formulation enhancements and business development initiatives.
We expect research and development expenditures to remain disciplined and aligned with our product roadmap. While investment levels may vary depending on the timing and scope of specific initiatives, we anticipate that overall research and development spending will remain measured and strategically focused.
Impairment of Goodwill from continuing operations
Impairment of goodwill from continuing operations represents non-cash charges recognized when the carrying value of goodwill allocated to a cash-generating unit exceeds its estimated recoverable amount.
Administrative and Other Operating Expenses from continuing operations
Administrative and other operating expenses from continuing operations primarily comprise staff costs, consultancy fees, legal and professional service fees, auditor remuneration, subscription and software expenses, depreciation and amortization, and licensing and royalty fees. These expenses support the Group's corporate functions, including finance, legal, compliance, information technology infrastructure and administrative operations, as well as certain licensing and royalty arrangements associated with our Consumer Health business.
We expect these expenses to increase in absolute terms in the near term as we continue to continue to strengthen our operating infrastructure and meet the requirements of operating as a public company. This includes costs related to regulatory compliance, SEC reporting obligations, director and officer insurance, investor relations, external advisory services and technology enhancements. Over the longer term, we expect administrative and other operating expenses to remain disciplined and to benefit from operating leverage as revenue scales, supported by enhanced systems, process efficiencies and greater cost absorption across a larger revenue base.
Fair Value Gain/(Loss) on Financial Assets at Fair Value Through Profit or Loss from continuing operations
Fair value gain/(loss) on financial assets at fair value through profit or loss represents unrealized gain or loss arising from changes in the fair value of such assets during the reporting period. This is a non-cash item and does not impact our cash flows.
Gain on Warrant Exchange
Gain on warrant exchange represents the gain arising from the exchange of certain warrants for the Company’s ordinary shares during the reporting period. Upon the exchange, the warrant liabilities were derecognized and replaced by equity instruments measured at fair value at the exchange date. The difference between the carrying amount of the warrant liabilities and the fair value of the consideration issued was recognized as a gain. This is a non-cash item and does not impact our cash flows.
60
Fair Value Gain on Warrant Liabilities from continuing operations
Fair value gain on warrant liabilities relates to the changes in the fair value of the warrants which are issued in connection with the de-SPAC and placement transaction and measured at fair value through profit or loss. The warrants are exercisable from May 18, 2022 and will expire on May 18, 2027. This is a non-cash item and does not impact our cash flows.
Unrealized Fair Value Loss on Digital Assets from continuing operations
Unrealized fair value loss on digital assets represents non-cash losses arising from decreases in the fair value of our digital assets, which consist primarily of Bitcoin holdings. These assets are measured at fair value, with changes in fair value recognized in profit or loss based on quoted market prices at the reporting date. As a result, this line item may fluctuate depending on market conditions and Bitcoin price movements.
Gain on Partial Disposal of an Equity-accounted Investee
Gain on partial disposal of an equity-accounted investee represents the gain recognized upon the sale of a portion of our ownership interest in an investee accounted for using the equity method. The gain is measured as the difference between the consideration received and the proportionate carrying amount of the investment disposed. This is a non-recurring, non-cash gain to the extent it relates to the reclassification of accumulated equity-accounted balances and does not impact our cash flows.
Share of Loss of Equity-accounted Investees, net of tax
Share of loss of equity-accounted investees, net of tax, represents our proportionate share of the net losses incurred by investees accounted for under the equity method. Such losses are recognized in our consolidated statement of profit or loss and other comprehensive income in accordance with the equity method of accounting.
Other Finance Costs from continuing operations
Other finance costs primarily comprise interest expenses recognized on lease liabilities in accordance with IFRS 16. We currently do not have bank borrowings or other interest-bearing financing arrangements.
Income Tax (Expense)/Credit from continuing operations
Income tax (expense)/credit from continuing operations represents the net tax benefit recognized in respect of our continuing operations for the year. We are subject to income taxes in the jurisdictions in which we operate, each with its own statutory tax rates. Our effective tax rate may vary based on the geographic mix of taxable income or losses, the availability and utilization of tax credits and loss carryforwards, changes in the valuation of deferred tax assets and liabilities, and amendments to applicable tax laws and regulations.
Profit/(loss) From Discontinued Operations, Net of Tax
Profit/(loss) from discontinued operations, net of tax, represents the post-tax results of businesses that have been disposed of or classified as discontinued operations in accordance with applicable accounting standards. During the year ended December 31, 2025, this primarily relates to the disposal of ACT Genomics on October 1, 2025 and the results for the year include both the operating results of ACT Genomics prior to disposal and the gain recognized on disposal of the subsidiary. In prior periods, discontinued operations included the cessation of COVID-19-related diagnostic services and other DNA testing operations in the EMEA region.
Other Comprehensive Income/(Expense)
Other comprehensive income/(expense) primarily comprises exchange differences arising from the translation of foreign operations, which are driven by fluctuations in foreign exchange rates relative to the prior reporting period. It also includes our share of other comprehensive income or expense of equity-accounted investees recognized under the equity method, as well as the reclassification of cumulative translation reserves to profit or loss upon disposal of foreign operations.
61
Results of Operations
The following table sets forth our consolidated statements of profit or loss and other comprehensive income and their respective dollar amount for the years presented. Following the table, we discuss our results of operations for the year ended December 31, 2025 compared to the year ended December 31, 2024.
| Year Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| (in thousands of U.S. dollars) | |||||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Revenue | $ | 92,390 | $ | 15,936 | $ | 6,155 | |||||||||||
| Direct costs | (43,600) | (6,659) | (4,982) | ||||||||||||||
| Gross profit | 48,790 | 9,277 | 1,173 | ||||||||||||||
| Other income and other net gains | 614 | 2,006 | 4,133 | ||||||||||||||
| Selling and marketing expenses | (35,540) | (5,413) | (5,462) | ||||||||||||||
| Research and development expenses | (5,132) | (9,051) | (9,172) | ||||||||||||||
| Impairment of goodwill | (6,815) | — | — | ||||||||||||||
| Administrative and other operating expenses | (46,398) | (33,090) | (29,116) | ||||||||||||||
| Operating loss from continuing operations | (44,481) | (36,271) | (38,444) | ||||||||||||||
| Fair value gain/(loss) on financial assets at fair value through profit or loss | 780 | (8,869) | (7,135) | ||||||||||||||
| Gain on warrant exchange | 36,657 | — | — | ||||||||||||||
| Fair value (loss)/gain on warrant liabilities | (17,943) | 49 | 3,351 | ||||||||||||||
| Unrealized fair value loss on digital assets | (9,725) | — | — | ||||||||||||||
| Gain on partial disposal of an equity-accounted investee | — | 1,244 | — | ||||||||||||||
| Share of loss of equity-accounted investees, net of tax | (1,260) | (2,010) | (670) | ||||||||||||||
| Other finance costs | (241) | (168) | (46) | ||||||||||||||
| Loss before taxation | (36,213) | (46,025) | (42,944) | ||||||||||||||
| Income tax (expense)/credit | (34) | 7,639 | (54) | ||||||||||||||
| Loss from continuing operations | (36,247) | (38,386) | (42,998) | ||||||||||||||
| Discontinued operations | |||||||||||||||||
| Loss from discontinued operations, net of tax | (3,731) | (11,420) | (21,779) | ||||||||||||||
| Loss for the year | (39,978) | (49,806) | (64,777) | ||||||||||||||
| Other comprehensive income/(expense) | |||||||||||||||||
| Items that will not be reclassified subsequently to profit or loss: | |||||||||||||||||
| Share of other comprehensive (expense)/income of equity-accounted investees | (26) | 303 | — | ||||||||||||||
| Item that may be reclassified subsequently to profit or loss: | |||||||||||||||||
| Exchange differences on translation of foreign operations | 716 | (1,024) | 1,795 | ||||||||||||||
| Reclassification of cumulative translation reserve upon disposal of foreign operations | (74) | — | — | ||||||||||||||
| Other comprehensive income/(expense) for the year | 616 | (721) | 1,795 | ||||||||||||||
| Total comprehensive expense for the year | $ | (39,362) | $ | (50,527) | $ | (62,982) | |||||||||||
62
Comparison of the Years Ended December 31, 2025 and 2024
Revenue From Continuing Operations
Year Ended December 31 | |||||||||||||||||||||||
| 2025 | 2024 | $ Change | % Change | ||||||||||||||||||||
| (in thousands of U.S. dollars, unless otherwise stated) | |||||||||||||||||||||||
| (Restated) | |||||||||||||||||||||||
| Prevention | $ | 12,945 | $ | 10,367 | $ | 2,578 | 25 | % | |||||||||||||||
| Consumer Health | 79,445 | 5,569 | 73,876 | 1327 | % | ||||||||||||||||||
| Total revenue from continuing operations | $ | 92,390 | $ | 15,936 | 76,454 | 480 | % | ||||||||||||||||
Our total revenue from continuing operations increased by $76.5 million, or 480%, from $15.9 million for the year ended December 31, 2024 to $92.4 million for the year ended December 31, 2025. The increase was primarily driven by the rapid growth of our consumer health business, particularly IM8, which contributed $60.1 million in revenue during 2025 following its launch in December 2024. The significant scale-up of IM8 reflects strong customer acquisition, expanding international presence and increased subscription adoption.
Revenue growth from continuing operations was further supported by the full-year contribution from Europa, acquired in August 2024, which expanded our fulfillment and distribution capabilities for sports nutrition products. In addition, revenue from genetic testing services, under the CircleDNA brand, increased year-over-year, contributing to the overall growth.
Prevention. Revenue from continuing operations derived from our Prevention segment increased by $2.6 million, or 25%, from $10.4 million for the year ended December 31, 2024 to $12.9 million for the year ended December 31, 2025. The increase was primarily attributable to a higher number of CircleDNA reports released during the year ended December 31, 2025, resulting in revenue recognition upon delivery of testing results, and to and revenue recognized from the expiration of unused test kits, which had previously been recorded as contract liabilities.
Consumer Health. Revenue from continuing operations derived from our Consumer Health segment increased by $73.9 million, or 1,327%, from $5.6 million for the year ended December 31, 2024 to $79.4 million for the year ended December 31, 2025. The increase was primarily driven by the strong performance of our consumer health product, IM8, which generated $60.1 million in revenue in its first full fiscal year following launch, as well as contributions from Europa's fulfillment and distribution services.
Direct Costs, Gross Profit and Gross Margin From Continuing Operations
Total direct costs from continuing operations increased by $36.9 million, or 555%, from $6.7 million for the year ended December 31, 2024 to $43.6 million for the year ended December 31, 2025. The increase was primarily attributable to the significant growth in our Consumer Health segment, particularly IM8, as well as the full-year contribution from Europa following its acquisition in August 2024. The increase reflects higher cost of inventories, logistics expenses, service-related charges, and staffing costs required to support the expanded scale of operations.
Gross profit from continuing operations increased by $39.5 million, or 426%, from $9.3 million for the year ended December 31, 2024 to $48.8 million for the year ended December 31, 2025. The increase was primarily driven by strong revenue growth in IM8, which generated $60.1 million in revenue during 2025, together with continued contributions from CircleDNA and Europa.
Gross margin from continuing operations decreased from 58% for the year ended December 31, 2024 to 53% for the year ended December 31, 2025. The decline in consolidated gross margin was primarily attributable to the revenue mix effect resulting from the inclusion of the lower-margin Europa distribution business. With the divestment of the Europa 3PL business completed in January 2026, we expect our blended gross margin profile to improve, assuming continued growth in our higher-margin Consumer Health products.
63
Other Income and Other Net Gains From Continuing Operations
Other income and other net gains from continuing operations decreased by $1.4 million, or 69%, from $2.0 million for the year ended December 31, 2024 to $0.6 million for the year ended December 31, 2025. The decrease was primarily attributable to lower bank interest income and other incidental income compared to the prior year.
Selling and Distribution Expenses From Continuing Operations
Selling and distribution expenses from continuing operations increased by $30.1 million, or 557%, from $5.4 million for the year ended December 31, 2024 to $35.5 million for the year ended December 31, 2025. The increase was primarily attributable to higher advertising and promotional expenses, customer acquisition costs, commission expenses and fulfillment-related costs associated with the rapid expansion of IM8 and the scaling of our Consumer Health segment.
Selling and marketing expenses from continuing operations represented 38% of revenue from continuing operations for the year ended December 31, 2025, compared to 34% in 2024, reflecting our deliberate investment in marketing initiatives to drive growth.
Research and Development Expenses From Continuing Operations
Research and development expenses from continuing operations decreased by $3.9 million, or 43%, from $9.1 million for the year ended December 31, 2024 to $5.1 million for the year ended December 31, 2025. The decrease was primarily attributable to reduced development activities compared to the prior year, which included product development activities at a greater scale.
Impairment Loss of Goodwill
An impairment loss of $6.8 million was recognized for the year ended December 31, 2025 in respect of goodwill allocated to the Europa cash-generating unit. The impairment was recognized as the carrying amount of the cash-generating unit exceeded its recoverable amount based on management's assessment. This impairment loss is a non-cash charge and does not impact our liquidity or cash flows. No impairment loss of goodwill was recognized for the year ended December 31, 2024.
Administrative and Other Operating Expenses From Continuing Operations
Administrative and other operating expenses from continuing operations increased by $13.3 million, or 40%, from $33.1 million for the year ended December 31, 2024 to $46.4 million for the year ended December 31, 2025. The increase was primarily attributable to higher professional fees, consultancy expenses, licensing and royalty expenses and general corporate expenses associated with the expansion of our Consumer Health segment and the operational scaling of the business during the year.
Fair Value Gain/(Loss) on Financial Assets at Fair Value Through Profit or Loss From Continuing Operations
Fair value changes on financial assets at fair value through profit or loss from continuing operations changed from a loss of $8.9 million for the year ended December 31, 2024 to a gain of $0.8 million for the year ended December 31, 2025. The movement reflects changes in the fair value of such financial assets during the reporting period. These fair value adjustments are non-cash in nature and do not impact our cash flows.
Gain on Warrant Exchange
Gain on warrant exchange was $36.7 million for the year ended December 31, 2025, compared to nil for the year ended December 31, 2024. The gain arose from the exchange of certain warrants for the Company’s ordinary shares during the year. The gain represents the difference between the carrying amount of the warrant liabilities derecognized and the fair value of the instruments issued at the exchange date. This is a non-cash item and does not impact our liquidity or cash flows.
64
Fair Value (Loss)/Gain on Warrant Liabilities From Continuing Operations
Fair value loss on warrant liabilities was $17.9 million for the year ended December 31, 2025, compared to a fair value gain of $49.0 thousand for the year ended December 31, 2024. The movement reflects changes in the fair value of warrants issued in connection with the de-SPAC and the placement transaction, which are measured at fair value through profit or loss. These fair value adjustments are non-cash in nature and do not affect our liquidity or cash flows.
Unrealized Fair Value Loss on Digital Assets
Unrealized fair value loss on digital assets was $9.7 million for the year ended December 31, 2025, compared to nil in 2024. The loss represents non-cash changes in the fair value of our digital assets, which primarily consist of Bitcoin holdings, measured based on quoted market prices at the reporting date. The fair value movements reflect volatility in market prices during the year and do not impact our operating cash flows.
Gain on Partial Disposal of an Equity-accounted Investee
No gain on partial disposal of an equity-accounted investee was recognized for the year ended December 31, 2025. In 2024, a gain of $1.2 million was recognized upon the sale of a portion of our interest in an investee accounted for using the equity method. The gain represented the difference between the consideration received and the proportionate carrying amount of the investment disposed.
Share of Loss of Equity-accounted Investees, net of tax
Share of loss of equity-accounted investees, net of tax, decreased by $0.7 million, or 37% from $2.0 million for the year ended December 31, 2024 to $1.3 million for the year ended December 31, 2025. This amount represents our proportionate share of the net losses incurred by investees accounted for under the equity method.
Other Finance Costs From Continuing Operations
Other finance costs from continuing operations remained relatively stable at $0.2 million for the year ended December 31, 2025, compared to to $0.2 million for the year ended December 31, 2024. These costs primarily represent interest expenses recognized on lease liabilities.
Income Tax (Expense)/Credit From Continuing Operations
Income tax credit from continuing operations changed from a credit of $7.6 million for the year ended December 31, 2024 to $33.6 thousand for the year ended December 31, 2025. The tax credit recognized in 2024 was primarily attributable to the reversal of an overprovision of income tax recorded in prior years. In 2025, the tax impact was not material and reflects the geographic mix of taxable income and losses during the year.
Profit/(Loss) From Discontinued Operations, Net of Tax
Loss from discontinued operations, net of tax, decreased from $11.4 million for the year ended December 31, 2024, to $3.7 million for the year ended December 31, 2025, representing an improvement of $7.7 million. The results for 2025 primarily reflect the operating results and disposal impact of ACT Genomics, which was classified as a discontinued operation in accordance with IFRS 5 and disposed of on October 1, 2025. The results for the year include both the operating results of ACT Genomics prior to disposal and the gain of $2.0 million recognized upon disposal of the subsidiary. In 2024, discontinued operations primarily related to the cessation of COVID-19-related diagnostic services and certain DNA testing operations in the EMEA region.
Other Comprehensive Income/(Expense)
Other comprehensive income was $0.6 million for the year ended December 31, 2025, compared to other comprehensive expense of $0.7 million for the year ended December 31, 2024.
The movement was primarily attributable to exchange differences arising from the translation of foreign operations, reflecting fluctuations in foreign currency exchange rates during the year. In addition, 2025 included a reclassification of cumulative translation reserves to profit or loss upon the disposal of foreign operations, as well as our share of other comprehensive income or expense of equity-accounted investees.
65
Non-IFRS Financial Measures
Management uses certain non-IFRS financial measures to supplement the financial measures prepared in accordance with IFRS Accounting Standards, which include adjusted EBITDA. Management believes that adjusted EBITDA is helpful to investors as they provide useful information to understand our core financial and operating performance from period to period because they exclude (1) depreciation and amortization, (2) interest income, (3) other finance costs, (4) income tax expense/(credit), (5) amortization of deferred expenses, (6) equity-settled share-based payment expenses, (7) non-recurring expenses related to acquisition, disposal and fundraising, (8) strategic realignment and discontinued products impact, (9) exchange gain or loss, net, (10) fair value loss on financial assets at fair value through profit or loss, (11) gain on warrant exchange, (12) fair value loss/(gain) on warrant liabilities, (13) unrealized fair value loss on digital asset, (14) gain on partial disposal of an equity-accounted investee, (15) share of loss of equity-accounted investees, net of tax, (16) impairment loss of goodwill, and (17) (profit)/loss from discontinued operations, net of tax. These adjustments are made for items that may not be indicative of our business performance, including non-cash and/or non-recurring items.
In addition to our results determined in accordance with IFRS Accounting Standards, we present the non-IFRS measure as supplemental information. We believe this measure is useful to management and investors in evaluating our operating performance, as it excludes certain items that we do not consider indicative of our ongoing operating results. We use this measure for internal planning, forecasting and performance evaluation purposes. We believe this non-IFRS financial measure may provide investors with additional information regarding our results of operations and enhance consistency and comparability with our historical financial performance. However, this measure may not be comparable to similarly titled measures provided by other companies, as other companies may calculate similarly titled measures differently. This non-IFRS financial measure is presented for supplemental informational purposes only and should not be considered in isolated from, or as a substitute for, financial information prepared and presented in accordance with IFRS Accounting Standards. This measure has limitations as an analytical tool and should not be viewed as an alternative to loss for the year, loss before taxation or any other performance measure derived in accordance with IFRS Accounting Standards.
66
The tables below reconciles adjusted EBITDA to its most directly comparable IFRS Accounting Standards measure for the periods presented. Investors are encouraged to review the related IFRS Accounting Standards financial measures and the reconciliations of this non-IFRS measure to its most directly comparable IFRS Accounting Standards financial measure.
| Year Ended | |||||||||||
| December 31, | December 31, | ||||||||||
| 2025 | 2024 | ||||||||||
| (Unaudited) | (Unaudited) | ||||||||||
| Loss for the year under IFRS Accounting Standards | $ | (39,978) | $ | (49,806) | |||||||
| Depreciation and amortization | 2,340 | 2,479 | |||||||||
| Interest income | (1,042) | (1,846) | |||||||||
| Other finance costs | 241 | 168 | |||||||||
| Income tax expense/(credit) | 34 | (7,639) | |||||||||
| EBITDA (Non-IFRS) | (38,405) | (56,644) | |||||||||
| Amortization of deferred expenses | 3,549 | 8,294 | |||||||||
| Equity-settled share-based payment expenses | 6,354 | 5,842 | |||||||||
| Non-recurring expenses related to acquisition, disposal and fundraising | 7,952 | 3,605 | |||||||||
| Strategic realignment and discontinued products impact | 3,761 | 173 | |||||||||
| Exchange gain or loss, net | 838 | (8) | |||||||||
| Fair value loss on financial assets at fair value through profit or loss | (780) | 8,869 | |||||||||
| Gain on warrant exchange | (36,657) | — | |||||||||
| Fair value loss/(gain) on warrant liabilities | 17,943 | (49) | |||||||||
| Unrealized fair value loss on digital asset | 9,725 | — | |||||||||
| Gain on partial disposal of an equity-accounted investee | — | (1,244) | |||||||||
| Share of loss of equity-accounted investees, net of tax | 1,260 | 2,010 | |||||||||
| Impairment loss of goodwill | 6,815 | — | |||||||||
| Loss from discontinued operations, net of tax | 3,731 | 11,420 | |||||||||
| Adjusted EBITDA (Non-IFRS) | $ | (13,914) | $ | (17,732) | |||||||
67
| Three Months Ended | |||||||||||
| December 31, | December 31, | ||||||||||
| 2025 | 2024 | ||||||||||
| (Unaudited) | (Unaudited) | ||||||||||
| Loss for the period under IFRS Accounting Standards | $ | (7,542) | $ | (17,543) | |||||||
| Depreciation and amortization | 493 | 668 | |||||||||
| Interest income | (210) | (522) | |||||||||
| Other finance costs | 45 | 86 | |||||||||
| Income tax expense/(credit) | 37 | (7,440) | |||||||||
| EBITDA (Non-IFRS) | (7,177) | (24,751) | |||||||||
| Amortization of deferred expenses | — | 2,099 | |||||||||
| Equity-settled share-based payment expenses | 1,299 | 1,176 | |||||||||
| Non-recurring expenses related to acquisition, disposal and fundraising | 4,366 | 1,781 | |||||||||
| Strategic realignment and discontinued products impact | 3,750 | 10 | |||||||||
| Exchange gain or loss, net | 199 | (546) | |||||||||
| Fair value (gain)/loss on financial assets at fair value through profit or loss | (879) | 8,728 | |||||||||
| Gain on warrant exchange | (36,657) | — | |||||||||
| Fair value loss/(gain) on warrant liabilities | 17,339 | (31) | |||||||||
| Unrealized fair value loss/(gain) on digital asset | 9,725 | — | |||||||||
| Gain on partial disposal of an equity-accounted investee | — | (1,244) | |||||||||
| Share of loss of equity-accounted investees, net of tax | 196 | 961 | |||||||||
| Impairment loss of goodwill | 6,815 | — | |||||||||
| (Profit)/loss from discontinued operations, net of tax | (2,153) | 4,202 | |||||||||
| Adjusted EBITDA (Non-IFRS) | $ | (3,177) | $ | (7,615) | |||||||
B.Liquidity and Capital Resources
We have historically financed our operations primarily through the issuance of ordinary shares, proceeds from capital markets transactions and cash generated from investing activities. Our primary requirements for liquidity and capital are to finance working capital, capital expenditures and general corporate purposes, as well as investment in research and development and potential mergers and acquisition opportunities.
As of December 31, 2025 and 2024, our principal source of liquidity was our cash balance of $32.1 million and $52.3 million, respectively. The decrease in cash balance year-over-year was primarily attributable to operating cash outflows and investments in digital assets and financial assets, partially offset by proceeds from financing activities and disposal transactions. We incurred a net loss after tax of $40.0 million and $49.8 million for the years ended December 31, 2025 and 2024, respectively. Net cash used in operating activities was $21.8 million in 2025, compared to $28.9 million in 2024, reflecting continued operating losses and working capital investments to support business expansion, partially offset by improved revenue scale.
Between Prenetics HK and its subsidiaries, the cash is transferred from Prenetics HK to its subsidiaries in the form of capital contributions or through intercompany advances. If needed, cash may be transferred between Prenetics HK and its subsidiaries in the United States, Taiwan and Singapore through intercompany fund advances and capital contributions, and there are currently no restrictions on transferring funds between Prenetics HK and its subsidiaries in the United States, Taiwan and Singapore. Under our cash management policy, the amount of intercompany transfer of funds is determined based on the working capital needs of the subsidiaries and intercompany transactions, and is subject to internal approval process and funding arrangements. Our management reviews and monitors our cash flow forecast and working capital needs of the subsidiaries on a regular basis.
68
Assuming the exercise of all outstanding Warrants for cash, we would receive aggregate proceeds of approximately $154.6 million. However, we will only receive such proceeds if all the Warrant holders exercise all of their Warrants. The exercise price of our Warrants was $8.91 per 1.29 shares (or an effective price of $6.91 per share), subject to adjustment. After completion of our reverse stock split of 15-to-1 in November 2023, the exercise price of our Warrants has been adjusted to $133.65 per 1.29 shares (or an effective price of $103.60 per share). Assuming the exercise of all outstanding Class A Warrants, Class B Warrants, Class C Warrants and Placement Agent Warrants, we would receive aggregate proceeds of approximately $65.0 million (subject to adjustment of exercise prices). However, we will only receive such proceeds if all the holders of Class A Warrants, Class B Warrants, Class C Warrants and Placement Agent Warrants exercise all of their such warrants. We believe that the likelihood that warrant holders determine to exercise their warrants, and therefore the amount of cash proceeds that we would receive, is dependent upon the market price of our Class A Ordinary Shares. If the market price for our Class A Ordinary Shares is less than the exercise price of the warrants (on a per share basis), we believe that warrant holders will be very unlikely to exercise any of their warrants, and accordingly, we will not receive any such proceeds. There is no assurance that the warrants will be “in the money” prior to their expiration or that the warrant holders will exercise their warrants. Holders of the Private Warrants have the option to exercise the Private Warrants on a cashless basis in accordance with the Existing Warrant Agreement. Holders of our Class A Warrants, Class B Warrants, Class C Warrants and Placement Agent Warrants have the option to exercise such warrants on a cashless basis in accordance with the terms of such warrants. To the extent that any warrants are exercised on a cashless basis, the amount of cash we would receive from the exercise of the warrants will decrease.
On December 30, 2022, we acquired 74.39% of equity interest in ACT Genomics for a total consideration of $10 million in cash and 19,891,910 Class A Ordinary Shares (which were subsequently consolidated into approximately 1,326,128 Class A Ordinary Shares, due to the reverse stock split). On October 1, 2025, we sold our equity interest in ACT Genomics to Delta Electronics, Inc. for up to approximately $72 million in cash (of which approximately $46 million are gross proceeds to Prenetics).
On July 20, 2023, we initiated a 50/50 equity-accounted investee, Insighta. This establishment involved a total consideration of $80 million in cash and 22,222,222 Class A Ordinary Shares (which were subsequently consolidated into 1,481,482 Class A Ordinary Shares, due to the reverse stock split). On October 14, 2024, our shareholding in Insighta was reduced to 35% following a disposal of 15% interest for a consideration of $30 million to Tencent. On February 17, 2026, we sold our remaining 35% equity stake in Insighta to Tencent for $70 million in cash.
On January 1, 2026, we entered into an Asset Purchase Agreement with an independent buyer pursuant to which we divested substantially all of the assets of the logistics, fulfillment, warehousing and distribution business operated under Europa Sports Partners, LLC. The aggregate consideration comprises share consideration in the buyer’s parent, consisting of (i) closing shares with an aggregate value of approximately $3.0 million, subject to a vesting condition linked to specified performance milestones, and (ii) contingent share consideration of up to $10.0 million, payable in tranches upon achievement of additional performance targets during the earn-out period. The performance milestones are primarily linked to the operational and commercial performance of the disposed business, including shipment volume–based targets and referral revenue generated under post-closing commercial arrangements. The buyer also assumed certain liabilities associated with the transferred business relating to the assigned contracts and operations subsequent to the closing date.
In addition, during the year ended December 31, 2025, we deployed a portion of our capital into financial assets at fair value through profit or loss and digital assets, primarily Bitcoin, as part of our broader capital allocation strategy. During the year, we invested approximately $54.4 million in digital assets and $20.0 million in financial assets at fair value through profit or loss. As of December 31, 2025, the carrying amount of our digital assets was approximately $44.6 million. Investments in financial assets at fair value through profit or loss are generally made with the objective of achieving returns on surplus cash and may include equity or other marketable instruments. Our investment in digital assets reflects a treasury diversification approach and is subject to significant market price volatility. These investments are non-operating in nature and are measured at fair value, with changes in fair value recognized in profit or loss or other comprehensive income. As a result, they may introduce volatility to our reported earnings that is not directly related to our core operating performance. While these assets are generally liquid, their realizable value may fluctuate significantly depending on prevailing market conditions, which may impact the timing and amount of liquidity available if we elect to monetize such positions. We continuously monitor our investment portfolio and may adjust our holdings from time to time based on market conditions, liquidity requirements and risk considerations.
We believe our existing cash and cash equivalents, together with proceeds from financing activities, will be sufficient to meet our operating working capital requirements, and capital expenditures at least for the next twelve months. Our future liquidity and capital requirements will depend on a number of factors, including the growth and performance of our
69
business, the level of investment in marketing and inventory, and the timing and scale of any strategic investments or acquisitions.
While we currently do not have any committed external financing arrangements or agreements for future investments or acquisitions, we may seek additional capital through equity or debt financing if required to support our strategic initiatives. Our ability to obtain additional financing will depend on market conditions, investor demand and our financial performance.
Cash flows from operations may also be affected by customer demand, and other risks described under “Item 3. Key Information — D. Risk Factors.” We intend to maintain financing flexibility and access to the capital markets where appropriate to support the execution of our long-term growth strategy.
The following table summarizes our cash flows for the periods presented:
Year Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| (in thousands of U.S. dollars) | |||||||||||||||||
| Net cash used in operating activities | (21,826) | (28,874) | (13,765) | ||||||||||||||
| Net cash (used in)/from investing activities | (35,694) | 38,541 | (82,952) | ||||||||||||||
| Net cash from/(used in) financing activities | 37,467 | (3,343) | (4,704) | ||||||||||||||
Operating Activities
Net cash used in operating activities was $21.8 million for the year ended December 31, 2025, compared to $28.9 million for the year ended December 31, 2024. The operating cash outflow in 2025 was primarily driven by a net loss of $40.0 million, which included several significant non-cash items. These non-cash adjustments included an unrealized fair value loss on digital assets of $9.7 million, an impairment loss of goodwill of $6.8 million, equity-settled share-based payment expenses of $7.6 million, depreciation of $2.7 million, amortization of intangible assets of $1.1 million, share of loss of equity-accounted investees of $2.1 million, write-off on inventories of $0.7 million, fair value loss on financial assets at fair value through profit or loss of $0.8 million, fair value loss on warrant liabilities of $17.9 million, and other finance costs of $0.3 million, partially offset by gain on warrant exchange of $36.7 million, gain on disposal of a subsidiary of $2.0 million and bank interest income of $1.1 million. These items do not have an immediate impact on cash flows and may vary from period to period depending on market conditions, valuation assumptions and the performance of investees.
Changes in operating assets and liabilities resulted in a net cash outflow of $9.1 million for the year ended December 31, 2025. This was primarily attributable to increases in inventories of $2.8 million, an increase in deposits, prepayments and other receivables of $3.8 million, reflecting inventory build-up and upfront operating expenditures to support the expansion of our Consumer Health business. These outflows were partially offset by increases in accrued expenses and other current liabilities of $15.5 million and increase in trade payables of $0.3 million, which reflect the timing of vendor payments and the scaling of operations. In addition, contract liabilities decreased by $3.3 million during the year, primarily due to the recognition of revenue upon the satisfaction of performance obligations associated with previously contract liabilities. Compared to the prior year, the reduction in net cash used in operating activities reflects the continued scaling of our revenue base and operating efficiencies, notwithstanding ongoing investments to support growth.
Net cash used in operating activities for the year ended December 31, 2024 was $28.9 million, primarily driven by a loss for the year of $49.8 million, adjusted for non-cash items. These adjustments included fair value loss on financial assets at fair value through profit or loss of $8.9 million, equity-settled share-based payment expenses of $7.8 million, depreciation of $4.0 million, amortization of intangible assets of $1.9 million, share of loss of equity-accounted investees of $1.8 million, write-off on inventories of $0.7 million, bank interest income of $2.0 million, and other finance costs of $0.2 million.
Changes in operating assets and liabilities resulted in a net cash outflow of $2.0 million, primarily due to a decrease in deposits and prepayments and other receivables of $2.1 million, a decrease in trade receivables of $0.6 million, and an increase in inventories of $1.3 million, partially offset by a decrease in trade payables of $4.7 million, a decrease in accrued expenses and other current liabilities of $0.9 million, an increase in contract liabilities of $0.4 million, and a decrease in deferred expenses of $8.3 million.
70
Investing Activities
Net cash used in investing activities was $35.7 million for the year ended December 31, 2025, compared to net cash provided by investing activities of $38.5 million for the year ended December 31, 2024. Investing cash outflows in 2025 were primarily attributable to $0.2 million for the purchase of property, plant and equipment, $54.4 million used for the purchase of digital assets, primarily Bitcoin, and $20.0 million invested in financial assets at fair value through profit or loss, reflecting our capital allocation strategy and investment in digital assets. These outflows were partially offset by $37.8 million of net cash inflow from the disposal of a subsidiary, and $1.1 million in interest income received. The shift from net inflows in 2024 to net outflows in 2025 reflects a transition from divestment-related activities to investment deployment and treasury diversification.
Net cash provided by investing activities for the year ended December 31, 2024 was primarily attributable to $30.0 million in proceeds from the partial disposal of an equity-accounted investee and $16.0 million from the redemption of short-term deposits, partially offset by $8.3 million paid for acquisition of a business, and $1.0 million paid for the purchase of property, plant and equipment.
Financing Activities
Net cash provided by financing activities was $37.5 million for the year ended December 31, 2025, compared to net cash used in financing activities of $3.3 million for the year ended December 31, 2024. Financing inflows in 2025 were primarily attributable to $40.2 million in proceeds from a public placement, which strengthened our liquidity position and provided additional capital to support the expansion of our Consumer Health business and other strategic initiatives. These inflows were partially offset by $2.4 million in capital element of lease rentals paid and $0.3 million in interest element of lease rentals paid. The financing inflow in 2025 represents a non-recurring capital raising activity and may not be indicative of future financing cash flows.
Net cash used in financing activities for the year ended December 31, 2024 primarily consisted of $2.6 million in capital element of lease rentals paid, $0.2 million in interest element of lease rentals paid, and $0.6 million in payments for purchase of treasury shares.
Contractual and Other Obligations
Other than the ordinary cash requirements for our operations and our capital expenditure, our material cash requirements as of December 31, 2025 and any subsequent interim period primarily include lease liabilities, warrant liabilities, and liabilities for puttable financial instruments. The following table sets forth their details as of December 31, 2025:
| Payment Due by Period | |||||||||||||||||
| Total | Less than 1 year | 1 – 2 Years | |||||||||||||||
(in thousands of U.S. dollars) | |||||||||||||||||
| Lease liabilities | 1,876 | 1,428 | 448 | ||||||||||||||
| Warrant liabilities | 20,319 | 20,319 | — | ||||||||||||||
Off-Balance Sheet Arrangements
Since the date of our incorporation, we have not engaged in any off-balance sheet arrangements, as defined in the rules and regulations of the SEC.
C.Research and Development, Patents and Licenses, Etc.
See “Item 4. Information on the Company — B. Business Overview — Research and Development” and “Item 4. Information on the Company — B. Business Overview — Intellectual Property.”
D.Trend Information
Other than as disclosed elsewhere in this annual report, we are not aware of any trends, uncertainties, demands, commitments or events since January 1, 2025 to December 31, 2025 that are reasonably likely to have a material adverse
71
effect on our net revenues, income, profitability, liquidity or capital resources, or that caused the disclosed financial information to be not necessarily indicative of future operating results or financial conditions.
E.Critical Accounting Estimates
Our consolidated financial statements are prepared in accordance with IFRS Accounting Standards, and the preparation of these consolidated financial statements requires us to make estimates that affect the application of the Group's accounting policies and the reported amounts of assets, liabilities, revenue, costs and expenses. Actual results may differ from these estimates. Estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to estimates are recognized prospectively.
Impairment Test of Cash Generating Units Containing Goodwill and Intangible Assets
Internal and external sources of information are reviewed at the end of each reporting period to identify indications that the following assets may be impaired or, except in the case of goodwill, an impairment loss previously recognized no longer exists or may have decreased:
•intangible assets; and
•goodwill
If any such indication exists, the asset’s recoverable amount is estimated. In addition, for goodwill, intangible assets that are not yet available for use and intangible assets that have indefinite useful lives, the recoverable amount is estimated annually whether or not there is any indication of impairment.
Calculation of recoverable amount
The recoverable amount of an asset is the greater of its fair value less costs of disposal and value in use. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset. Where an asset does not generate cash inflows largely independent of those from other assets, the recoverable amount is determined for the smallest group of assets that generates cash inflows independently (i.e. a cash-generating unit).
The determination of recoverable amount involves significant judgement and the use of estimates. In particular, value-in-use calculations require management to make assumptions regarding future cash flows, including expected revenue growth, operating margins, terminal growth rates and appropriate discount rates. These assumptions are based on management’s expectations of future business performance and market conditions.
Where fair value less costs of disposal is applied, the determination of recoverable amount may involve the use of valuation techniques based on market participant assumptions, including probability-weighted assessments of expected future consideration where relevant. Such valuations may incorporate both fixed and contingent share consideration linked to the achievement of specified performance milestones.
Management exercised significant judgement in determining the appropriate valuation methodology for certain cash-generating units, including the use of fair value less costs of disposal where appropriate. This assessment required consideration of the Group’s strategic direction, including decision to divest certain businesses, as well as market participant assumptions.
The estimation of recoverable amount is inherently uncertain and sensitive to changes in key assumptions. Changes in future operating performance, discount rates, probability of achieving performance milestones or market conditions could result in material changes to the recoverable amount and may lead to impairment charges in future periods.
Recognition of impairment losses
An impairment loss is recognized in profit or loss if the carrying amount of an asset, or the cash-generating unit to which it belongs, exceeds its recoverable amount. Impairment losses recognized in respect of cash-generating units are allocated first to reduce the carrying amount of any goodwill allocated to the cash-generating unit (or group of units) and then, to reduce the carrying amount of the other assets in the unit (or group of units) on a pro rata basis, except that the carrying value of an asset will not be reduced below its individual fair value less costs of disposal (if measurable) or value in use (if determinable).
72
Fair Value of Warrant Liabilities
The Group’s warrant liabilities are classified as financial liabilities and measured at fair value through profit or loss. The fair value of these instruments is determined using valuation techniques, including binomial option pricing models and Monte Carlo simulation models, due to the absence of quoted market prices for certain warrants.
The valuation of warrant liabilities requires the use of significant estimates and assumptions, including the Company’s share price, expected volatility, expected term and risk-free interest rates. The Group applies consistent valuation methodologies based on market-observable data where available, though these assessments involve inherent estimation.
Changes in these assumptions, particularly fluctuations in the Company’s share price and market volatility, are recorded as non-cash gains or losses in the consolidated statement of profit or loss from period to period. The Group monitors these valuations to ensure they reflect current market data in accordance with relevant accounting standards.
ITEM 6. DIRECTORS, SENIOR MANAGEMENT AND EMPLOYEES
A.Directors and Senior Management
The following table sets forth certain information relating to our executive officers and directors as of the date of this annual report. Our board of directors is comprised of four directors.
The terms of two former members of the Board, Ms. Chiu Wing Kwan Winnie, an independent director and member of our Audit Committee, the Compensation Committee and the Nominating and Corporate Government Committee, and Ms. Kathryn Henry, an independent director, ended on May 17, 2025 and June 12, 2025 respectively and were not renewed. The decision did not arise or result from any disagreement between Ms. Chiu Wing Kwan Winnie and Ms. Kathryn Henry with the Company. We appointed Kong Yiu (Andy) Cheung as an independent director to our Board of Directors and as a member of the Audit Committee, effective June 16, 2025.
Mr. Kong Yiu (Andy) Cheung resigned from his position as director of the Company, effective February 16, 2026. Mr. Cheung’s decision to step down followed the Company’s strategic shift to focus its resources and management attention entirely on the continued global expansion and scaling of its flagship health and longevity brand, IM8. The decision did not arise or result from any disagreement between Mr. Cheung and the Company. The Company appointed Dr. Darshan Shah to its Board as an independent director and member of the Audit Committee, Compensation Committee, and Nominating and Corporate Governance Committee, effective February 16, 2026.
| Name | Age | Position/Title | ||||||||||||
Danny Sheng Wu Yeung | 47 | Director, Chairperson and Chief Executive Officer | ||||||||||||
| Cheng Yin Pan (Ben) | 38 | Independent Director | ||||||||||||
Dr. Darshan Shah | 53 | Independent Director | ||||||||||||
| David Eric Vanderveen | 57 | Director and President of Americas | ||||||||||||
| Lo Hoi Chun (Stephen) | 41 | Chief Financial Officer | ||||||||||||
Danny Sheng Wu Yeung is the co-founder of Prenetics and has served as Chief Executive Officer and Chairman of the Board since the Company's inception in 2014. Mr. Yeung also serves as co-founder and Chief Executive Officer of IM8, a premium health and longevity brand co-founded with David Beckham and wholly owned by Prenetics. Under Mr. Yeung's leadership, Prenetics performed 28 million COVID-19 tests and generated approximately $700 million in revenue during the pandemic, and subsequently completed a public listing on the Nasdaq Stock Exchange. Prior to Prenetics, Mr. Yeung was a founding partner at SXE Ventures, where he led investments in genetic testing companies and in Honey Science Corporation, which was acquired by PayPal Holdings, Inc. for approximately $4 billion in 2019. Mr. Yeung founded uBuyiBuy in 2010, which was subsequently acquired by Groupon, Inc. Prior to leaving Groupon in early 2014, Mr. Yeung served as Chief Executive Officer of Groupon East Asia, establishing it as the largest e-commerce platform in the region. Mr. Yeung's earlier ventures include franchising Hong Kong dessert chain Hui Lau Shan into the United States and operating a hospitality furniture business that executed multi-million-dollar projects for MGM Resorts International.
Cheng Yin Pan (Ben) is the Chairman of the Board of Directors and Chief Executive Officer of Youngtimers AG, a Switzerland-listed global asset management firm (SIX: YTME), since January 2025, and CEO of C Capital since March 2016. Under his leadership, C Capital has made significant strides in the investment landscape, focusing on transformative opportunities across various sectors. With extensive experience in venture capital and private equity, Mr. Cheng has played
73
a pivotal role in high-profile investments, including CASETiFY, the fastest-growing global tech accessories brand; XiaoPeng (NYSE: XPEV), a leading Chinese electric vehicle manufacturer; Animoca brands, a multinational blockchain technology; NOTHING, a U.K.-based consumer electronics manufacturer; and Lalamove, a prominent Chinese on-demand logistics firm. Mr. Cheng is an independent director of Prenetics and holds a non-executive director position at Ocean Line Port Development Limited (8502.HK). In addition to his operational roles, Mr. Cheng actively contributes to the community as a committee member of the Hong Kong Trade Development Council Mainland Business Advisory Committee and the Venture Committee of Hong Kong Venture Capital and Private Equity Association. Prior to C Capital, Mr. Cheng honed his expertise as an investment banker at Bank of Americal Merrill Lynch and Standard Chartered Bank, where he was involved in landmark transactions such as the US$3.3 billion take-private of WuXi Pharma Tech, Temasek’s US$5.7 billion investment in Watson’s and the US$2.1 billion acquisition of ING’s insurance business in Hong Kong, Macau and Thailand. Mr. Cheng holds a bachelor’s degree in Quantitative Finance with honors from the Chinese University of Hong Kong. He is a member of CPA Australia.
Dr. Darshan Shah is a board-certified surgeon and a recognized expert in longevity medicine with over 20 years of experience in clinical practice and healthcare entrepreneurship. He currently serves as an independent director of Prenetics. Dr. Shah earned his medical degree at the age of 21 from the University of Missouri, Kansas City, and completed his surgical training at the Mayo Clinic. During his career as a specialist in trauma and reconstructive surgery, he personally performed over 20,000 surgical procedures. Since 2016, Dr. Shah has served as the Founder and Chief Executive Officer of Next Health, one of the world's premier health optimization and longevity centers. In addition to his role at Next Health, Dr. Shah is the host of the EXTEND Podcast, where he engages with leading scientists on advancements in health, wellness and longevity. He is an alumnus of Harvard Business School and Singularity University, and has authored numerous peer-reviewed articles in academic medical journals. In recent years, Dr. Shah has transitioned his clinical focus to longevity and healthspan optimization, advising thousands of patients on data-driven wellness protocols. This deep medical foundation provides the Board with critical technical oversight as Prenetics continues to scale its science-backed product ecosystem, including the IM8 brand.
David Eric Vanderveen has had a successful 30-year track record in beverage, nutrition, and technology, leading disruptive change at both private and public companies. He currently serves as our director and President of Americas. From 2022 until recently, he was CEO of Nirvana Water Sciences, where he developed a team of industry beverage, functional beverage, and supplement experts to transform the brand, product portfolio, go-to-market strategy, and execution of the business. From 2002 to 2015, Mr. Vanderveen was the founder and CEO of XS Worldwide, a company with a portfolio of energy drinks and sports nutrition products. XS partnered with Amway Corporation and became the fastest-growing business segment and category for the world’s largest direct seller, reaching annual sales of over $400 million with distribution in more than 60 countries. XS was sold to Amway in 2015, where Mr. Vanderveen served as Vice President and Global General Manager for the XS brand, achieving maximum earn-out through 2018. Mr. Vanderveen is also an advisor and limited partner at RX3 Growth Partners, a private equity firm that partners with consumer brands and other leading investment firms to maximize value by leveraging its unique influencer base of athletes, celebrities, and thought leaders, and is a general partner in LLB Global Fund, an international arbitration fund in partnership with French law firm, Lazareff Le Bars.
Lo Hoi Chun (Stephen) has served as our Chief Financial Officer since 2018, and oversees our financial operations, corporate accounting and reporting, treasury, financial and tax planning and analysis, and investor relations. Prior to joining us, Mr. Lo served as the Vice President in the Asia Pacific Investment Banking team of Citigroup, where he worked extensively on initial public offering transactions, placements, debt issuances and cross border mergers and acquisitions in Asia and the United States. Previously, Mr. Lo was an auditor with Ernst & Young. Mr. Lo received a Master of Business Administration from Yale University’s School of Management, a Master of Science in Accounting and Finance from the London School of Economics and Political Science, and a bachelor’s degree in Accounting from Hong Kong Baptist University. Mr. Lo is a Fellow of the Hong Kong Institute of Certified Public Accountants, a Chartered Accountant of the Institute of Chartered Accountants in England and Wales (ICAEW) and a CFA Charter Holder. He serves on the Hong Kong Committee of ICAEW.
B.Compensation
Compensation of Directors and Executive Officers
In 2025, we paid an aggregate of US$5.91 million and US$3.94 million in cash compensation and benefits in kind to our directors and executive officers as a group, respectively. Our directors and executive officers do not receive pension, retirement or other similar benefits, and we have not set aside or accrued any amount to provide such benefits to our
74
executive officers. Our subsidiary in Hong Kong is required by the applicable local laws and regulations to make contributions to Mandatory Provident Fund.
Employment Agreements and Indemnification Agreements
Each director has entered into a director service agreement with us. The director service agreement provides for an initial term of engagement of one year, with automatic one-year renewals, upon mutual agreement between the parties on the terms and conditions of such renewal, and subject to early termination by either party with or without cause at any time upon a 30-day notice. The director service agreement also includes certain restrictive covenants, which include confidentiality and non-disclosure restrictions, non-competition and non-solicitation restrictions that apply during the term and for certain periods following specified terminations of employment, an inventions assignment provision, and certain rights to indemnification by us.
Each of the executive officers is party to an employment agreement with Prenetics Limited or IM8 (US) LLC, which are our wholly-owned subsidiaries. Under these agreements, the employment of each executive officer is for a specified time period, and may be terminated for cause, at any time, for certain acts of the executive officer, such as continued failure to satisfactorily perform, willful misconduct or gross negligence in the performance of agreed duties, conviction or entry of a guilty or nolo contendere plea of any felony or any misdemeanor involving moral turpitude, or dishonest act that results materially to our detriment or material breach of the employment agreement. The employment may also be terminated without cause upon 90 days’ advance written notice. The executive officer may resign at any time with a 90-day advance written notice.
The employment agreements with the other executive officers also include confidentiality and nondisclosure restrictions and non-competition and non-solicitation restrictions that apply during employment and for certain periods following termination of employment.
We have entered into indemnification agreements with each of our directors. Under these agreements, we have agreed to indemnify our directors against certain liabilities and expenses incurred by such persons in connection with claims made by reason of their being our director.
Share Incentive Plans
Prenetics 2021 Plan
In June 2021, Prenetics’ board of directors adopted the 2021 Share Incentive Plan of Prenetics, or Prenetics 2021 Plan, which provides for the issuance of up to 14,814,113 shares (equivalent to approximately 987,608 shares after the reverse stock split) pursuant to all awards, including shares underlying the 2014 and 2016 Option Scheme and the 2017 Restricted Share Scheme.
In addition, in connection with the Business Combination, all 17,549,116 RSUs with respect to the ordinary shares of Prenetics, par value $0.0001 per share (“Prenetics Ordinary Shares”) that were outstanding under the Prenetics 2021 Plan at the time of consummation of the Business Combination were replaced with 17,549,116 RSUs with respect to Class A Ordinary Shares (and in the case of Danny Yeung, Class B Ordinary Shares) under the 2022 Plan.
The 2022 Plan
On May 18, 2022, we adopted the 2022 Share Incentive Plan, or the 2022 Plan, which became effective on the same day. The following summarizes the material terms of the 2022 Plan:
Shares Subject to the Plan. Initially, the maximum number of ordinary shares that may be issued under the 2022 Plan is (a) 16,479,399 (consolidated into 1,098,627 due to the 15:1 reverse stock split), which will be increased on the first day of each calendar year beginning in the year immediately following closing of the Business Combination and during the term of the 2022 Plan, in an amount equal to the lesser of (i) three percent (3%) of the total number of shares issued and outstanding on an as-converted fully-diluted basis on the last day of the immediately preceding fiscal year and (ii) such number of shares determined by our board of directors, plus (b) the number of shares reserved for issuance in accordance with an employee share purchase program (the “Employee Share Purchase Program”) to be adopted by a committee consisting of one or more members of our board of directors (the “Committee”) following the consummation of the Business Combination. The maximum number that may be issued subject to RSUs with respect to our ordinary shares (“Prenetics RSUs”) granted pursuant to the Employee Share Purchase Program is 3,295,880 (consolidated into 219,726 due
75
to the 15:1 reverse stock split), which will automatically increase on the first day of each calendar year for a period of not more than ten years from the Acquisition Merger Effective Date, in an amount equal to the lesser of (a) one percent (1%) of our fully-diluted share capital on the last day of the immediately preceding calendar year or (b) such small number determined by the Committee.
If an award terminates, expires, or lapses for any reason without having been exercised or settled in full, the number of shares subject to the award shall again be available for the grant of an award pursuant to the 2022 Plan. If any award is forfeited or repurchased, the shares underlying such award may again be granted or awarded under the 2022 Plan, provided that if an award granted pursuant to the Employee Share Purchase Program terminates, expires, or lapses for any reason without having been settled in full, the shares subject to such award shall only may again be available for the grant of an award pursuant to the Employee Share Purchase Program.
Capitalization Adjustment. In the event there is a specified type of change in our capital structure, such as a dividend, share split, reverse share split, combination or exchange of shares, amalgamation, arrangement or consolidation, spin-off, recapitalization or other distribution (other than normal cash dividends), appropriate adjustments will be made to (i) the aggregate number and type of shares that may be issued under the 2022 Plan, (ii) the terms and conditions of any outstanding awards (including, without limitation, any applicable performance targets or criteria with respect thereto), (iii) the grant or exercise price per share for any outstanding awards under the 2022 Plan, and (iv) in the case of a spin-off, the additional number and type of shares (including shares in the entities being spun-off) that shall be issued or an appropriate decrease of exercise price in connection with the spin-off.
Types of Awards. The 2022 Plan permits the awards of options, share appreciation rights, restricted shares, RSUs and other awards approved by the plan administrator or the board of directors.
Eligibility. We may grant awards to our employees, directors and consultants, and our subsidiaries. However, we may grant options that are intended to qualify as incentive share options only to our employees and our subsidiaries.
Plan Administration. The 2022 Plan shall be administered by a committee of one or more members of our board of directors and/or one or more of our executive officers delegated by our board of directors. The administrator determines the participants to receive awards, when and how awards will be granted, the type of award to be granted, the number of awards to be granted, and the other terms and conditions of each award. The administrator may delegate certain authorities under the 2022 Plan to our Chief Executive Officer.
Award Agreements. Awards granted under the 2022 Plan are evidenced by award agreements that set forth, consistent with the 2022 Plan, the terms, conditions and limitations for each award, the provisions applicable in the event that the grantee’s employment or service terminates, and our authority to unilaterally or bilaterally amend, modify, suspend, cancel or rescind the award.
Vesting Schedule. In general, the plan administrator determines the vesting schedule, which is specified in the relevant award agreement.
Conditions of Awards. The administrator determines the provisions, terms and conditions of each award granted under the 2022 Plan, including but not limited to the vesting schedule of the awards.
Termination. Unless terminated earlier, the 2022 Plan has a term of ten years from the date of its effectiveness. With the approval of our board of directors, the 2022 Plan can be terminated at any time; provided, however, no such termination shall adversely affect in any material way any awards previously granted without the prior written consent of the participant.
As of April 29, 2026, Prenetics RSUs underlying 388,036 Class A Ordinary Shares and 53,953 Class B Ordinary Shares were outstanding under the 2022 Plan.
RSU
As of April 29, 2026, there were a total of 78,161 Class A Ordinary Shares and 53,953 Class B Ordinary Shares underlying grants of outstanding RSUs that were held by the directors and executive officers as a group. The following
76
table summarizes, as of April 29, 2026, the number of outstanding ordinary shares underlying outstanding RSUs that we granted to our directors and executive officers.
| Name | Number of Ordinary Shares Underlying Outstanding RSUs | Date of Grant | ||||||||||||
Danny Sheng Wu Yeung | 53,953 | June 23, 2023 | ||||||||||||
Cheng Yin Pan (Ben)(1) | 15,873 | July 15, 2025 | ||||||||||||
David Eric Vanderveen | 8,681 | July 15, 2025 | ||||||||||||
Dr. Darshan Shah | 5,599 | February 16, 2026 | ||||||||||||
| Lo Hoi Chun (Stephen) | 32,736 | June 23, 2023 | ||||||||||||
| 15,272 | July 15, 2025 | |||||||||||||
_______________
Note:
(1)Represents outstanding RSUs granted to M13 Capital Management Holdings Limited, a British Virgin Islands company. M13 Capital Management Holdings Limited is wholly owned by Cheng Yin Pan. The registered address of M13 Capital Management Holdings Limited is Portcullis Chambers, 4th Floor, Ellen Skelton Building, 3076 Sir Francis Drake Highway, Road Town, Tortola, VG1110, British Virgin Islands.
C.Board Practices
Board of Directors
Our board of directors consists of four directors as of the date of this annual report. Of these four directors, two are independent. The Articles provide that the minimum number of directors shall be two and the exact number of directors shall be determined from time to time by our board of directors. A director is not required to hold any shares in us by way of qualification. A director may vote in respect of any contract or proposed contract or arrangement in which such director may be interested provided that (a) the nature of his/her interest is declared at a meeting of the directors, either specifically or by way of a general notice, and such director’s vote may be counted in the quorum at any meeting of directors at which any such contract or proposed contract or arrangement is considered, and (b) if such contract or arrangement is a transaction with a related party, such transaction has been approved by the audit committee. The directors may exercise all our powers to borrow money, mortgage its undertaking, property and uncalled capital, and issue debentures or other securities whenever money is borrowed or as security for any obligation of us or of any third party. No non-employee director has a service contract with us that provides for benefits upon termination of service.
Board Committees
Our board of directors has an audit committee, a compensation committee and a nominating and corporate governance committee. Each committee’s members and functions are described below.
Audit Committee
The audit committee consists of Cheng Yin Pan (Ben) and Dr. Darshan Shah. Mr. Cheng is the chairperson of the audit committee. Mr. Cheng satisfies the criteria of an audit committee financial expert as set forth under the applicable rules of the SEC. Our board of directors has determined that each of Mr. Cheng and Dr. Shah satisfies the requirements for an “independent director” within the meaning of the Nasdaq listing rules and the criteria for independence set forth in Rule 10A-3 of the Exchange Act.
The audit committee oversees our accounting and financial reporting processes. The audit committee is responsible for, among other things:
•appointing the independent auditors and pre-approving all auditing and non-auditing services permitted to be performed by the independent auditors;
•reviewing with the independent auditors any audit problems or difficulties and management’s response;
•discussing the annual audited financial statements with management and the independent auditors;
77
•reviewing the adequacy and effectiveness of our accounting and internal control policies and procedures and any steps taken to monitor and control major financial risk exposures;
•reviewing and approving all proposed related party transactions;
•meeting separately and periodically with management and the independent auditors;
•monitoring compliance with our code of business conduct and ethics, including reviewing the adequacy and effectiveness of our procedures to ensure proper compliance.
Compensation Committee
The compensation committee consists of Cheng Yin Pan (Ben) and Dr. Darshan Shah. Mr. Cheng is the chairperson of the compensation committee. Our board of directors has determined that each of Mr. Cheng and Dr. Shah satisfies the requirements for an “independent director” within the meaning of the Nasdaq listing rules.
The compensation committee is responsible for, among other things:
•reviewing and approving, or recommending to the board for its approval, the compensation for our chief executive officer and other executive officers;
•reviewing and recommending to the board for determination with respect to the compensation of our non-employee directors;
•reviewing periodically and approving any incentive compensation or equity plans, programs or similar arrangements; and
•the selection of compensation consultant, legal counsel or other adviser only after taking into consideration all factors relevant to that person’s independence from management.
Nominating and Corporate Governance Committee
The nominating and corporate governance committee consists of Cheng Yin Pan (Ben), Dr. Darshan Shah and Danny Yeung. Mr. Yeung is the chairperson of the nominating and corporate governance committee. Our board of directors has determined that each of Mr. Cheng and Dr. Shah satisfies the requirements for an “independent director” within the meaning of the Nasdaq listing rules.
The nominating and corporate governance committee is responsible for, among other things:
•selecting and recommending to the board of directors nominees for election by the shareholders or appointment by the board of directors;
•reviewing annually with the board of directors the current composition of the board of directors with regard to characteristics such as independence, knowledge, skills, experience and diversity;
•making recommendations on the frequency and structure of our board of directors meetings and monitoring the functioning of the committees of our board of directors; and
•advising our board of directors periodically with regard to significant developments in the law and practice of corporate governance as well as our compliance with applicable laws and regulations, and making recommendations to our board of directors on all matters of corporate governance and on any remedial action to be taken.
Duties of Directors
Under the laws of the Cayman Islands, directors have a fiduciary duty to act honestly in good faith with a view to the company’s best interests. Our directors also have a duty to exercise the care, diligence and skill that a reasonably prudent person would exercise in comparable circumstances. We have the right to seek damages if a duty owed by our directors is breached. In limited exceptional circumstances, a shareholder may have the right to seek damages in the company’s name if a duty owed by the directors is breached.
78
Appointment and Removal of Directors
Our amended and restated memorandum and articles of association provide that all directors may be appointed by ordinary resolution and removed by ordinary resolution. The Articles also provide that the directors may, so long as a quorum of directors remains in office, appoint any person to be a director so as to fill a casual vacancy or as an addition to the existing board of director. Our directors do not serve for a fixed term and there is no requirement for them to retire by rotation nor to make themselves eligible for re-election.
The office of a director shall be vacated if (a) such director resigns their office by notice in writing signed by such director and left at our registered office; (b) such director becomes bankrupt or makes any arrangement or composition with such director’s creditors generally; (c) such director dies or is found to be or becomes of unsound mind; (d) such director ceases to be a director by virtue of, or becomes prohibited from being a director by reason of, an order made under any provisions of any law or enactment; (e) such director is removed from office by notice addressed to such director at their last known address and signed by all of the co-directors (not being less than two in number); or (f) such director is removed from office by ordinary resolution.
Terms of Directors
A director shall hold office until such time as he or she resigns his office by notice in writing to us, is removed from office by ordinary resolution or is otherwise disqualified from acting as a director or removed in accordance with our amended and restated memorandum and articles of association.
D.Employees
As of December 31, 2025, we had 98 full-time employees and 30 service contractors across eight countries, including Hong Kong, Malaysia and the United States. Our employees and service contractors are primarily located in Hong Kong and Malaysia. The following table sets forth the number of our employees categorized by function and geographic region as of December 31, 2025:
| Function: | As of December 31, 2025 | ||||
| General and administrative | 52 | ||||
| Operations | 16 | ||||
| Research and development | 9 | ||||
| Sales and marketing | 21 | ||||
| Total | 98 | ||||
| Geographic Region: | As of December 31, 2025 | ||||
| Hong Kong | 55 | ||||
Malaysia | 38 | ||||
United States | 4 | ||||
United Kingdom | 1 | ||||
| Total | 98 | ||||
We believe we generally have good relationships with our employees and service contractors. Our human capital resources objectives include, as applicable, identifying, recruiting, retaining, incentivizing and integrating our existing and new employees, advisors and consultants. The principal purposes of our equity and cash incentive plans are to attract, retain and reward personnel through the granting of stock-based and cash-based compensation awards, in order to increase
79
stockholder value and the success of our company by motivating such individuals to perform to the best of their abilities and achieve our objectives.
E.Share Ownership
The following table sets forth information regarding the beneficial ownership of our ordinary shares as of April 29, 2026 by:
•each person known by us to be the beneficial owner of more than 5% of the outstanding ordinary shares;
•each of our directors and executive officers; and
•all our directors and executive officers as a group.
The number of ordinary shares beneficially owned by each entity, person, executive officer or director is determined in accordance with the rules of the SEC, and the information is not necessarily indicative of beneficial ownership for any other purpose. Under such rules, beneficial ownership includes any ordinary shares over which the individual has sole or shared voting power or investment power as well as any such ordinary shares that the individual has the right to acquire within 60 days of April 29, 2026 through the exercise of any option or other right. Ordinary shares that a person has the right to acquire within 60 days of April 29, 2026 are deemed outstanding for purposes of computing the percentage ownership of the person holding such rights, but are not deemed outstanding for purposes of computing the percentage ownership of any other person, except with respect to the percentage ownership of all executive officers and directors as a group. Except as otherwise indicated, we believe that the persons named in the table have sole voting and investment power with respect to all ordinary shares held by that person based on information provided to us by such person. Each holder of Class A Ordinary Shares is entitled to one vote per share and each holder of Class B Ordinary Shares is entitled to twenty (20) votes per share.
The percentage of our ordinary shares beneficially owned is computed on the basis of 15,562,491 Class A Ordinary Shares and 1,580,972 Class B Ordinary Shares issued and outstanding as of April 29, 2026.
| Ordinary Shares Beneficially Owned | |||||||||||||||||||||||||||||
| Class A Ordinary Shares | Class B Ordinary Shares | Total Ordinary Shares | % of Total Ordinary Shares | % of Voting Power(2) | |||||||||||||||||||||||||
Directors and Executive Officers(1) | |||||||||||||||||||||||||||||
Danny Sheng Wu Yeung(3) | 138,694 | 1,611,050 | 1,749,744 | 10.19 | % | 67.72 | % | ||||||||||||||||||||||
Cheng Yin Pan (Ben)(4) | 75,021 | — | 75,021 | 0.44 | % | 0.16 | % | ||||||||||||||||||||||
David Eric Vanderveen(5) | 39,953 | — | 39,953 | 0.23 | % | 0.08 | % | ||||||||||||||||||||||
| Dr. Darshan Shah | 4,506 | — | 4,506 | 0.03 | % | 0.01 | % | ||||||||||||||||||||||
Lo Hoi Chun (Stephen)(6) | 524,311 | — | 524,311 | 3.05 | % | 1.11 | % | ||||||||||||||||||||||
| All Directors and Executive Officers as a Group | 782,485 | 1,611,050 | 2,393,535 | 13.91 | % | 69.02 | % | ||||||||||||||||||||||
| Principal Shareholders | |||||||||||||||||||||||||||||
Lo Yuk Ming Dennis(7) | 962,963 | — | 962,963 | 5.62 | % | 2.04 | % | ||||||||||||||||||||||
Danny Sheng Wu Yeung(3) | 138,694 | 1,611,050 | 1,749,744 | 10.19 | % | 67.72 | % | ||||||||||||||||||||||
_______________
(1)The business address for the directors and executive officers of the Company is Unit 703-706, K11 Atelier King’s Road, 728 King’s Road, Quarry Bay, Hong Kong.
(2)For each person or group included in this column, percentage of total voting power represents voting power based on both Class A Ordinary Shares and Class B Ordinary Shares held by such person or group with respect to all outstanding Ordinary Shares as a single class. Each holder of Class A Ordinary Shares is entitled to one vote per share. Each holder of Class B Ordinary Shares is entitled to twenty (20) votes per share. Class B Ordinary Shares are convertible at any time by the holder into Class A Ordinary Shares on a one-for-one basis, while Class A Ordinary Shares are not convertible into Class B Ordinary Shares under any circumstances.
(3)Represents 647,592 Class B Ordinary Shares held by Da Yeung Limited, a British Virgin Islands company, 138,694 Class A Ordinary Shares and 933,380 Class B Ordinary Shares held by Danny Sheng Wu Yeung, and 30,078 Class B Ordinary Shares that Danny Sheng Wu Yeung has the contingent right to acquire within 60 days by settlement of his
80
vested Restricted Stock Units granted under the Company’s 2022 Share Incentive Plan. Settlement and delivery of such 30,078 Class B Ordinary Shares occurs at Danny Sheng Wu Yeung’s election. Da Yeung Limited is wholly owned by Danny Sheng Wu Yeung. The registered address of Da Yeung Limited is Coastal Building, Wickham’s Cay II, P. 0. Box 2221, Road Town, Tortola, VG 1110, British Virgin Islands.
(4)Represents 75,021 Class A Ordinary Shares held by M13 Capital Management Holdings Limited, a British Virgin Islands company. M13 Capital Management Holdings Limited is wholly owned by Cheng Yin Pan. The registered address of M13 Capital Management Holdings Limited is Portcullis Chambers, 4th Floor, Ellen Skelton Building, 3076 Sir Francis Drake Highway, Road Town, Tortola, VG1110, British Virgin Islands.
(5)Represents 38,305 Class A Ordinary Shares held by David Eric Vanderveen and 1,648 Class A Ordinary Shares held by his spouse.
(6)Represents 487,462 Class A Ordinary Shares held by Lo Hoi Chun, and 36,849 Class A Ordinary Shares that Lo Hoi Chun has the contingent right to acquire within 60 days by settlement of his vested Restricted Stock Units granted under the Company’s 2022 Share Incentive Plan. Settlement and delivery of such 36,849 Class A Ordinary Shares occurs at Lo Hoi Chun’s election.
(7)Based on the most recently available Schedule 13D filed by Lo Yuk Ming Dennis on August 7, 2023.
F.Disclosure of A Registrant’s Action to Recover Erroneously Awarded Compensation
Not applicable.
ITEM 7. MAJOR SHAREHOLDERS AND RELATED PARTY TRANSACTIONS
A.Major Shareholders
Please refer to “Item 6. Directors, Senior Management and Employees — E. Share Ownership.”
B.Related Party Transactions
Business Combination
Related party transactions entered into in connection with our business combination in 2022, including the issuance of Class B founder shares and sponsor-related arrangements, are described in our Form 20-F for the year ended December 31, 2024, and are incorporated herein by reference. There have been no material changes to those arrangements as of the date of this Annual Report.
Employment Agreements and Indemnification Agreements
See “Item 6. Directors, Senior Management and Employees — B. Compensation.”
Share Incentive Plans
See “Item 6. Directors, Senior Management and Employees — B. Compensation — Share Incentive Plans.”
Other Related Party Transactions
For details on certain related party transactions that the Company has entered into in the normal course of the Company’s business, see Note 36 to our audited consolidated financial statements included elsewhere in this annual report.
C.Interests of Experts and Counsel
Not applicable.
ITEM 8. FINANCIAL INFORMATION
A.Consolidated Statements and Other Financial Information
We have appended consolidated financial statements filed as part of this annual report.
81
Legal Proceedings
From time to time, we may be subject to litigation and/or other claims incidental to our ordinary course of business. There are currently no claims or actions pending against us, that, in the view of our management, are likely to have a material adverse effect on our business.
Litigation or any other legal or administrative proceeding, regardless of the outcome, is likely to result in substantial costs and diversion of our resources, including our management’s time and attention. For potential impact of legal or administrative proceedings on us, see “Item 3. Key Information — D. Risk Factors — Risks Relating to Intellectual Property and Legal Proceedings — We may be subject to legal proceedings and litigation, which are costly to defend, and adverse publicity about any investigation, litigation, regulatory or legal action against us or our senior management could harm our reputation and business.”
Dividend Policy
We have not declared or paid any cash dividend on our Class A Ordinary Shares as of the date of this annual report. We currently intend to retain any future earnings and do not expect to pay any dividends in the foreseeable future. Any further determination to pay dividends on our ordinary shares would be at the discretion of our board of directors, subject to applicable laws, and would depend on our financial condition, results of operations, capital requirements, general business conditions, and other factors that our board of directors may deem relevant.
B.Significant Changes
Except as disclosed elsewhere in this annual report, we have not experienced any significant changes since the date of our audited consolidated financial statements included in this annual report.
ITEM 9. THE OFFER AND LISTING
A.Offer and Listing Details
See “— C. Markets.”
B.Plan of Distribution
Not applicable.
C.Markets
Our Class A Ordinary Shares and Warrants are listed on Nasdaq under the symbols “PRE” and “PRENW,” respectively.
D.Selling Shareholders
Not applicable.
E.Dilution
Not applicable.
F.Expenses of the Issue
Not applicable.
ITEM 10. ADDITIONAL INFORMATION
A.Share Capital
Not applicable.
82
B.Memorandum and Articles of Association
The following is a summary of certain material provisions of our Amended and Restated Memorandum and Articles of Association, as amended by special resolution on August 1, 2025 (as so amended, the “Amended Articles”), and the Companies Act (As Revised) of the Cayman Islands (the “Companies Act”), insofar as they relate to the material terms of the Company’s ordinary shares. This summary is qualified in its entirety by reference to the complete text of the Amended Articles, which is filed as an exhibit to this annual report on Form 20-F. We encourage you to read the Amended Articles in their entirety for a complete description of the rights and obligations of holders of our ordinary shares.
Registration
The Company was incorporated in the Cayman Islands as an exempted company with limited liability on May 28, 2021. The Company is registered with the Registrar of Companies of the Cayman Islands under file number 378682. The Company’s registered office is at the offices of Mourant Governance Services (Cayman) Limited, 94 Solaris Avenue, Camana Bay, P.O. Box 1348, Grand Cayman KY1-1108, Cayman Islands, or at such other place within the Cayman Islands as the directors may from time to time decide.
Objects and Purposes
The objects of the Company are unrestricted and the Company has full power and authority to carry out any object not prohibited by the Companies Act or any other law of the Cayman Islands. The Company’s Amended Articles do not impose any limitation on the objects or purposes of the Company.
Description of Share Capital
As originally adopted, the Company’s authorized share capital was US$50,000 divided into 500,000,000 shares of US$0.0001 par value each. Following the 1-for-15 reverse stock split authorized by an ordinary resolution of the shareholders on October 20, 2023 and effected on November 15, 2023, the par value of each ordinary share was consolidated to US$0.0015. Subsequently, on August 1, 2025, the Company’s shareholders approved a special resolution to increase the authorized share capital from US$50,000 divided into 33,333,334 shares to US$320,000 divided into 213,333,334 shares of US$0.0015 par value each, of which (i) 186,666,667 are designated as Class A Ordinary Shares, (ii) 15,333,333 are designated as convertible Class B Ordinary Shares and (iii) 11,333,334 are designated as shares of such class or classes (however designated) as the board of directors may determine in accordance with Article 10 of the Amended Articles. All ordinary shares issued and outstanding are fully paid and non-assessable.
Holders of Class A Ordinary Shares and Class B Ordinary Shares generally have the same rights and powers except for voting and conversion rights as described below.
The Company maintains a register of its shareholders and a shareholder will only be entitled to a share certificate if the board of directors determines that share certificates be issued.
Danny Yeung, our chief executive officer, controls the voting power of all of the outstanding Class B Ordinary Shares. Although Mr. Yeung controls the voting power of all of the outstanding Class B Ordinary Shares, his control over those shares is not permanent and is subject to reduction or elimination at any time or after certain periods as a result of a variety of factors, as further described below under “—Optional and Mandatory Conversion.”
Dividends
The holders of ordinary shares are entitled to such dividends as the board of directors may in its discretion lawfully declare from time to time, or as shareholders may declare by ordinary resolution (provided that no dividend shall exceed the amount recommended by the board of directors). Dividends may be declared and paid out of the funds of the Company lawfully available therefor. The directors may also declare and pay dividends out of the share premium account or any other fund or account which can be authorized for this purpose in accordance with the Companies Act.
Class A Ordinary Shares and Class B Ordinary Shares rank equally as to dividends and other distributions. Dividends may be paid either in cash or in specie, provided that no dividend can be made in specie on any Class A Ordinary Shares unless a dividend in specie in equal proportion is made on Class B Ordinary Shares.
Voting Rights
83
Holders of ordinary shares have the right to receive notice of, attend, speak and vote at general meetings of the shareholders. In respect of all matters upon which holders of ordinary shares are entitled to vote, each Class A Ordinary Share is entitled to one (1) vote and each Class B Ordinary Share is entitled to twenty (20) votes. At any meeting of shareholders a resolution put to the vote of the meeting shall be decided by way of a poll and not by way of a show of hands. A poll shall be taken in such manner and at such place as the chairperson of the meeting may direct (including the use of a ballot or voting papers, or tickets) and the result of a poll shall be deemed to be the resolution of the meeting.
Class A Ordinary Shares and Class B Ordinary Shares vote together on all matters, except that the Company will not, without the approval of holders of a majority of the voting power of the Class B Ordinary Shares, voting exclusively and as a separate class:
•increase the number of authorized Class B Ordinary Shares;
•issue any Class B Ordinary Shares or securities convertible into or exchangeable for Class B Ordinary Shares, other than to any Key Executive or his or her affiliates, or on a pro rata basis to all holders of Class B Ordinary Shares permitted to hold such shares under the Amended Articles;
•create, authorize, issue, or reclassify into, any preference shares in the capital of the Company or any shares in the capital of the Company that carry more than one (1) vote per share;
•reclassify any Class B Ordinary Shares into any other class of shares or consolidate or combine any Class B Ordinary Shares without proportionately increasing the number of votes per Class B Ordinary Share; or
•amend, restate, waive, adopt any provision inconsistent with or otherwise vary or alter any provision of the Amended Articles relating to the voting, conversion or other rights, powers, preferences, privileges or restrictions of the Class B Ordinary Shares.
An ordinary resolution to be passed by the shareholders will require a simple majority of votes cast at a meeting of shareholders, while a special resolution will require not less than two-thirds of votes cast at a meeting of shareholders.
Liquidation Rights
The Class A Ordinary Shares and Class B Ordinary Shares rank equally upon the occurrence of any liquidation, dissolution or winding up of the Company, in the event of which the Company’s assets will be distributed to, or the losses will be borne by, shareholders in proportion to the par value of the shares held by them. The liquidator may, with the sanction of a special resolution of the shareholders, divide amongst the shareholders in specie or in kind the whole or any part of the assets of the Company, and may for such purpose set such value as the liquidator deems fair upon any property to be divided and may determine how such division shall be carried out as between the shareholders or different classes of shareholders.
Sinking Fund Provisions
There are no sinking fund provisions applicable to the Company’s ordinary shares.
Redemption of Ordinary Shares
Subject to the provisions of the Companies Act, the Company may issue shares that are to be redeemed or are liable to be redeemed at the option of the shareholder or the Company. The redemption of such shares will be effected in such manner and upon such other terms as the Company may, by either resolution of the board of directors or special resolution of shareholders, determine before the issue of the shares.
Liability to Further Capital Calls
The board of directors may from time to time make calls upon shareholders for any amounts unpaid on their ordinary shares. The ordinary shares that have been called upon and remain unpaid are, after a notice period, subject to forfeiture. All issued and outstanding ordinary shares are fully paid. Accordingly, no additional capital contributions may be required of our existing shareholders.
Provision Discriminating Against Shareholders Owning a Substantial Number of Shares
There is no provision in the Amended Articles that discriminates against any existing or prospective holder of ordinary shares as a result of such shareholder owning a substantial number of shares.
84
Optional and Mandatory Conversion
Each Class B Ordinary Share is convertible into one (1) Class A Ordinary Share (as adjusted for share splits, share combinations and similar transactions occurring after the Acquisition Effective Time) at any time at the option of the holder thereof. Class A Ordinary Shares are not convertible into Class B Ordinary Shares under any circumstances.
Any number of Class B Ordinary Shares held by a holder thereof will automatically and immediately be converted into an equal number of Class A Ordinary Shares (as adjusted for share splits, share combinations and similar transactions occurring after the Acquisition Effective Time) upon the occurrence of any of the following:
•Any direct or indirect sale, transfer, assignment, or disposition of such number of Class B Ordinary Shares by the holder thereof or the direct or indirect transfer or assignment of the voting power attached to such number of Class B Ordinary Shares through voting proxy or otherwise to any person that is not a Permitted Transferee of such holder;
•The direct or indirect sale, transfer, assignment, or disposition of a majority of the issued and outstanding voting securities of, or the direct or indirect transfer or assignment of the voting power attached to such voting securities through voting proxy or otherwise, or the direct or indirect sale, transfer, assignment, or disposition of all or substantially all of the assets of, a holder of Class B Ordinary Shares that is an entity to any person that is not a Permitted Transferee of such holder; or
•A person becoming a holder of Class B Ordinary Shares by will or intestacy.
All Class B Ordinary Shares issued and outstanding will be automatically and immediately converted into an equal number of Class A Ordinary Shares upon the occurrence of any of the following:
•On Danny Yeung’s death or Incapacity;
•On the date on which Danny Yeung is terminated for cause (as defined in the employment agreement with Danny Yeung and in the event of a dispute regarding whether there was cause, cause will be deemed not to exist unless and until an affirmative ruling regarding such cause has been made by a court or arbitral panel of competent jurisdiction, and such ruling has become final and non-appealable); or
•On the first date that both of the following conditions are satisfied: (I) Danny Yeung and his Affiliates and Permitted Transferees together own less than thirty three percent (33%) of the number of Class B Ordinary Shares (which for these purposes shall be deemed to include all Class B Ordinary Shares issuable upon exercise of all outstanding restricted share units to acquire Class B Ordinary Shares that are held by Danny Yeung immediately following the Acquisition Effective Time) that Danny Yeung and his Affiliates and Permitted Transferees owned immediately following the Acquisition Effective Time, as adjusted for share splits, share combinations and similar transactions occurring after the Acquisition Effective Time; and (II) Danny Yeung ceases to be a Director or officer of the Company.
No Class B Ordinary Shares shall be issued by the Company after conversion of all Class B Ordinary Shares into Class A Ordinary Shares.
Transfer of Ordinary Shares
Subject to applicable laws, including securities laws, and the restrictions contained in the Amended Articles, any shareholders may transfer all or any of their Class A Ordinary Shares by an instrument of transfer in the usual or common form, in a form prescribed by Nasdaq or any other form approved by the board of directors of the Company.
Class B Ordinary Shares may be transferred only to a Permitted Transferee of the holder and any Class B Ordinary Shares transferred otherwise will be converted into Class A Ordinary Shares as described above. See “— Optional and Mandatory Conversion.”
A “Permitted Transferee” with respect to the Class B Ordinary Shareholders, means any or all of the following:
(a)Danny Yeung and his Permitted Entities and Permitted Transferees of each of them (each a “Key Executive”);
(b)any Key Executive’s Permitted Entities;
(c)the transferee or other recipient in any transfer of any Class B Ordinary Shares by any Class B Ordinary Shareholder: (i) to (A) his or her Family Members; (B) any other relative or individual approved by the board of directors of the Company; or (C) any trust or estate planning entity (including partnerships, limited companies, and limited liability companies), that is primarily for the benefit of, or the ownership interests of which are Controlled by, such Class B Ordinary Shareholder, his or her Family Members, and/or other trusts or
85
estate planning entities described in this paragraph (c), or any entity Controlled by such Key Executive or a trust or estate planning entity; or (ii) occurring by operation of law, including in connection with divorce proceedings;
(d)any charitable organization, foundation, or similar entity;
(e)the Company or any of its subsidiaries; or
(f)in connection with a transfer as a result of, or in connection with, the death or incapacity of a Key Executive: any Key Executive’s Family Members, another Class B Ordinary Shareholder, or a designee approved by majority of all directors of the Company, provided that in case of any transfer of Class B Ordinary Shares pursuant to clauses (b) through (e) above to a person who at any later time ceases to be a Permitted Transferee under the relevant clause, the Company shall be entitled to refuse registration of any subsequent transfer of such Class B Ordinary Shares except back to the transferor of such Class B Ordinary Shares pursuant to clauses (b) through (e) (or to a Key Executive or his or her Permitted Transferees) and in the absence of such transfer back to the transferor (or to a Key Executive or his or her Permitted Transferees), the applicable Class B Ordinary Shares shall be subject to mandatory conversion as set out above.
A “Permitted Entity” with respect to any Key Executive means: (a) any person in respect of which such Key Executive has, directly or indirectly: (i) control with respect to the voting of all the Class B Ordinary Shares held by or to be transferred to such person; (ii) the ability to direct or cause the direction of the management and policies of such person or any other person having the authority referred to in the preceding clause (a)(i) (whether by contract, as executor, trustee, trust protector or otherwise); or (iii) the operational or practical control of such person, including through the right to appoint, designate, remove or replace the person having the authority referred to in the preceding clauses (a)(i) or (ii); (b) any trust the beneficiaries of which consist primarily of a Key Executive, his or her Family Members, and/or any persons Controlled directly or indirectly by such a trust; and (c) any person Controlled by a trust described in the immediately preceding clause (b).
“Family Member” means the following individuals: the applicable individual, the spouse of the applicable individual (including former spouses), the parents of the applicable individual, the lineal descendants of the applicable individual, the siblings of the applicable individual, and the lineal descendants of a sibling of the applicable individual. For purposes of the preceding sentence, the descendants of any individual shall include adopted individuals and their issue but only if the adopted individual was adopted prior to attaining age 18.
“Controlled” means directly or indirectly: (i) the ownership or control of a majority of the outstanding voting securities of such person; (ii) the right to control the exercise of a majority of the votes at a meeting of the board of directors (or equivalent governing body) of such person; or (iii) the ability to direct or cause the direction of the management and policies of such person (whether by contract, through other legally enforceable rights or howsoever arising).
The board of directors may decline to register any transfer of any share in the event that any of the following is known by the directors not to be both applicable and true with respect to such transfer:
•the instrument of transfer is lodged with the Company, or the Company’s designated transfer agent or share registrar, accompanied by the certificate for the shares to which it relates (if any) and such other evidence as the board of directors may reasonably require to show the right of the transferor to make the transfer;
•the instrument of transfer is in respect of only one class of shares;
•the instrument of transfer is properly stamped, if required;
•the transferred shares are fully paid up and free of any lien in favor of the Company (it being understood and agreed that all other liens, e.g., pursuant to a bona fide loan or indebtedness transaction, shall be permitted); or
•a fee of such maximum sum as Nasdaq may determine to be payable, or such lesser sum as the board of directors may from time to time require, is paid to the Company in respect thereof.
If the board of directors refuses to register a transfer they shall, within two months after the date on which the instrument of transfer was lodged, send to each of the transferor and the transferee notice of such refusal stating the facts which are considered to justify the refusal to register the transfer.
Variation of Rights of Shares
Subject to certain Amended Articles provisions governing the Class B Ordinary Shares, if at any time the share capital of the Company is divided into different classes of shares, the rights attached to any class (unless otherwise
86
provided by the terms of issue of the shares of that class) may be varied without the consent of the holders of the issued shares of that class where such variation is considered by the directors not to have a material adverse effect upon such rights. Otherwise, any such variation will be made only with the consent in writing of the holders of not less than two-thirds of the issued shares of that class, or with the approval of a resolution passed by a majority of not less than two-thirds of the votes cast at a separate meeting of the holders of the shares of that class.
General Meetings of Shareholders
The Company will hold an annual general meeting at such time and place as the board of directors will determine. The board of directors may call extraordinary general meetings whenever they think fit, and must convene an extraordinary general meeting upon the requisition of (a) shareholders holding at least one third of the votes that may be cast at such meeting, or (b) the holders of Class B Ordinary Shares entitled to cast a majority of the votes that all Class B Ordinary Shares are entitled to cast. At least seven (7) calendar days’ notice in writing shall be given for any general meeting.
One or more shareholders holding not less than one-third of the total issued share capital of the Company present in person or by proxy and entitled to vote will be a quorum for all purposes, provided that, from and after the Acquisition Effective Time where Class B Ordinary Shares are in issue, the presence in person or by proxy of holders of a majority of Class B Ordinary Shares will be required in any event.
Any action required or permitted to be taken at a meeting of shareholders may be effected by a written resolution passed in accordance with the Amended Articles.
Limitations on the Rights to Own Shares
There are no limitations on the rights to own shares in the Company, including rights of non-resident or foreign shareholders, imposed by Cayman Islands law or by the Amended Articles.
Change of Control
There are no provisions in the Amended Articles that would have the effect of delaying, deferring or preventing a change of control of the Company, other than the dual-class share structure described above and the following provisions:
•The board of directors has the authority, without further action of the shareholders, to issue shares of such class or classes as the board may determine, including preference shares with such rights, preferences and restrictions as the board determines, which could be used to dilute the holdings of a potential acquirer;
•The dual-class ordinary share structure, which provides Danny Yeung with significant influence over matters requiring shareholder approval, including the election of directors and significant corporate transactions;
•Danny Yeung serves as the permanent Chairperson of the board of directors so long as he remains a Director; and
•There is no cumulative voting with respect to the election of directors, which limits the ability of minority shareholders to elect director candidates.
However, under Cayman Islands law, our directors may only exercise the rights and powers granted to them under the Amended Articles for a proper purpose and for what they believe in good faith to be in the best interests of the Company.
Disclosure of Share Ownership
There are no provisions in the Amended Articles governing the ownership threshold above which shareholder ownership must be disclosed. The Company is subject to the disclosure requirements of the United States securities laws, including the Securities Exchange Act of 1934 (as amended), which requires disclosure of beneficial ownership of more than 5% of the Company’s voting shares, as well as beneficial ownership reporting by the Company’s directors and officers.
Changes in Capital
The Company may from time to time by ordinary resolution, subject to the rights of holders of Class B Ordinary Shares:
87
•increase its share capital by such sum, to be divided into shares of such amount, as the resolution will prescribe;
•consolidate and divide all or any of its share capital into shares of a larger amount than existing shares;
•sub-divide its existing shares or any of them into shares of a smaller amount; provided that in the subdivision the proportion between the amount paid and the amount, if any, unpaid on each reduced share will be the same as it was in case of the share from which the reduced share is derived; or
•cancel any shares that at the date of the passing of the resolution have not been taken or agreed to be taken by any person and diminish the amount of its share capital by the amount of the shares so cancelled.
Subject to the rights of Class B Ordinary Shares, the Company may by special resolution reduce its share capital or any capital redemption reserve fund in any manner permitted by law.
Directors
Power to Vote on Matters in Which a Director Is Materially Interested
A director may vote with respect to any contract, proposed contract, or arrangement in which he or she is interested. Subject to the Amended Articles, if a director has an interest in a transaction, he or she must declare the nature and extent of that interest. However, a director shall not be deemed to be interested in a transaction if he or she is interested solely by reason of holding shares in, or being a director or officer of, a company in which the director is interested, provided that the director shall not vote on any resolution of the board of directors relating to the granting of any security or guarantee in connection with a debt or obligation of the Company for the benefit of such director personally.
Remuneration of Directors
The directors shall receive such remuneration as the board of directors may from time to time determine. Each director is entitled to be repaid or prepaid all travelling, hotel and incidental expenses reasonably incurred or expected to be incurred by him or her in attending meetings of the board of directors or committees of the board of directors or shareholder meetings or otherwise in connection with the discharge of his or her duties as a director.
Appointment and Removal of Directors
Unless otherwise determined by the Company by ordinary resolution, the number of directors shall not be less than two (2) and the exact number of directors shall be determined from time to time by the board of directors. Directors may be appointed by shareholders in a general meeting by ordinary resolution, or by the board of directors. The Chairperson of the board of directors shall be Danny Yeung, so long as Danny Yeung is a Director. In the event that Danny Yeung is not a Director, the board of directors shall elect and appoint a Chairperson by the affirmative vote of a simple majority of the directors then in office. An appointment of a director may be on terms that the director shall automatically retire from office (unless the director has sooner vacated office) at the next or a subsequent annual general meeting or upon any specified event or after any specified period. The office of a director may be vacated if the director resigns, becomes bankrupt or is found to be or becomes of unsound mind.
Borrowing Powers
The board of directors may exercise all of the powers of the Company to borrow money and to mortgage or charge all or any part of the undertaking, property and assets (present and future) and uncalled capital of the Company, and to issue debentures, bonds and other securities of the Company, whether outright or as collateral security for any debt, liability or obligation of the Company or any third party.
Retirement and Age Limit
There is no requirement in the Amended Articles for retirement of directors upon reaching any age limit.
Number of Shares Required to Qualify as a Director
There is no requirement for a director to hold shares in the Company in order to qualify to serve as a director.
88
Calls on Ordinary Shares and Forfeiture of Ordinary Shares
The board of directors may from time to time make calls upon shareholders for any amounts unpaid on their ordinary shares. The ordinary shares that have been called upon and remain unpaid are, after a notice period, subject to forfeiture.
Inspection of Books and Records
The board of directors will determine whether, to what extent, at what times and places and under what conditions or articles the accounts and books of the Company will be open to the inspection by shareholders, and no shareholder (not being a director) will otherwise have any right of inspecting any account or book or document of the Company except as required by the Companies Act or authorized by ordinary resolution of shareholders. Holders of our ordinary shares will have no general rights under Cayman Islands law to inspect or obtain copies of our list of shareholders or our corporate records (except for the memorandum and articles of association, any special resolutions and the register of mortgages and charges).
Exempted Company
The Company is an exempted company with limited liability incorporated under the laws of the Cayman Islands. The Companies Act distinguishes between ordinary resident companies and exempted companies. Any company that is registered in the Cayman Islands but conducts business mainly outside of the Cayman Islands may apply to be registered as an exempted company. The requirements for an exempted company are essentially the same as for an ordinary resident company except for the exemptions and privileges listed below:
•an exempted company (other than an exempted company holding a license to carry on business in the Cayman Islands) does not have to file an annual return of its shareholders with the Registrar of Companies of the Cayman Islands;
•an exempted company’s register of members is not open to inspection;
•an exempted company does not have to hold an annual general meeting;
•an exempted company may issue shares with no par value;
•an exempted company may obtain an undertaking against the imposition of any future taxation;
•an exempted company may register by way of continuation in another jurisdiction and be deregistered in the Cayman Islands;
•an exempted company may register as a limited duration company; and
•an exempted company may register as a segregated portfolio company.
“Limited liability” means that the liability of each shareholder is limited to the amount unpaid by the shareholder on the shares of the company (except in exceptional circumstances, such as involving fraud, the establishment of an agency relationship or an illegal or improper purpose or other circumstances in which a court may be prepared to pierce or lift the corporate veil).
Indemnification of Directors and Officers
Cayman Islands law does not limit the extent to which a company’s memorandum and articles of association may provide for indemnification of officers and directors, except to the extent any such provision may be held by the Cayman Islands courts to be contrary to public policy, such as to provide indemnification against willful default, willful neglect, civil fraud or the consequences of committing a crime.
The Amended Articles provide for indemnification of our officers and directors to the maximum extent permitted by law, including for any liability incurred in their capacities as such, except through their own actual fraud or willful default. We have entered into indemnification agreements with each of our directors and executive officers. In addition, we maintain standard policies of insurance under which coverage is provided to our directors and officers against loss arising from claims made by reason of breach of duty or other wrongful act, and to us with respect to payments which may be made by us to such directors and officers pursuant to the indemnification provision or otherwise as a matter of law.
Differences in Corporate Law
89
The Companies Act is derived, to a large extent, from the older Companies Acts of England but does not follow recent English statutory enactments, and accordingly there are significant differences between the Companies Act and the current Companies Act of England. In addition, the Companies Act differs from laws applicable to U.S. corporations and their shareholders. Set forth below is a summary of certain significant differences between the provisions of the Companies Act applicable to the Company and the laws applicable to companies incorporated in the State of Delaware, which is the jurisdiction where many publicly listed companies in the United States are incorporated.
Mergers and Similar Arrangements
The Companies Act permits mergers and consolidations between Cayman Islands companies and between Cayman Islands companies and non-Cayman Islands companies. For these purposes, (a) “merger” means the merging of two or more constituent companies and the vesting of their undertaking, property and liabilities in one of such companies as the surviving company, and (b) a “consolidation” means the combination of two or more constituent companies into a consolidated company and the vesting of the undertaking, property and liabilities of such companies to the consolidated company. In order to effect such a merger or consolidation, the directors of each constituent company must approve a written plan of merger or consolidation, which must then be authorized by (a) a special resolution of the shareholders of each constituent company, and (b) such other authorization, if any, as may be specified in such constituent company’s articles of association.
The Companies Act also permits a court-sanctioned plan of arrangement to be approved by a majority in number of each class of shareholders and creditors with whom the arrangement is to be made and who at a meeting convened by such court or pursuant to the Companies Act represent seventy-five percent (75%) or more in value of each such class of shareholders or creditors, as the case may be, that are present and voting either in person or by proxy at such meeting.
The Companies Act also contains a statutory power of compulsory acquisition which may facilitate a “squeeze out” of dissenting minority shareholders upon a tender offer. When a tender offer is made and accepted by holders of 90% of the shares affected within four months, the offeror may, within a two-month period commencing on the expiration of such four-month period, require the holders of the remaining shares to transfer such shares to the offeror on the terms of the offer. An objection can be made to the Grand Court of the Cayman Islands but this is unlikely to succeed in the case of an offer which has been so approved unless there is evidence of fraud, bad faith or collusion.
Shareholder Suits
In principle, we will normally be the proper plaintiff in any claim based on a breach of duty owed to us, and as a general rule a derivative action may not be brought by a minority shareholder. However, based on English authorities, which would in all likelihood be of persuasive authority in the Cayman Islands, the Cayman Islands courts can be expected to follow and apply the common law principles which permit a minority shareholder to commence a class action against or derivative actions in the name of the company to challenge actions where: (a) a company acts or proposes to act illegally or beyond the scope of its authority; (b) the act complained of, although not beyond the scope of its authority, could only be effected duly if authorized by more than a simple majority vote that has not been obtained; and (c) those who control the company are perpetrating a “fraud on the minority.”
Anti-Takeover Provisions
Some provisions of the Amended Articles may discourage, delay or prevent a change of control of the Company or management that shareholders may consider favorable, including a provision that authorizes the board of directors to issue shares in one or more series and to designate the price, rights, preferences, privileges and restrictions of such shares without any further vote or action by the shareholders. However, under Cayman Islands law, our directors may only exercise the rights and powers granted to them under the Amended Articles for a proper purpose and for what they believe in good faith to be in the best interests of the Company.
Directors’ Fiduciary Duties
Under Cayman Islands law, directors owe the following fiduciary duties: (a) a duty to act in good faith in what the directors consider to be in the best interests of the company as a whole; (b) a duty to exercise their powers for a proper purpose and not for a collateral purpose; (c) a duty not to put themselves in a position where their interests conflict with the duties they owe to the company (unless the company consents); and (d) a duty to exercise independent judgment.
In addition to the above, directors owe a duty of care to the company which is not fiduciary in nature. This duty has been defined as a requirement to act as a reasonably diligent person having both the general knowledge, skill and
90
experience that may reasonably be expected of a person carrying out the same functions as are carried out by that director in relation to the company, and the general knowledge, skill and experience of that particular director.
As set out above, directors have a duty not to put themselves in a position of conflict and this includes a duty not to engage in self-dealing transactions. However, in some instances, the duty not to be in a position of conflict may be released by shareholders provided certain conditions are satisfied.
Shareholder Action by Written Resolution
The Companies Act permits a Cayman Islands exempted company to adopt articles of association which provide for shareholder action by written resolution signed by all shareholders entitled to vote thereon. The Amended Articles provide that any action required or permitted to be taken at a meeting of shareholders may be effected by a written resolution passed in accordance with the Amended Articles.
Shareholder Proposals
Under the Companies Act, shareholders have no right to put any proposal before a meeting of shareholders unless such right is specified in the articles of association. The Amended Articles allow shareholders holding not less than one third of the votes that may be cast at such meeting to requisition an extraordinary general meeting. As a Cayman Islands exempted company, we are not obligated by law to call shareholders’ annual general meetings.
Cumulative Voting
Under Cayman Islands law, there is no requirement for cumulative voting. The Amended Articles do not provide for cumulative voting.
Removal of Directors
Under the Amended Articles, directors may be removed by ordinary resolution of the shareholders. The Companies Act also provides that a director may be removed by ordinary resolution.
Transactions with Interested Shareholders
The Companies Act has no comparable statute to Section 203 of the Delaware General Corporation Law, which regulates self-dealing transactions. However, Cayman Islands law provides that transactions entered into by a company in which a director has an interest are voidable by the company unless the director’s interest is disclosed and the directors approve the transaction.
Dissolution and Winding Up
Under the Companies Act, a company may be wound up by either an order of the courts of the Cayman Islands or by a special resolution of its members. The court has authority to order winding up in a number of specified circumstances, including where it is, in the opinion of the court, just and equitable to do so. Under the Companies Act and the Amended Articles, the Company may be dissolved, liquidated or wound up by a special resolution of our shareholders.
Variation of Rights of Shares
Under the Companies Act, if the share capital of a company is divided into different classes of shares, the rights attached to any class may only be varied with the written consent of the holders of two-thirds of the issued shares of that class or with the approval of a resolution passed by a majority of not less than two-thirds of the votes cast at a separate class meeting, subject to any additional provisions in the company’s articles of association. The Amended Articles include a provision permitting the directors to vary the rights of any class of shares if such variation is not materially adverse, as described above under “Variation of Rights of Shares.”
Transfer of Shares
Under Cayman Islands law, the transfer of shares in a Cayman Islands company may be restricted in the company’s articles of association. The Amended Articles contain provisions restricting the transfer of Class B Ordinary Shares as
91
described above under “Transfer of Ordinary Shares.” There are no restrictions on the transfer of Class A Ordinary Shares in the Amended Articles other than such restrictions as may be imposed by applicable securities laws.
Inspection of Corporate Records
Members of a Cayman Islands exempted company do not have a general right to inspect or obtain copies of the company’s list of shareholders or corporate records (other than copies of its memorandum and articles of association, any special resolutions, and its register of mortgages and charges). These rights may, however, be granted in the articles of association.
C.Material Contracts
Other than contracts entered into in the ordinary course of business, and other than those described in “Item 3. Risk Factors — Risks Relating to Our Business and Industry,” “Item 4. Information on the Company,” “Item 5. Operating and Financial Review and Prospects,” “Item 7. Major Shareholders and Related Party Transactions — B. Related Party Transactions,” or described elsewhere in this annual report, the following contracts summarized below are the material contracts that the Company has been a party to for the two years preceding the publication of this Annual Report.
Sale of ACT Genomics
On June 20, 2025, we entered into an Agreement for Sale and Purchase of the issued shares in ACT Genomics Holdings Company Limited ("ACT Genomics") with Delta Electronics, Inc. ("Delta") as buyer, certain other shareholders of ACT Genomics as co-sellers, and Chen Hua Chien as the holders' representative. Pursuant to the agreement, Delta agreed to acquire 100% of the issued shares of ACT Genomics from all shareholders, including Prenetics (which held approximately 73.3% of the issued shares). The aggregate headline price for all sale shares and option shares was capped at approximately US$71.8 million (multiplied by the applicable sale ratio). Prenetics' sale shares headline price was approximately US$46.3 million. The initial consideration was subject to customary post-completion adjustments for working capital, net debt and transaction expenses, determined in accordance with completion accounts prepared following closing. An adjustment escrow amount, an indemnity escrow amount and a tax indemnity escrow amount, collectively in the amount of approximately $6.3 million attributable to Prenetics, were held in escrow pending the completion of certain customary release conditions. The agreement contains customary representations, warranties and indemnities from the sellers. Completion was conditional on, among other things, regulatory approvals, third-party consents (including from key commercial partners), and delivery of audited financial statements. Completion of the sale and purchase transaction occurred on October 1, 2026.
Acquisition of Europa Group
On May 23, 2024, we entered into a Unit Purchase Agreement with ACM Europa VII-C LLC, AFI Partners Fund IV, and AFI Partners LLC to acquire Europa Partners Holdings, LLC, including its subsidiaries Europa Sports Partners, LLC and Hubmatrix Partners, LLC (collectively, the "Europa Group"). We acquired 100% of the membership interests in the Europa Group, a leading distributor of sports nutrition and health products in the United States, for a nominal cash consideration of $10, aligning with our strategic expansion into the United States health and wellness market.
In relation to the acquisition, we also issued a $9 million Secured Revolving Promissory Note to Europa to support the Europa Group's working capital needs, paid off $1.45 million under a reduced loan settlement with Rosenthal & Rosenthal, Inc., and assumed some other operational liabilities. As part of the acquisition, we restructured operations to integrate the Europa Group’s brick-and-mortar and e-commerce distribution platforms, supporting the launch of our consumer health brand, IM8.
Sale of Europa 3PL Business
On January 1, 2026, we entered into an Asset Purchase Agreement (the "APA") with Europa Sports Partners, LLC and Hubmatrix Partners, LLC (as sellers), IM8 Global Limited, Stord Fulfillment, LLC (as buyer, a wholly-owned subsidiary of Stord, Inc.) and Stord, Inc. (as buyer parent). Pursuant to the APA, we sold substantially all of the assets of our Europa third-party logistics (“3PL”) business, including inventory, assigned contracts (customer, vendor, warehouse and equipment lease agreements), intellectual property assets and tangible personal property, to Stord Fulfillment, LLC. The buyer assumed certain liabilities relating to the assigned contracts arising after closing, as well as liabilities arising from its ownership and operation of the purchased assets on or after closing. The aggregate purchase price consists of share consideration payable in shares of non-voting common stock of Stord, Inc., comprising: (1) closing shares having an aggregate value of $3 million, subject to a vesting condition tied to a performance milestone; and (2) contingent share consideration of up to $10 million, issuable in tranches of $2.5 million each upon achievement of certain additional performance milestones.
92
Insighta Share Sale Agreement and Share Subscription Agreement
On June 26, 2023, we entered into definitive transaction documents to establish a joint venture between Prenetics, Professor Dennis Lo, and others, namely Insighta, on June 25, 2023. The definitive agreements consisted of a share subscription agreement among Insighta Holdings Limited and Prenetics Global Limited, and a share sale agreement among Prenetics Global Limited, Professor Dennis Lo, and others. Pursuant to the definitive agreements, Prenetics received a 50% equity stake in Insighta for a total consideration of US$100 million, which comprised of US$80 million cash consideration, and a US$20 million share consideration consisting of 22,222,222 newly issued Class A Ordinary Shares issued at an issue price of US$0.90 per Class A Ordinary Share (equivalent to 1,481,482 shares at a price of US$13.5 after the reverse stock split).
Registration Rights Agreement
In connection with the Insighta Share Sale Agreement, we entered into a Registration Rights Agreement with Professor Dennis Lo and AC-Tech Investment Limited on July 20, 2023, pursuant to which, among other things, the Company agreed to undertake certain resale shelf registration obligations in accordance with the Securities Act and granted Professor Dennis Lo and AC-Tech Investment Limited customary demand and piggyback registration rights.
Tencent’s $30 Million Investment in Insighta
On October 14, 2024, Prenetics entered into a Share Purchase Agreement with Image Frame Investment (HK) Limited ("Image Frame"), an affiliate of Tencent Holdings Limited. Pursuant to this agreement, Image Frame invested $30 million in Insighta, acquiring a 15% equity interest from us. Consequently, our ownership in Insighta decreased from 50% to 35%, while Insighta's co-founders retained their 50% stake. As part of the transaction, we received $30 million in cash from the sale of secondary shares to Image Frame. Additionally, Tencent appointed Alexander Ng, President of Tencent Healthcare, to the seat on Insighta's board.
Tencent’s Subsequent $70 Million Investment in Insighta
On February 13, 2026, we entered into a Share Purchase Agreement with Image Frame. Pursuant to this agreement, we agreed to sell to Image Frame 700,000 Class A shares in Insighta, representing approximately 35% of Insighta's total issued shares on an as-converted and fully diluted basis, for a cash purchase price of US$70,000,000, of which US$1,000,000 is held in escrow pending satisfaction of certain post-completion obligations. Completion was conditional on customary pre-closing conditions. In connection with completion, Danny Yeung and Yin Pan Cheng resigned as directors of Insighta and were replaced by nominees of Image Frame and Professor Dennis Lo. This transaction represented our full divestiture of our remaining equity interest in Insighta.
October 2025 Equity Offering
On October 28, 2025, we closed a best-efforts public offering (the "October 2025 Offering") of 2,722,642 Class A Ordinary Shares, together with accompanying Class A Warrants to purchase up to 2,722,642 Class A Ordinary Shares at an exercise price of US$24.12 per share, and Class B Warrants to purchase up to 2,722,642 Class A Ordinary Shares at an exercise price of US$32.16 per share, at a combined offering price of US$16.08 per Class A Ordinary Share and accompanying Class A Warrant and Class B Warrant. The Class A Warrants and Class B Warrants are immediately exercisable upon issuance for a period of five years from the closing date. We received gross proceeds of approximately US$44 million from the October 2025 Offering, before deducting placement agent fees and other offering expenses. Dominari Securities LLC acted as sole placement agent pursuant to a Placement Agency Agreement dated October 27, 2025, for which it received a cash fee and warrants to purchase 131,829 Class A Ordinary Shares at an exercise price of US$16.08 per share. Continental Stock Transfer and Trust Company acts as warrant agent pursuant to a warrant agency agreement dated October 28, 2025. The October 2025 Offering was made pursuant to our effective registration statement on Form F-3 (File No. 333-288824), as supplemented. For further details, see the Company's report on Form 6-K filed with the SEC on October 28, 2025.
December 2025 Warrant Exchange
On December 23, 2025 and December 31, 2025, we entered into warrant exchange agreements with certain holders of Class A Warrants and Class B Warrants issued in the October 2025 Offering, pursuant to which such holders exchanged their existing Class A Warrants and Class B Warrants for new Class C Warrants at a ratio of two existing warrants for one Class C Warrant. Each Class C Warrant entitles its holder to purchase one Class A Ordinary Share at an exercise price of US$18.00 per share, for a period of two years following the effectiveness of a registration statement on Form F-3 registering the resale of the underlying shares. In connection with the warrant exchange, we agreed to prepare and file such registration statement with the SEC. Continental Stock Transfer and Trust Company acts as warrant agent for the Class C Warrants. For further details regarding the warrant exchange and the terms of the Class C Warrants, see the Company's report on Form 6-K filed with the SEC on December 23, 2025.
93
MZHCI, LLC Consulting Agreement
On April 25, 2024, we entered into an investor relations consulting agreement with MZHCI, LLC ("MZHCI"), a MZ Group company, pursuant to which MZHCI provides investor relations, investment awareness, financial media relations and public market advisory services to the Company. The initial agreement had an initial term of nine months from April 25, 2024, with automatic monthly renewals thereafter unless terminated by either party on 30 days' written notice.
On August 12, 2025, we entered into a new investor relations consulting agreement with MZHCI, as amended by an Amendment Agreement dated September 5, 2025, which superseded the 2024 agreement. The new agreement has an initial term of six months, renewable by mutual written agreement. As of the date of this annual report, the agreement remains effective. MZHCI is entitled to certain cash and equity compensation under the agreement, including upon the completion of certain performance-based milestones. MZHCI performs its services as an independent contractor.
Roth Consulting Agreement
On July 5, 2024, we entered into a capital markets advisory engagement letter with Roth Capital Partners, LLC ("Roth"), pursuant to which Roth agreed to provide capital markets advisory services to the Company for a period of twelve months, including advice relating to the development of our short and long-term capital markets strategy, analysis of our business, industry, competition and anticipated capital requirements as they relate to our valuation, strategic development of institutional investor relationships, attendance at Roth's sector and annual conferences, and meetings with Roth's institutional sales personnel.
As consideration for Roth's services, we agreed to pay a capital markets advisory fee equivalent to 130,000 Class A Ordinary Shares, to be satisfied twelve months after execution of the agreement, through the issuance of fully vested restricted share units convertible into 130,000 Class A Ordinary Shares at no exercise cost under our 2022 Share Incentive Plan. If we are unable to issue the award as restricted share units under the 2022 Share Incentive Plan, we are instead required to issue 130,000 Class A Ordinary Shares to Roth, together with customary registration rights requiring us to file a resale registration statement with the SEC no later than fifteen (15) days after issuance and to use commercially reasonable efforts to cause such registration statement to become and remain effective until the shares are freely tradable by non-affiliates under Rule 144 of the Securities Act.
D.Exchange Controls
There are no governmental laws, decrees, regulations or other legislation in the Cayman Islands that may affect the import or export of capital, including the availability of cash and cash equivalents for use by the Company, or that may affect the remittance of dividends, interest, or other payments by the Company to non-resident holders of its ordinary shares.
E.Taxation
U.S. Federal Income Tax Considerations to U.S. Holders
General
The following is a general discussion of the material U.S. federal income tax consequences to U.S. Holders (as defined below) of the acquisition, ownership and disposition of our Class A Ordinary Shares and Warrants. No ruling has been requested or will be obtained from the IRS regarding the U.S. federal income tax consequences of the acquisition, ownership and disposition of our Class A Ordinary Shares and Warrants; thus, there can be no assurance that the IRS will not challenge the U.S. federal income tax treatment described below or that, if challenged, such treatment will be sustained by a court.
This summary is limited to U.S. federal income tax considerations relevant to U.S. Holders that hold our Class A Ordinary Shares or Warrants as “capital assets” within the meaning of section 1221 of the Internal Revenue Code of 1986, as amended (the “Code”) (generally, property held for investment). This discussion does not address all aspects of U.S. federal income taxation that may be important to holders in light of their individual circumstances, including holders subject to special treatment under the U.S. tax laws, such as, for example:
•our officers or directors;
•banks, financial institutions or financial services entities;
•broker-dealers;
•taxpayers that are subject to the mark-to-market accounting rules;
94
•tax-exempt entities;
•S-corporations, partnerships and other pass-through entities or arrangements for U.S. federal income tax purposes, and any owners of any such S-corporations, partnerships or other pass-through entities or arrangements;
•governments or agencies or instrumentalities thereof;
•insurance companies;
•regulated investment companies;
•real estate investment trusts;
•expatriates or former long-term residents of the United States;
•accrual method taxpayers that file applicable financial statements as described in Section 451(b) of the Code;
•persons that actually or constructively own five percent or more of our shares by vote or value;
•persons that acquired our securities pursuant to an exercise of employee share options, in connection with employee share incentive plans or otherwise as compensation or in connection with services;
•persons subject to the alternative minimum tax or the base erosion and anti-abuse tax;
•persons that hold our securities as part of a straddle, constructive sale, hedging, conversion or other integrated or similar transaction; or
•U.S. Holders (as defined below) whose functional currency is not the U.S. dollar.
As used in this annual report, the term “U.S. Holder” means a beneficial owner of our Class A Ordinary Shares or Warrants that is for U.S. federal income tax purposes:
•an individual citizen or resident of the United States;
•a corporation (or other entity treated as a corporation for U.S. federal income tax purposes) that is created or organized (or treated as created or organized) in or under the laws of the United States, any state thereof or the District of Columbia;
•an estate the income of which is subject to U.S. federal income taxation regardless of its source; or
•a trust if (A) a court within the United States is able to exercise primary supervision over the administration of the trust and one or more U.S. persons have the authority to control all substantial decisions of the trust, or (B) it has in effect under applicable U.S. Treasury regulations a valid election to be treated as a U.S. person.
Moreover, the discussion below is based upon the provisions of the Code, the Treasury regulations promulgated thereunder and administrative and judicial interpretations thereof, all as of the date hereof. Those authorities may be repealed, revoked, modified or subject to differing interpretations, possibly on a retroactive basis, so as to result in U.S. federal income tax consequences different from those discussed below. Furthermore, this discussion does not address any aspect of U.S. federal non-income tax laws, such as gift or estate tax laws, state, local or non-U.S. tax laws, or the Medicare tax on net investment income.
This discussion does not consider the tax treatment of partnerships (including any entity or arrangement classified as a partnership for U.S. federal income tax purposes) or other pass-through entities for U.S. federal income tax purposes or persons who hold our securities through such entities. If a partnership (or other entity or arrangement classified as a partnership for U.S. federal income tax purposes) is the beneficial owner of our securities, the U.S. federal income tax treatment of the partnership or a partner in the partnership will generally depend on the status of the partner and the activities of the partner and the partnership. If you are a partnership or a partner of a partnership holding our securities, we urge you to consult your own tax advisor.
This summary does not purport to be a comprehensive analysis or description of all potential U.S. federal income tax consequences of acquiring, owning and disposing of our Class A Ordinary Shares or Warrants. Holders of our Class A Ordinary Shares or Warrants should consult with their tax advisors regarding the particular tax consequences to them of the acquisition, ownership and disposition of our Class A Ordinary Shares or Warrants, including the applicability and effects of U.S. federal, state, local, and other tax laws.
95
Taxation of Distributions
We do not anticipate paying any cash distributions on our Class A Ordinary Shares in the foreseeable future. However, subject to the possible applicability of the PFIC rules discussed below under “Passive Foreign Investment Company Status,” if we do make a distribution of cash or other property on our Class A Ordinary Shares, a U.S. Holder will generally be required to include in gross income as a dividend the amount of any distribution paid on our Class A Ordinary Shares to the extent the distribution is paid out of our current or accumulated earnings and profits (as determined under U.S. federal income tax principles). Subject to the PFIC rules described below, distributions in excess of such earnings and profits will generally be applied against and reduce the U.S. Holder’s basis in our Class A Ordinary Shares (but not below zero) and, to the extent in excess of such basis, will generally be treated as capital gain from the sale or exchange of such Class A Ordinary Shares (see “— Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of Class A Ordinary Shares and Warrants” below). We do not intend to provide calculations of our earnings and profits under U.S. federal income tax principles. A U.S. Holder should expect all cash distributions to be reported as dividends for U.S. federal income tax purposes. Any dividend will generally not be eligible for the dividends received deduction allowed to corporations in respect of dividends received from U.S. corporations.
With respect to non-corporate U.S. Holders, under tax laws currently in effect and subject to certain exceptions, dividends with respect to our Class A Ordinary Shares will generally be taxed at the lower applicable long-term capital gains rate (see “— Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of Class A Ordinary Shares and Warrants” below) provided that our Class A Ordinary Shares are readily tradable on an established securities market in the United States, and we are not treated as a PFIC in the year the dividend is paid or in the preceding year and certain holding period and other requirements are met. U.S. Treasury Department guidance indicates that shares listed on Nasdaq (on which the Class A Ordinary Shares are listed) will be considered readily tradable on an established securities market in the United States. Even if the Class A Ordinary Shares are listed on Nasdaq, there can be no assurance that our Class A Ordinary Shares will be considered readily tradable on an established securities market in future years. U.S. Holders should consult their tax advisors regarding the availability of such lower rate for any dividends paid with respect to Class A Ordinary Shares.
Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of Class A Ordinary Shares and Warrants
Subject to the PFIC rules described below under “Passive Foreign Investment Company Status,” a U.S. Holder will generally recognize capital gain or loss on the sale or other taxable disposition of our Class A Ordinary Shares or Warrants in an amount equal to the difference between (i) the amount of cash and the fair market value of any property received in the disposition, and (ii) such U.S. Holder’s adjusted tax basis in such Class A Ordinary Shares or Warrants disposed. A U.S. Holder’s adjusted tax basis in its Class A Ordinary Shares or Warrants will generally equal the U.S. Holder’s acquisition cost reduced by any prior distributions treated as a return of capital. Please see “— Exercise, Lapse or Redemption of a Warrant” below for a discussion regarding a U.S. Holder’s basis in a Class A Ordinary Share acquired pursuant to the exercise of a Warrant.
Any such capital gain or loss generally will be long-term capital gain or loss if the U.S. Holder’s holding period for such Class A Ordinary Shares or Warrants exceeds one year. Long-term capital gain realized by a non-corporate U.S. Holder is currently eligible to be taxed at reduced rates. The deduction of capital losses is subject to certain limitations.
Exercise, Lapse or Redemption of a Warrant
Subject to the PFIC rules described below under “Passive Foreign Investment Company Status” and except as discussed below with respect to the cashless exercise of a Warrant, a U.S. Holder will generally not recognize gain or loss upon the acquisition of a Class A Ordinary Share on the exercise of a Warrant for cash. A U.S. Holder’s tax basis in a Class A Ordinary Share received upon exercise of the Warrant will generally be an amount equal to the sum of the U.S. Holder’s tax basis in the Warrant exchanged therefor and the exercise price. The U.S. Holder’s holding period for a Class A Ordinary Share received upon exercise of the Warrant will begin on the date following the date of exercise (or possibly the date of exercise) of the Warrant and will not include the period during which the U.S. Holder held the Warrant. If a Warrant is allowed to lapse unexercised, a U.S. Holder will generally recognize a capital loss equal to such holder’s tax basis in the Warrant.
The tax consequences of a cashless exercise of a Warrant are not clear under current U.S. federal income tax law. Subject to the PFIC rules discussed below, a cashless exercise may be tax-free, either because the exercise is not a realization event or because the exercise is treated as a “recapitalization” for U.S. federal income tax purposes. Although we expect a U.S. Holder’s cashless exercise of our Warrants (including after we provide notice of our intent to redeem
96
warrants for cash) to be treated as a recapitalization, a cashless exercise could alternatively be treated as a taxable exchange in which gain or loss would be recognized.
In either tax-free situation, a U.S. Holder’s tax basis in the Class A Ordinary Shares received would generally equal the U.S. Holder’s tax basis in the Warrants exercised therefor. If the cashless exercise is not treated as a realization event, it is unclear whether a U.S. Holder’s holding period for the Class A Ordinary Share will commence on the date of exercise of the Warrant or the day following the date of exercise of the Warrant. If the cashless exercise is treated as a recapitalization, the holding period of the Class A Ordinary Shares would include the holding period of the Warrants exercised therefor.
It is also possible that a cashless exercise may be treated in part as a taxable exchange in which gain or loss would be recognized. In such event, a U.S. Holder could be deemed to have surrendered Warrants with an aggregate fair market value equal to the exercise price for the total number of Warrants to be exercised. Subject to the PFIC rules discussed below, the U.S. Holder would recognize capital gain or loss in an amount equal to the difference between the fair market value of the warrants deemed surrendered and the U.S. Holder’s adjusted tax basis in such Warrants. In this case, a U.S. Holder’s tax basis in the Class A Ordinary Shares received would equal the U.S. Holder’s tax basis in the Warrants exercised plus the exercise price of such warrants. It is unclear whether a U.S. Holder’s holding period for the Class A Ordinary Shares would commence on the date of exercise of the warrant or the day following the date of exercise of the Warrants.
Due to the absence of authority on the U.S. federal income tax treatment of a cashless exercise, there can be no assurance which, if any, of the alternative tax consequences and holding periods described above would be adopted by the IRS or a court of law. Accordingly, a U.S. Holder should consult its tax advisor regarding the tax consequences of a cashless exercise.
Subject to the PFIC rules described below, if we redeem Warrants for cash or purchase Warrants in an open market transaction, such redemption or purchase will generally be treated as a taxable disposition to the U.S. Holder, taxed as described above under “— Gain or Loss on Sale, Taxable Exchange or Other Taxable Disposition of Class A Ordinary Shares and Warrants.”
Possible Constructive Distributions
The terms of each Warrant provide for an adjustment to the number of Class A Ordinary Shares for which the Warrant may be exercised or to the exercise price of the warrant in certain events, as discussed in the section titled “Description of Warrants” in Exhibit 2.5 to this annual report. An adjustment which has the effect of preventing dilution generally is not taxable. The U.S. Holders of the Warrants would, however, be treated as receiving a constructive distribution from us if, for example, the adjustment increases such U.S. Holders’ proportionate interests in our assets or earnings and profits (e.g. through an increase in the number of Class A Ordinary Shares that would be obtained upon exercise or through a decrease to the exercise price of a Warrant) as a result of a distribution of cash or other property to the holders of Class A Ordinary Shares which is taxable to the U.S. Holders of such Class A Ordinary Shares as described under “— Taxation of Distributions” above. Such constructive distribution would be subject to tax as described under that section in the same manner as if the U.S. Holders of the Warrants received a cash distribution from us equal to the fair market value of such increased interest.
Passive Foreign Investment Company Status
The treatment of U.S. Holders of our Class A Ordinary Shares and Warrants could be materially different from that described above if we are or were treated as a passive foreign investment company (“PFIC”) for U.S. federal income tax purposes.
A non-U.S. corporation will be classified as a PFIC for U.S. federal income tax purposes if either (i) at least 75% of its gross income in a taxable year, including its pro rata share of the gross income of any corporation in which it is considered to own at least 25% of the shares by value, is passive income or (ii) at least 50% of its assets in a taxable year (ordinarily determined based on fair market value and averaged quarterly over the year), including its pro rata share of the assets of any corporation in which it is considered to own at least 25% of the shares by value, are held for the production of, or produce, passive income. Passive income generally includes dividends, interest, rents and royalties (other than rents or royalties derived from the active conduct of a trade or business) and gains from the disposition of passive assets. Due to the lack of authority and guidance, the application of such rules with respect to digital assets, or transactions involving digital assets, is subject to uncertainty, although digital assets are likely generally treated as passive assets.
97
Based on our composition of assets and market capitalization (which is subject to fluctuation), and treating our digital assets as passive assets for such purposes, we believe that we were not a PFIC for the taxable year ended December 31, 2025. There can be no assurance regarding our PFIC status for the current taxable year or foreseeable future taxable years, however, because our PFIC status is a factual determination made annually that will depend, in part, upon the composition of our income and assets, and includes uncertainty under the PFIC rules with respect to digital assets and transactions involving digital assets. The value of our assets for purposes of the asset test, including the value of our goodwill and unbooked intangibles, may be determined in part by reference to the market price of our Class A Ordinary Shares from time to time (which may be volatile). Because we will generally take into account our current market capitalization in estimating the value of our goodwill and other unbooked intangibles, our PFIC status for the current taxable year and foreseeable future taxable years may be affected by our market capitalization, and therefore, fluctuations in our market capitalization create a risk that we may be classified as a PFIC for the current taxable year and foreseeable future taxable years. In addition, the composition of our income and assets will be affected by how, and how quickly, we spend our liquid assets. Under circumstances in which our revenue from activities that produce passive income significantly increases relative to our revenue from activities that produce non-passive income, or in which we determine not to deploy significant amounts of cash for active purposes or our holdings of digital assets increase relative to our other assets, our risk of becoming classified as a PFIC may substantially increase.
Because there are uncertainties in the application of the relevant rules, it is possible that the Internal Revenue Service (the “IRS”) may challenge our classification of certain income or assets as non-passive, or our valuation of our goodwill and other unbooked intangibles, each of which could cause us to become classified as a PFIC for the current or subsequent taxable years. If we are classified as a PFIC for any taxable year during which a U.S. Holder holds our Class A Ordinary Shares or Warrants, the PFIC rules discussed below will generally apply to such U.S. Holder for such taxable year, and unless the U.S. Holder makes certain elections, will apply in future taxable years even if we cease to be a PFIC.
It is not entirely clear how various aspects of the PFIC rules apply to the Warrants. Section 1298(a)(4) of the Code provides that, to the extent provided in Treasury Regulations, any person who has an option to acquire stock in a PFIC shall be considered to own such stock in the PFIC for purposes of the PFIC rules. No final Treasury Regulations are currently in effect under Section 1298(a)(4) of the Code. However, proposed Treasury Regulations under Section 1298(a)(4) of the Code have been promulgated that, if finalized in their current form, would have a retroactive effective date (the “Proposed PFIC Option Regulations”). Each prospective investor is urged to consult its tax advisors regarding the possible application of the Proposed PFIC Option Regulations to the Warrants. The following discussion assumes that the Proposed PFIC Option Regulations will apply to the Warrants.
If we are determined to be a PFIC for any taxable year (or portion thereof) that is included in the holding period of a U.S. Holder of Class A Ordinary Shares or Warrants and, in the case of Class A Ordinary Shares, the U.S. Holder did not make an applicable purging election or a mark-to-market election, such U.S. Holder would generally be subject to special and adverse rules with respect to (i) any gain recognized by the U.S. Holder on the sale or other disposition of its Class A Ordinary Shares or Warrants and (ii) any “excess distribution” made to the U.S. Holder (generally, any distributions to such U.S. Holder during a taxable year of the U.S. Holder that are greater than 125% of the average annual distributions received by such U.S. Holder in respect of the Class A Ordinary Shares during the three preceding taxable years of such U.S. Holder or, if shorter, such U.S. Holder’s holding period for the Class A Ordinary Shares).
Under these rules:
•the U.S. Holder’s gain or excess distribution will be allocated ratably over the U.S. Holder’s holding period for the Class A Ordinary Shares or Warrants;
•the amount allocated to the U.S. Holder’s taxable year in which the U.S. Holder recognized the gain or received the excess distribution, or to the period in the U.S. Holder’s holding period before the first day of our first taxable year in which we were a PFIC, will be taxed as ordinary income;
•the amount allocated to other taxable years (or portions thereof) of the U.S. Holder and included in its holding period will be taxed at the highest tax rate in effect for that year and applicable to the U.S. Holder; and
•an additional tax equal to the interest charge generally applicable to underpayments of tax will be imposed on the U.S. Holder with respect to the tax attributable to each such other taxable year of the U.S. Holder.
If we are a PFIC and, at any time, have a non-U.S. subsidiary that is classified as a PFIC, a U.S. Holder would generally be deemed to own a portion of the shares of such lower-tier PFIC, and generally could incur liability for the deferred tax and interest charge described above if we (or our subsidiary) receive a distribution from, or dispose of all or
98
part of the interest in, the lower-tier PFIC or the U.S. Holders otherwise were deemed to have disposed of an interest in the lower-tier PFIC. U.S. Holders are urged to consult their tax advisors regarding the tax issues raised by lower-tier PFICs.
QEF Election, Market-Market Election and Purging Election
In general, a U.S. Holder may avoid the adverse PFIC tax consequences described above in respect of such U.S. Holder’s Class A Ordinary Shares (but not Warrants) by making and maintaining a timely and valid QEF election (if eligible to do so) to include in income its pro rata share of our net capital gains (as long-term capital gain) and other earnings and profits (as ordinary income), on a current basis, in each case whether or not distributed, in the taxable year of the U.S. Holder in which or with which our taxable year ends. A U.S. Holder generally may make a separate election to defer the payment of taxes on undistributed income inclusions under the QEF rules, but if deferred, any such taxes will be subject to an interest charge.
As discussed above, it is not entirely clear how various aspects of the PFIC rules apply to the Warrants. However, a U.S. Holder may not make a QEF election with respect to its Warrants. As a result, if a U.S. Holder sells or otherwise disposes of such Warrants (other than upon exercise of such Warrants for cash) and we were a PFIC at any time during the U.S. Holder’s holding period of such Warrants, any gain recognized will generally be treated as an excess distribution, taxed as described above. If a U.S. Holder that exercises such Warrants properly makes and maintains a QEF election with respect to the newly acquired Class A Ordinary Shares (or has previously made a QEF election with respect to Class A Ordinary Shares), the QEF election will apply to the newly acquired Class A Ordinary Shares. Notwithstanding such QEF election, the adverse tax consequences relating to PFIC shares, adjusted to take into account the current income inclusions resulting from the QEF election, will continue to apply with respect to such newly acquired Class A Ordinary Shares (which will generally be deemed to have a holding period for purposes of the PFIC rules that includes the period the U.S. Holder held the Warrants), unless the U.S. Holder makes a purging election under the PFIC rules.
Under one type of purging election, the U.S. Holder will be deemed to have sold such shares at their fair market value and any gain recognized on such deemed sale will be treated as an excess distribution, as described above. As a result of this purging election, the U.S. Holder will have a new basis and holding period in the Class A Ordinary Share acquired upon the exercise of the Warrants solely for purposes of the PFIC rules.
The QEF election is made on a shareholder-by-shareholder basis and, once made, can be revoked only with the consent of the IRS. A U.S. Holder generally makes a QEF election by attaching a completed IRS Form 8621 (Information Return by a Shareholder of a Passive Foreign Investment Company or Qualified Electing Fund), including the information provided in a PFIC Annual Information Statement, to a timely filed U.S. federal income tax return for the tax year to which the election relates. Retroactive QEF elections generally may be made only by filing a “protective statement” (as defined in Treasury regulations) with such timely filed return and if certain other conditions are met (including that the shareholder reasonably believed that as of the election due date we were not a PFIC for our taxable year that ended during the retroactive election year) or with the consent of the IRS. U.S. Holders are urged to consult their tax advisors regarding the availability and tax consequences of a retroactive QEF election under their particular circumstances.
A U.S. Holder that does not make a timely QEF election in the first taxable year (or portion thereof) in which we are a PFIC that is included in the holding period of the Class A Ordinary Shares of such U.S. Holder may be able to mitigate the adverse PFIC tax consequences by making a QEF election in a subsequent taxable year and simultaneously making a purging election under the PFIC rules. Under one type of purging election, the U.S. Holder will be deemed to have sold our Class A Ordinary Shares at their fair market value and any gain recognized on such deemed sale will be treated in the same manner as an excess distribution, taxed as described above. As a result of this purging election, the U.S. Holder will have additional basis (to the extent of any gain recognized on the deemed sale). U.S. Holders are strongly urged to consult with and rely solely upon their tax advisors regarding the application of the rules governing purging elections to their particular circumstances.
Please note that in order to comply with the requirements of any QEF election, a U.S. Holder must receive a PFIC Annual Information Statement from us. We have not determined whether we will provide U.S. Holders this information if we determine that we are or will become a PFIC. As noted above, we currently do not intend to maintain or provide calculations of our earnings and profits under U.S. federal income tax principles. There is also no assurance that we will have timely knowledge of our PFIC status in the future or of the required information to be provided. Moreover, we are not required to endeavor to cause any lower-tier PFIC to provide such U.S. Holder the information that may be required to make or maintain a QEF election with respect to such lower-tier PFIC. There can be no assurance that we will have timely knowledge of the status of any such lower-tier PFIC. In addition, we may not hold a controlling interest in any such lower-
99
tier PFIC and thus there can be no assurance could (even if we elected to try) cause the lower-tier PFIC to provide such required information.
Alternatively, if a U.S. Holder makes a valid mark-to-market election for the first taxable year of the U.S. Holder in which the U.S. Holder holds (or is deemed to hold) our Class A Ordinary Shares and in which we are treated as a PFIC, such U.S. Holder generally will not be subject to the PFIC rules described above in respect of such Class A Ordinary Shares. Such U.S. Holder will generally include for each of its taxable years as ordinary income the excess, if any, of the fair market value of its Class A Ordinary Shares at the end of such year over its adjusted basis in its Class A Ordinary Shares. The U.S. Holder also will recognize an ordinary loss in respect of the excess, if any, of its adjusted basis of its Class A Ordinary Shares over the fair market value of its Class A Ordinary Shares at the end of its taxable year (but only to the extent of the net amount of previously included income as a result of the mark-to-market election). The U.S. Holder’s basis in its Class A Ordinary Shares will be adjusted to reflect any such income or loss amounts, and any further gain recognized on a sale or other taxable disposition of its Class A Ordinary Shares will be treated as ordinary income. Currently, a mark-to-market election may not be made with respect to the Warrants.
Any mark-to-market election made by a U.S. Holder for the Class A Ordinary Shares will also apply to such U.S. Holder’s Class A Ordinary Shares received upon exercise of Warrants. As a result, if a mark-to-market election has been made by a U.S. Holder with respect to our Class A Ordinary Shares, any Class A Ordinary Shares received upon exercise of Warrants will automatically be marked-to-market in the year of exercise. Because a U.S. Holder’s holding period for Class A Ordinary Shares received upon exercise of a Warrant should, be treated for purposes of the Proposed PFIC Option Regulations to include the period during which such U.S. Holder held such Warrant, a U.S. Holder will be treated as making a mark-to-market election with respect to such Class A Ordinary Shares after the beginning of such U.S. Holder’s holding period for such Class A Ordinary Shares unless such Class A Ordinary Shares are acquired in the same tax year as the year in which the U.S. Holder acquired its Warrants. Consequently, the excess distribution regime described above generally will apply to the mark-to-market gain realized in the tax year in which such Class A Ordinary Shares are received upon the exercise of the Warrants. However, the general mark-to-market rules will apply to subsequent tax years.
If the U.S. Holder did not make a valid mark-to-market election for the first taxable year of the U.S. Holder in which the U.S. Holder held (or was deemed to hold) our Class A Ordinary Shares in which we were treated as a PFIC and if we were not a “pedigreed QEF” (as defined in Treasury regulations) during a U.S. Holder’s entire holding period for the Class A Ordinary Shares prior to such U.S. Holder’s mark-to-market election with respect to our Class A Ordinary Shares, U.S. Treasury regulations provide that the excess distribution regime applies to any distributions and dispositions occurring in the first year in which the mark-to-market election is made, as well as to the excess of the value of the Class A Ordinary Shares over its basis as of the effective date of the election. The U.S. Holder's basis in our Class A Ordinary Shares would then be increased by these inclusions. U.S. Holders are strongly urged to consult with and rely solely upon their tax advisors regarding the application of the rules governing purging elections to their particular circumstances.
The mark-to-market election is available only for “marketable stock,” generally, stock that is regularly traded on a national securities exchange that is registered with the SEC, including Nasdaq (on which the Class A Ordinary Shares are listed), or on a foreign exchange or market that the IRS determines has rules sufficient to ensure that the market price represents a legitimate and sound fair market value. Moreover, a mark-to-market election made with respect to Class A Ordinary Shares would not apply to a U.S. Holder’s indirect interest in any lower tier PFICs in which we own shares. U.S. Holders should consult their tax advisors regarding the availability and tax consequences of a mark-to-market election with respect to the Class A Ordinary Shares under their particular circumstances.
A U.S. Holder that owns (or is deemed to own) shares in a PFIC during any taxable year of the U.S. Holder may have to file an IRS Form 8621 and such other information as may be required by the U.S. Treasury Department. Failure to do so, if required, will extend the statute of limitations until such required information is furnished to the IRS.
The rules dealing with PFICs are very complex and are affected by various factors in addition to those described above. Accordingly, U.S. Holders of the Class A Ordinary Shares or Warrants should consult their tax advisors concerning the application of the PFIC rules to our Class A Ordinary Shares or Warrants under their particular circumstances.
Cayman Islands Tax Considerations
Prospective investors should consult their professional advisers on the possible tax consequences of buying, holding or selling shares under the laws of their country of citizenship, residence or domicile.
100
The following is a discussion on certain Cayman Islands income tax consequences of an investment in our securities. The discussion is a general summary of present law, which is subject to prospective and retroactive change. It is not intended as tax advice, does not consider any investor’s particular circumstances, and does not consider tax consequences other than those arising under Cayman Islands law.
Under Existing Cayman Islands Laws:
Payments of dividends and capital in respect of our securities will not be subject to taxation in the Cayman Islands and no withholding will be required on the payment of a dividend or capital to any holder of Class A Ordinary Shares, as the case may be, nor will gains derived from the disposal of the Class A Ordinary Shares be subject to Cayman Islands income or corporation tax. The Cayman Islands currently have no income, corporation or capital gains tax and no estate duty, inheritance tax or gift tax.
No stamp duty is payable in respect of the issue of our securities or on an instrument of transfer in respect of a Class A Ordinary Share or a Warrant.
We have been incorporated under the laws of the Cayman Islands as an exempted company with limited liability and, as such, have obtained undertakings from the Governor in Cabinet of the Cayman Islands in the following form:
The Tax Concessions Law
Undertaking as to Tax Concessions
In accordance with the Tax Concessions Act (as amended) of the Cayman Islands, the Governor in Cabinet of the Cayman Islands has undertaken with the Company:
(a)no law which is thereafter enacted in the Cayman Islands imposing any tax to be levied on profits, income, gains or appreciations shall apply to the Company or its operations; and
(b)in addition, that no tax to be levied on profits, income, gains or appreciations or which is in the nature of estate duty or inheritance tax shall be payable:
(i)on or in respect of the shares, debentures or other obligations of the Company; or
(ii)by way of the withholding in whole or in part of any relevant payment as defined in the Tax Concessions Act (as amended).
The concessions apply for a period of TWENTY years from September 21, 2021.
The Cayman Islands currently levy no taxes on individuals or corporations based upon profits, income, gains or appreciations and there is no taxation in the nature of inheritance tax or estate duty. There are no other taxes likely to be material to the Company levied by the Government of the Cayman Islands save certain stamp duties which may be applicable, from time to time, on certain instruments executed in or brought within the jurisdiction of the Cayman Islands.
F.Dividends and Paying Agents
Not applicable.
G.Statement by Experts
Not applicable.
H.Documents on Display
We are subject to the periodic reporting and other informational filing requirements of the Exchange Act, and are required to file reports and other information with the SEC. Specifically, we are required to file annually a Form 20-F within four months after the end of each fiscal year, which is December 31. Copies of reports and other information, when so filed, may be inspected without charge and may be obtained at prescribed rates at the public reference facilities maintained by the SEC at 100 F Street, N.E., Washington, D.C. 20549. The public may obtain information regarding the Washington, D.C. Public Reference Room by calling the SEC at 1-800-SEC-0330. The SEC also maintains a website at www.sec.gov that contains reports, proxy and information statements, and other information regarding registrants that make electronic filings with the SEC using its EDGAR system. As a foreign private issuer, we are exempt from the rules
101
under the Exchange Act prescribing the furnishing and content of quarterly reports and proxy statements, and officers, directors and principal shareholders are exempt from certain of the reporting and short-swing profit recovery provisions contained in Section 16 of the Exchange Act. Section 8103 of the National Defense Authorization Act for Fiscal Year 2026, named the “Holding Foreign Insiders Accountable Act”, which was signed into law on December 18, 2025, will require directors and officers of foreign private issuers to make insider reports under Section 16(a) of the Exchange Act, effective March 18, 2026. Our principal shareholders continue to remain exempt from the reporting under Section 16(a) of the Exchange Act and our directors, officers and principal shareholders continue to remain exempt from the short-swing profit recovery provisions contained in Section 16(b) of the Exchange Act.
In accordance with Nasdaq Stock Market Rule 5250(d), we will post this annual report on our website at https://ir.prenetics.com. In addition, we will provide hardcopies of our annual report free of charge to shareholders upon request.
I.Subsidiary Information
Not applicable.
J.Annual Report to Security Holders
Not applicable.
ITEM 11. QUANTITATIVE AND QUALITATIVE DISCLOSURES ABOUT MARKET RISK
We are exposed to credit risk, liquidity risk and foreign currency risk in the ordinary course of our business. For more information about financial and market risks that we are exposed to, see Note 31 to our audited consolidated financial statements included elsewhere in this annual report.
Credit Risk
Credit risk refers to the risk of financial loss to us if a customer or counterparty to a financial instrument fails to meet its contractual obligations. Our credit risk arises principally from our trade receivables and other financial assets. The carrying amounts of financial assets represent our maximum exposure to credit risk.
Our exposure to credit risk arising from cash and cash equivalents is limited because the counterparties are banks and financial institutions with good credit rating for which we consider to have low credit risk. Our exposure to credit risk arising from trade receivables is influenced mainly by the individual characteristics of each counterparty. As of December 31, 2025, 37% and 81% of the total trade receivables were due from our largest counterparty and our five largest counterparties, respectively. We limit our credit risk arising from trade receivables by performing individual credit evaluations on all customers requiring credit over a certain amount, which take into account the customer’s past payment history, financial position and other factors.
Liquidity Risk
We manage our liquidity risk by regularly monitoring our liquidity requirements to ensure that we maintain sufficient reserves of cash to meet our liquidity requirements in the short and longer term.
Foreign Currency Risk
We are exposed to foreign currency risk primarily from transactions denominated in currencies other than the functional currencies of our subsidiaries. The functional currencies of our subsidiaries are primarily the U.S. dollar (“USD”) and Hong Kong dollar (“HKD”). Foreign currency exposure arises when sales, purchases and receivables are denominated in currencies that differ from the functional currency of the respective Group entities. As HKD is pegged to USD, we consider the risk of movements in exchange rates between HKD and USD to be insignificant. We currently do not engage in hedging activities with respect to foreign currency transactions. Although our overall exposure to foreign exchange risk is limited, fluctuations in exchange rates may affect the reporting of our results of operations and financial position as our consolidated financial statements are presented in USD.
102
ITEM 12. DESCRIPTION OF SECURITIES OTHER THAN EQUITY SECURITIES
A.Debt Securities
Not applicable.
B.Warrants and Rights
See “Warrants and Rights” in Exhibit 2.11 to this annual report.
C.Other Securities
Not applicable.
D.American Depositary Shares
Not applicable.
PART II
ITEM 13. DEFAULTS, DIVIDEND ARREARAGES AND DELINQUENCIES
None.
ITEM 14. MATERIAL MODIFICATIONS TO THE RIGHTS OF SECURITY HOLDERS AND USE OF PROCEEDS
None.
ITEM 15. CONTROLS AND PROCEDURES
Disclosure Controls and Procedures
Our management, with the participation of our chief executive officer and chief financial officer, has performed an evaluation of the effectiveness of our disclosure controls and procedures (as defined in Rule 13a-15(e) under the Exchange Act) as of the end of the period covered by this report, as required by Rule 13a-15(b) under the Exchange Act.
Based upon that evaluation, our management, with the participation of our chief executive officer and chief financial officer, has concluded that, as of December 31, 2025, our disclosure controls and procedures were effective in ensuring that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is recorded, processed, summarized and reported, within the time periods specified in the SEC’s rules and forms, and that the information required to be disclosed by us in the reports that we file or submit under the Exchange Act is accumulated and communicated to our management, including our chief executive officer and chief financial officer, as appropriate, to allow timely decisions regarding required disclosure.
Management’s Annual Report on Internal Control over Financial Reporting and Attestation Report of the Registered Public Accounting Firm
Our management is responsible for establishing and maintaining adequate internal control over financial reporting (as defined in Rules 13a-15(f) and 15d(f) under the Exchange Act). Internal control over financial reporting is a process designed to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external reporting purposes in accordance with international financial reporting standards (IFRS Accounting Standards). Because of its inherent limitations, internal control over financial reporting may not prevent or detect misstatements. Also, projections of any evaluation of effectiveness to future periods are subject to the risk that controls may become inadequate because of changes in conditions, or that the degree of compliance with the policies or procedures may deteriorate. A material weakness is a deficiency, or a combination of deficiencies, in internal control over financial reporting, such that there is a reasonable possibility that a material misstatement of the company’s annual or interim financial statements will not be prevented or detected on a timely basis.
103
The Company conducted an assessment of the effectiveness of its internal control over financial reporting as of December 31, 2025 based on the criteria established in Internal Control-Integrated Framework issued by the Committee of Sponsoring Organizations of the Treadway Commission (2013 framework). Based on such assessment, our management concluded that our internal control over financial reporting was effective as of December 31, 2025.
This annual report does not include an attestation report of the Company’s registered public accounting firm due to a transition period established by rules of the Securities and Exchange Commission for emerging growth companies.
Changes in Internal Control over Financial Reporting
There were no changes in our internal controls over financial reporting that occurred during the period covered by this annual report that have materially affected, or are reasonably likely to materially affect, our internal control over financial reporting.
ITEM 16. [Reserved]
ITEM 16A. AUDIT COMMITTEE FINANCIAL EXPERT
Our board of directors has determined that Cheng Yin Pan (Ben), an independent director (within the meaning of the Nasdaq listing rules and the criteria for independence set forth in Rule 10A-3 of the Exchange Act) satisfies the criteria of an audit committee financial expert as set forth under the applicable rules of the SEC.
ITEM 16B. CODE OF ETHICS
Our board of directors adopted a Code of Business Conduct and Ethics applicable to our directors, officers and employees. The code applies to all directors, officers, employees and extended workforce, including the Chairperson and Chief Executive Officer and Chief Financial Officer. A copy of our Code of Business Conduct and Ethics is available on our website at https://ir.prenetics.com/corporate-governance/documents-charters.
ITEM 16C. PRINCIPAL ACCOUNTANT FEES AND SERVICES
Our independent registered public accounting firm is Deloitte Touche Tohmatsu (PCAOB ID: 1104), which is located in Hong Kong.
The following table sets forth the aggregate fees by categories specified below in connection with certain professional services rendered by Deloitte Touche Tohmatsu (PCAOB ID: 1104) and KPMG (PCAOB ID: 1181), our principal external auditors, for the years indicated. KPMG served as our independent auditor in 2023, and Deloitte Touche Tohmatsu served as our independent auditor since September 13, 2024. We did not pay any other fees to our auditors during the periods indicated below.
| For the Year Ended December 31, | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
(in thousands of U.S. dollars) | |||||||||||||||||
Audit fees(1) | $ | 1,085 | $ | 1,244 | $ | 1,324 | |||||||||||
Audit-related fees(2) | 124 | — | — | ||||||||||||||
Tax fees(3) | 17 | 30 | 4 | ||||||||||||||
| All other fees | — | — | — | ||||||||||||||
Notes:
(1)“Audit fees” represent the audit work performed each fiscal year necessary to allow the auditor to issue an opinion on our financial statements and to issue an opinion on the local statutory financial statements. Audit fees also include services such as reviews of quarterly financial results and limited review procedures of quarterly financial results.
(2)“Audit-related fees” represent fees for assurance and related services that were reasonably related to the performance of the audit or review of our financial statements or for services that were traditionally performed by the external auditor.
104
(3)“Tax fees” represent fees billed for professional services for tax compliance and tax advice.
The policy of our audit committee is to pre-approve all audit and other service provided by Deloitte Touche Tohmatsu and KPMG as described above, other than those for de minimis services which are approved by the Audit Committee prior to the completion of the audit.
ITEM 16D. EXEMPTIONS FROM THE LISTING STANDARDS FOR AUDIT COMMITTEES
Not applicable.
ITEM 16E. PURCHASES OF EQUITY SECURITIES BY THE ISSUER AND AFFILIATED PURCHASERS
On March 6, 2026, the Company's board of directors authorized a share repurchase program under which the Company may repurchase up to US$40 million of its outstanding Class A Ordinary Shares over the following 12 months. Repurchases under the program may be made from time to time in open market transactions, privately negotiated transactions, block trades or otherwise, in accordance with applicable securities laws and regulations. The timing, manner, price and amount of any repurchases are determined at the discretion of the Company's management based on a variety of factors, including market conditions, share price, trading volume, general business and economic conditions, and applicable legal and regulatory requirements. The program does not obligate the Company to repurchase any specific number of shares and may be suspended, modified or discontinued at any time without prior notice.
In connection with the share repurchase program, the Company entered into a written trading plan with B. Riley Securities, Inc. under Rule 10b5-1 of the Securities Exchange Act of 1934, as amended, to facilitate repurchases of Class A Ordinary Shares under the program during periods in which the Company might otherwise be restricted from purchasing shares under applicable insider trading laws or its own trading policies.
105
The table below is a summary of the shares repurchased by us for the periods indicated. All shares were repurchased in the open market pursuant to the Rule 10b5-1 written trading plan entered into in connection with the share repurchase program announced on March 6, 2026.
| Total Number of Ordinary Shares Purchased | Average Price Paid Per Ordinary Share (US$)6 | Total Cumulative Number of Ordinary Shares Purchased as Part of the Publicly Announced Program | Approximate Dollar Value of Ordinary Shares that May Yet Be Purchased Under the Program (US$, in millions) | ||||||||||||||||||||
| Period | |||||||||||||||||||||||
| January 1, 2025 – January 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| February 1, 2025 – February 28, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| March 1, 2025 – March 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| April 1, 2025 – April 30, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| May 1, 2025 - May 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| June 1, 2025 - June 30, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| July 1, 2025 - July 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| August 1, 2025 - August 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| September 1, 2025 - September 30, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| October 1, 2025 - October 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| November 1, 2025 - November 30, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| December 1, 2025 - December 31, 2025 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| January 1, 2026 - January 31, 2026 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| February 1, 2026 – February, 2026 | 0 | N/A | 0 | N/A | |||||||||||||||||||
| March 1, 2026 - March 31, 2026 | 648,534 | 20.74 | 648,534 | 26.72 | |||||||||||||||||||
April 1, 2026 - April 29, 2026 | 211,867 | 18.53 | 860,401 | 22.80 | |||||||||||||||||||
| Total | 860,401 | 19.58 | 860,401 | 22.80 | |||||||||||||||||||
ITEM 16F. CHANGE IN REGISTRANT’S CERTIFYING ACCOUNTANT
The information required by this Item 16F was previously reported in Item 16F of our Annual Report on Form 20-F for the fiscal year ended December 31, 2024, filed with the Securities and Exchange Commission on April 30, 2025.
ITEM 16G. CORPORATE GOVERNANCE
We are a foreign private issuer and a “controlled company” as defined under the Nasdaq rules. Mr. Yeung, chairman of our board of directors and our chief executive officer, owns more than 50% of the total voting power of all issued and outstanding ordinary shares. For so long as we remain a foreign private issuer or a “controlled company” under that definition, we are permitted to elect to rely, and may rely, on certain exemptions from certain corporate governance rules, including: an exemption from the rule that a majority of the board of directors must be independent directors; an exemption from the rule that director nominees must be selected or recommended solely by independent directors or by a nominations committee that is comprised entirely of independent directors; an exemption from the rule that our board of directors must have a compensation committee that is comprised solely of independent directors; an exemption from the requirement that an audit committee be comprised of at least three members; an exemption from the requirement that an annual general meeting must be held; an exemption from the requirement that we must obtain shareholder approval prior to a plan or other equity compensation arrangement is established or materially amended; and an exemption from the requirement to obtain shareholder approval for issuing additional securities exceeding 20% of our outstanding ordinary shares.
6 The average price paid per share, net of brokerage commissions and other costs of execution.
106
We intend to rely on some or all of the exemptions listed above available to foreign private issuers and “controlled company.” As a result, you will not have the same protection afforded to shareholders of companies that are subject to all of the Nasdaq corporate governance requirements. See also “Item 10. Additional Information.”
ITEM 16H. MINE SAFETY DISCLOSURE
Not applicable.
ITEM 16I. DISCLOSURE REGARDING FOREIGN JURISDICTIONS THAT PREVENT INSPECTIONS.
Not applicable.
ITEM 16J. INSIDER TRADING POLICIES.
We have adopted insider trading policies and procedures governing the purchase, sale, and other dispositions of our securities by directors, senior management, and employees that are reasonably designed to promote compliance with applicable insider trading laws, rules and regulations, and any listing standards applicable to us. A copy of our insider trading policies is filed as Exhibit 11.1 to this annual report on Form 20-F.
ITEM 16K. CYBERSECURITY.
Cybersecurity Risk Management and Strategy
We recognize the importance of assessing, identifying, and managing material risks associated with cybersecurity threats, as such term is defined in Item 106(a) of Regulation S-K. These risks include, among other things: operational risks, intellectual property theft, fraud, extortion, harm to our employees, customers or third-party vendors and service providers and violation of data privacy or security laws.
We have implemented a multi-layered cybersecurity approach which includes physical, technical and administrative measures to protect our systems against cybersecurity incidents. Our measures for assessing, identifying and managing material risks from cybersecurity threats and security incidents include:
•Our information is encrypted and securely stored in the cloud following best practices such as OWASP Top 10 and the Amazon’s Well Architected framework, and adhering to internationally recognized security and privacy standards such as ISO 27001.
•We conduct periodic internal and external assessments, such as penetration testing and vulnerability scans.
•We implement system safeguards, including email filtering and access control.
•We ensure continuous threat surveillance and have incident response plans in place for prompt identification, reporting, and resolution.
•We provide cybersecurity and privacy training to our employees.
•We regularly review our internal cybersecurity policies.
We have also implemented incident response and breach management policies and procedures. Such incident response processes are overseen by leaders from our Information Security, People Operations, Compliance and Legal teams. As part of these processes, we engage external consultants to assess our internal cybersecurity programs and compliance with applicable practices and standards.
107
Cybersecurity Governance
Cybersecurity is an important part of our risk management processes and an area of focus for our board of directors and management. Our Audit Committee is responsible for the oversight of risks from cybersecurity threats and responses to incidents, should they arise. Members of the Audit Committee receive updates as necessary on a quarterly basis regarding matters of cybersecurity or when there is cybersecurity incident. The internal auditor communicates this information to the Audit Committee. This includes existing and new cybersecurity risks, status on how management is addressing and/or mitigating those risks, cybersecurity and data privacy incidents (if any) and status on key information security initiatives.
Our Head of Technology Operations maintains our security incident response plan and manages day-to-day incident identification, assessment and management, leads our overall cybersecurity risk management program, including ongoing assessments of system vulnerabilities and mitigation efforts. Our Head of Technology Operations has been involved in cybersecurity for approximately 10 years, with project experience relating to SOC-2, ISO 27001, GDPR, Business Continuity Planning, Disaster Recovery Planning, Incident Response Planning. Our Head of Technology Operations escalates cybersecurity incidents to other members of the Company’s leadership as appropriate, including our CFO, CEO and internal auditor. The IT and engineering team provides regular briefings to the Head of Technology Operations regarding the Company’s cybersecurity risks and activities, including any recent cybersecurity incidents and related responses, cybersecurity systems testing, activities of third parties, and the like.
108
PART III.
ITEM 17. FINANCIAL STATEMENTS
We have elected to provide consolidated financial statements pursuant to Item 18.
ITEM 18. FINANCIAL STATEMENTS
The consolidated financial statements of Prenetics Global Limited are included at the end of this annual report.
ITEM 19. EXHIBITS
| Exhibit Number | Description | |||||||
1.1* | ||||||||
| 2.1 | ||||||||
| 2.2 | Specimen Warrant Certificate of Prenetics Global Limited (incorporated by reference to Exhibit 4.2 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 2.3 | Warrant Agreement, dated May 13, 2021, between Artisan and Continental Stock Transfer & Trust Company (incorporated by reference to Exhibit 4.3 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 2.4 | Assignment, Assumption and Amendment Agreement, dated as of September 15, 2021, by and among Prenetics Global Limited, Artisan Acquisition Corp. and Continental Stock Transfer & Trust Company (incorporated by reference to Exhibit 10.8 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 2.5 | Warrant Agency Agreement, dated October 28, 2025, by and between Prenetics Global Limited and Continental Stock Transfer and Trust Company (relating to Class A Warrants, Class B Warrants and Placement Agent Warrants) (incorporated by reference to Exhibit 4.1 to the Report on Form 6-K (File No. 001-41401), filed with the SEC on October 28, 2025) | |||||||
| 2.6 | Form of Class A Warrant (incorporated by reference to Exhibit 4.2 to the Report on Form 6-K (File No. 001-41401), filed with the SEC on October 28, 2025) | |||||||
| 2.7 | Form of Class B Warrant (incorporated by reference to Exhibit 4.3 to the Report on Form 6-K (File No. 001-41401), filed with the SEC on October 28, 2025) | |||||||
| 2.8 | Form of Placement Agent Warrant (incorporated by reference to Exhibit 4.4 to the Report on Form 6-K (File No. 001-41401), filed with the SEC on October 28, 2025) | |||||||
| 2.9 | Form of Warrant Agency Agreement by and between Prenetics Global Limited and Continental Stock Transfer and Trust Company (relating to Class C Warrants) (incorporated by reference to Exhibit 4.1 to the Registration Statement on Form F-3 (File No. 333-294765), filed on March 31, 2026) | |||||||
| 2.10 | Form of Class C Warrant (incorporated by reference to Exhibit 4.2 to the Report on Form 6-K (File No. 001-41401), filed with the SEC on December 23, 2025) | |||||||
2.11* | ||||||||
| 4.1 | Business Combination Agreement, dated as of September 15, 2021, by and among Artisan Acquisition Corp., Prenetics Global Limited, Prenetics Group Limited, AAC Merger Limited, and PGL Merger Limited (incorporated by reference to Exhibit 2.1 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
109
| Exhibit Number | Description | |||||||
| 4.2 | Amendment to Business Combination Agreement, dated as of March 30, 2022, by and among Artisan Acquisition Corp., Prenetics Global Limited, Prenetics Group Limited, AAC Merger Limited, and PGL Merger Limited (incorporated by reference to Exhibit 2.2 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.3 | Registration Rights Agreement, dated as of September 15, 2021, by and among Prenetics Global Limited, Artisan Acquisition Corp., Artisan LLC and other parties named therein (incorporated by reference to Exhibit 10.5 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.4† | 2022 Share Incentive Plan (incorporated by reference to Exhibit 4.4 to Shell Company Report on Form 20-F (Reg. No. 001-41401), filed with the SEC on May 27, 2022) | |||||||
| 4.5 | Form of Indemnification Agreement between Prenetics Global Limited and each executive officer of Prenetics Global Limited (incorporated by reference to Exhibit 10.10 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.6 | Form of PIPE Subscription Agreements (incorporated by reference to Exhibit 10.1 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.7 | Deed of Novation and Amendment, dated as of September 15, 2021, by and among Artisan Acquisition Corp., Prenetics Global Limited, Artisan LLC and Aspex Master Fund (incorporated by reference to Exhibit 10.2 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.8 | Deed of Novation and Amendment, dated as of September 15, 2021, by and among Artisan Acquisition Corp., Prenetics Global Limited, Artisan LLC and Pacific Alliance Asia Opportunity Fund L.P. (incorporated by reference to Exhibit 10.3 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.9 | Shareholder Support Agreement and Deed, dated as of September 15, 2021, by and among Prenetics Global Limited, Prenetics Group Limited, Artisan Acquisition Corp., and certain management shareholders named therein (incorporated by reference to Exhibit 10.6 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.10 | Shareholder Support Agreement and Deed, dated as of September 15, 2021, by and among Prenetics Global Limited, Prenetics Group Limited, Artisan Acquisition Corp., and certain shareholders named therein (incorporated by reference to Exhibit 10.7 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
| 4.11 | Sponsor Support Agreement, dated as of September 15, 2021, by and among Prenetics Global Limited, Prenetics Group Limited, Artisan Acquisition Corp., Artisan LLC and other parties named therein (incorporated by reference to Exhibit 10.4 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
4.12# | Agreement for Sale and Purchase of the Issued Shares in ACT Genomics Holdings Company Limited, dated December 16, 2022, by and among Prenetics Global Limited, ACT Genomics and certain shareholders of ACT Genomics (incorporated by reference to Exhibit 4.14 of our annual report on Form 20-F (Reg. No. 001-41401), filed with the SEC on April 30, 2025) | |||||||
4.13# | Agreement for Sale and Purchase of the Issued Shares in ACT Genomics Holdings Company Limited, dated January 3, 2023, by and among Prenetics Global Limited, Hongkong Berry Genomics Co., Limited and ACT Genomics (incorporated by reference to Exhibit 4.14 of our annual report on Form 20-F (Reg. No. 001-41401), filed with the SEC on April 30, 2025) | |||||||
| 4.14 | Deed of Joinder, dated October 1, 2021, by and among Prenetics Global Limited, Prenetics Group Limited, Artisan Acquisition Corp. and Prudential Hong Kong Limited (incorporated by reference to Exhibit 10.18 to Amendment No. 8 to the Registration Statement on Form F-4 (Reg. No. 333-260928), filed with the SEC on March 30, 2022) | |||||||
110
111
| Exhibit Number | Description | |||||||
4.28*# | ||||||||
4.29*# | ||||||||
4.30*# | ||||||||
4.31*# | ||||||||
4.32*# | ||||||||
4.33*# | ||||||||
| 8.1* | ||||||||
| 11.1* | ||||||||
| 12.1* | ||||||||
| 12.2* | ||||||||
| 13.1** | ||||||||
| 13.2** | ||||||||
| 15.1* | ||||||||
| 15.2* | ||||||||
| 15.3* | ||||||||
| 97.10 | Policy for Recovery of Erroneously Awarded Compensation (incorporated by reference to Exhibit 97.1 of our annual report on Form 20-F (Reg. No. 001-41401), filed with the SEC on May 1, 2024) | |||||||
| 101.INS* | Inline XBRL Instance Document—this instance document does not appear on the Interactive Data File because its XBRL tags are not embedded within the Inline XBRL document | |||||||
| 101.SCH* | Inline XBRL Taxonomy Extension Scheme Document | |||||||
| 101.CAL* | Inline XBRL Taxonomy Extension Calculation Linkbase Document | |||||||
| 101.DEF* | Inline XBRL Taxonomy Extension Definition Linkbase Document | |||||||
| 101.LAB* | Inline XBRL Taxonomy Extension Label Linkbase Document | |||||||
| 101.PRE* | Inline XBRL Taxonomy Extension Presentation Linkbase Document | |||||||
| 104 | Cover Page Interactive Data File (embedded within the Inline XBRL document) | |||||||
_______________
*Filed with this Annual Report on Form 20-F.
**Furnished with this Annual Report on Form 20-F.
†Indicates a management contract or any compensatory plan, contract or arrangement.
112
# Portions of this exhibit have been omitted pursuant to Item 601(b)(10)(iv) of Regulation S-K on the basis that the Company customarily and actually treats that information as private or confidential and the omitted information is not material.
113
SIGNATURES
The registrant hereby certifies that it meets all of the requirements for filing on Form 20-F and that it has duly caused and authorized the undersigned to sign this annual report on its behalf.
| Prenetics Global Limited | ||||||||
| By: | /s/ Danny Sheng Wu Yeung | |||||||
| Name: | Danny Sheng Wu Yeung | |||||||
| Title: | Chief Executive Officer | |||||||
Date: April 30, 2026
114
Prenetics Global Limited
(Incorporated in the Cayman Islands)
and its Subsidiaries
Annual Report
Index
Report of Independent Registered Public Accounting Firm ( | F-2 | ||||
F-3 | |||||
F-4 | |||||
F-5 | |||||
F-6 | |||||
F-8 | |||||
F-10 | |||||
F-1
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the shareholders and the Board of Directors of Prenetics Global Limited:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated statements of financial position of Prenetics Global Limited and subsidiaries (the "Company") as of December 31, 2025 and 2024, the related consolidated statements of profit or loss and other comprehensive income, changes in equity, and cash flows, for each of the two years then ended December 31, 2025 and 2024, and the related notes (collectively referred to as the "financial statements"). In our opinion, the financial statements present fairly, in all material respects, the financial position of the Company as of December 31, 2025 and 2024, and the results of its operations and its cash flows for each of the two years ended December 31, 2025 and 2024, in conformity with IFRS Accounting Standards as issued by the International Accounting Standards Board ("IASB").
Basis for Opinion
These financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on the Company’s financial statements based on our audits. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audits in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audits to obtain reasonable assurance about whether the financial statements are free of material misstatement, whether due to error or fraud. The Company is not required to have, nor were we engaged to perform, an audit of its internal control over financial reporting. As part of our audits, we are required to obtain an understanding of internal control over financial reporting but not for the purpose of expressing an opinion on the effectiveness of the Company’s internal control over financial reporting. Accordingly, we express no such opinion.
Our audits included performing procedures to assess the risks of material misstatement of the financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the financial statements. Our audits also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the financial statements. We believe that our audits provide a reasonable basis for our opinions.
/s/ Deloitte Touche Tohmatsu
Hong Kong SAR, the People's Republic of China
April 30, 2026
We have served as the Company’s auditor since 2024.
F-2
REPORT OF INDEPENDENT REGISTERED PUBLIC ACCOUNTING FIRM
To the Shareholders and Board of Directors
Prenetics Global Limited:
Opinion on the Consolidated Financial Statements
We have audited the accompanying consolidated statements of profit or loss and other comprehensive income, changes in equity and cash flows of Prenetics Global Limited and subsidiaries (the Company) for the year ended December 31, 2023, and the related notes (collectively, the consolidated financial statements). In our opinion, the consolidated financial statements present fairly, in all material respects, the results of operations of the Company and its cash flows for the year ended December 31, 2023, in conformity with IFRS Accounting Standards as issued by the International Accounting Standards Board.
Basis for Opinion
These consolidated financial statements are the responsibility of the Company’s management. Our responsibility is to express an opinion on these consolidated financial statements based on our audit. We are a public accounting firm registered with the Public Company Accounting Oversight Board (United States) (PCAOB) and are required to be independent with respect to the Company in accordance with the U.S. federal securities laws and the applicable rules and regulations of the Securities and Exchange Commission and the PCAOB.
We conducted our audit in accordance with the standards of the PCAOB. Those standards require that we plan and perform the audit to obtain reasonable assurance about whether the consolidated financial statements are free of material misstatement, whether due to error or fraud.
Our audit included performing procedures to assess the risks of material misstatement of the consolidated financial statements, whether due to error or fraud, and performing procedures that respond to those risks. Such procedures included examining, on a test basis, evidence regarding the amounts and disclosures in the consolidated financial statements. Our audit also included evaluating the accounting principles used and significant estimates made by management, as well as evaluating the overall presentation of the consolidated financial statements. We believe that our audit provide a reasonable basis for our opinion.
/s/ KPMG
We served as the Company’s auditor from 2017 to 2024.
Hong Kong, China
April 30, 2024, except for the effects of discontinued operations of ACT Genomics discussed in note 10, as to which the date is April 30, 2026
F-3
Consolidated statements of profit or loss and other comprehensive income for each of the three years in the period ended December 31, 2025
(All amounts in thousands of U.S. dollars ("$"), except for share and per share data)
| Notes | 2025 | 2024 | 2023 | ||||||||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||||||||
| Continuing operations | |||||||||||||||||||||||
| Revenue | 5, 6 | $ | $ | $ | |||||||||||||||||||
| Direct costs | ( | ( | ( | ||||||||||||||||||||
| Gross profit | |||||||||||||||||||||||
| Other income and other net gains | 7 | ||||||||||||||||||||||
| Selling and marketing expenses | ( | ( | ( | ||||||||||||||||||||
| Research and development expenses | ( | ( | ( | ||||||||||||||||||||
| Impairment of goodwill | 12 | ( | |||||||||||||||||||||
| Administrative and other operating expenses | ( | ( | ( | ||||||||||||||||||||
| Operating loss from continuing operations | ( | ( | ( | ||||||||||||||||||||
| Fair value gain/(loss) on financial assets at fair value through profit or loss | 20 | ( | ( | ||||||||||||||||||||
| Gain on warrant exchange | 27 | ||||||||||||||||||||||
| Fair value (loss)/gain on warrant liabilities | 27 | ( | |||||||||||||||||||||
| Unrealized fair value loss on digital assets | 15 | ( | |||||||||||||||||||||
| Gain on partial disposal of an equity-accounted investee | 16 | ||||||||||||||||||||||
| Share of loss of equity-accounted investees, net of tax | 16 | ( | ( | ( | |||||||||||||||||||
| Other finance costs | 8(a) | ( | ( | ( | |||||||||||||||||||
| Loss before taxation | 8 | ( | ( | ( | |||||||||||||||||||
| Income tax (expense)/credit | 9 | ( | ( | ||||||||||||||||||||
| Loss from continuing operations | ( | ( | ( | ||||||||||||||||||||
| Discontinued operations | |||||||||||||||||||||||
| Loss from discontinued operations, net of tax | 10 | ( | ( | ( | |||||||||||||||||||
| Loss for the year | ( | ( | ( | ||||||||||||||||||||
| Other comprehensive income/(expense) | |||||||||||||||||||||||
| Items that will not be reclassified subsequently to profit or loss: | |||||||||||||||||||||||
| Share of other comprehensive (expense)/income of equity-accounted investees | 16 | ( | |||||||||||||||||||||
| Item that may be reclassified subsequently to profit or loss: | |||||||||||||||||||||||
| Exchange differences on translation of foreign operations | ( | ||||||||||||||||||||||
| Reclassification of cumulative translation reserve upon disposal of foreign operations | 34 | ( | |||||||||||||||||||||
| Other comprehensive income/(expense) for the year | ( | ||||||||||||||||||||||
| Total comprehensive expense for the year | $ | ( | $ | ( | $ | ( | |||||||||||||||||
| Loss attributable to: | |||||||||||||||||||||||
| Equity shareholders of the Company | $ | ( | $ | ( | $ | ( | |||||||||||||||||
| Non-controlling interests | 35 | ( | ( | ( | |||||||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||||||||
| Total comprehensive expense attributable to: | |||||||||||||||||||||||
| Equity shareholders of the Company | $ | ( | $ | ( | $ | ( | |||||||||||||||||
| Non-controlling interests | 35 | ( | ( | ( | |||||||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||||||||
| Loss per share | |||||||||||||||||||||||
| Basic | 11 | $ | ( | $ | ( | $ | ( | ||||||||||||||||
| Diluted | 11 | ( | ( | ( | |||||||||||||||||||
| Loss per share - Continuing operations | |||||||||||||||||||||||
| Basic | 11 | ( | ( | ( | |||||||||||||||||||
| Diluted | 11 | ( | ( | ( | |||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-4
Consolidated statements of financial position as of December 31, 2025 and 2024
(All amounts in thousands of U.S. dollars ("$"))
| Notes | 2025 | 2024 | |||||||||||||||
| Assets | |||||||||||||||||
| Property, plant and equipment | 13 | $ | $ | ||||||||||||||
| Intangible assets | 14 | ||||||||||||||||
| Digital assets | 15 | ||||||||||||||||
| Goodwill | 12 | ||||||||||||||||
| Interests in equity-accounted investees | 16 | ||||||||||||||||
| Financial assets at fair value through profit or loss - non-current | 20 | ||||||||||||||||
| Other non-current assets | 17 | ||||||||||||||||
| Non-current assets | |||||||||||||||||
| Deferred expenses | 19 | ||||||||||||||||
| Inventories | 18 | ||||||||||||||||
| Trade receivables | 19 | ||||||||||||||||
| Deposits, prepayments and other receivables | 19 | ||||||||||||||||
| Amounts due from related companies | |||||||||||||||||
| Financial assets at fair value through profit or loss - current | 20 | ||||||||||||||||
| Cash and cash equivalents | 21(b) | ||||||||||||||||
| Current assets | |||||||||||||||||
| Total assets | $ | $ | |||||||||||||||
| Liabilities | |||||||||||||||||
| Deferred tax liabilities | 24 | $ | $ | ||||||||||||||
| Warrant liabilities | 27 | ||||||||||||||||
| Lease liabilities - non-current | 25 | ||||||||||||||||
| Other non-current liabilities | 22 | ||||||||||||||||
| Non-current liabilities | |||||||||||||||||
| Trade payables | |||||||||||||||||
| Accrued expenses and other current liabilities | 22 | ||||||||||||||||
| Contract liabilities | 23 | ||||||||||||||||
| Lease liabilities - current | 25 | ||||||||||||||||
| Liabilities for puttable financial instrument | 28 | ||||||||||||||||
| Tax payable | |||||||||||||||||
| Current liabilities | |||||||||||||||||
| Total liabilities | |||||||||||||||||
| Equity | |||||||||||||||||
Class A ordinary shares (par value of $ | 29 | ||||||||||||||||
Class B ordinary shares (par value of $ | 29 | ||||||||||||||||
| Reserves | |||||||||||||||||
| Total equity attributable to equity shareholders of the Company | |||||||||||||||||
| Non-controlling interests | ( | ||||||||||||||||
| Total equity | |||||||||||||||||
| Total equity and liabilities | $ | $ | |||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-5
Consolidated statements of changes in equity for each of the three years in the period ended December 31, 2025
(All amounts in thousands of U.S. dollars (“$”))
| Attributable to equity shareholders of the Company | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes | Share capital | Share premium | Treasury stock | Translation reserve | Other reserve | Capital reserve | Accumulated losses | Sub-total | Non- controlling interests | Total | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at January 1, 2023 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income/(expense) for the year | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the year | — | — | — | — | — | — | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income for the year | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income/(expense) for the year | — | — | — | — | — | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in fair value of liabilities for puttable financial instrument | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity-settled share-based transactions | 30 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adjustments of non-controlling interests | — | — | — | — | — | — | — | — | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for acquisition | — | — | — | ( | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for investment in an equity-accounted investee | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for restricted share unit | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Shares issued upon conversion of exchange loan notes | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for reverse stock split | — | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of shares | — | — | ( | — | — | — | — | ( | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cancellation of treasury stock | 29(b) | — | ( | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2023 and January 1, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive expense for the year | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the year | — | — | — | — | — | — | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive expense for the year | — | — | — | ( | — | — | — | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive expense for the year | — | — | — | ( | — | — | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in fair value of liabilities for puttable financial instrument | 28 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in non-controlling interest due to change in shareholding in a subsidiary | 28 | — | — | — | — | ( | — | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity-settled share-based transactions | 30 | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares to advisors | 29(a) | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for restricted share unit | 29(a) | — | — | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Repurchase of shares | 29(b) | — | — | ( | — | — | — | — | ( | — | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2024 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||||||||||||||
F-6
Consolidated statements of changes in equity for each of the three years in the period ended December 31, 2025 (continued)
(All amounts in thousands of U.S. dollars (“$”))
| Attributable to equity shareholders of the Company | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes | Share capital | Share premium | Treasury stock | Translation reserve | Other reserve | Capital reserve | Accumulated losses | Sub-total | Non- controlling interests | Total | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at January 1, 2025 | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income/(expense) for the year | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the year | — | — | — | — | — | — | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income for the year | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income/(expense) for the year | — | — | — | — | — | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Change in fair value and derecognition of liabilities for puttable financial instrument | 28 | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Derecognition of non-controlling interests upon disposal of a subsidiary | 34 | — | — | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity-settled share-based transactions | 30 | — | — | — | — | — | — | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of new shares and allocation of warrant | 27, 29(a) | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Issuance of shares for restricted share unit | 29(a) | — | — | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2025 | $ | $ | $ | ( | $ | ( | $ | ( | $ | $ | ( | $ | $ | ( | $ | ||||||||||||||||||||||||||||||||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-7
Consolidated statements of cash flows for each of the three years in the period ended December 31, 2025
(All amounts in thousands of U.S. dollars (“$”))
| Notes | 2025 | 2024 | 2023 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash flows from operating activities | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Loss for the year | $ | ( | $ | ( | $ | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||
| Adjustments for: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bank interest income | 7,10(b) | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Depreciation | 13 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amortization of intangible assets | 14 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other finance costs | 8(a),10(c) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value (gain)/loss on financial assets at fair value through profit or loss | 20 | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Fair value loss/(gain) on warrant liabilities | 27 | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gain on warrant exchange | 27 | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unrealized fair value loss on digital assets | 15 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net foreign exchange losses | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Impairment loss of goodwill | 12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Gain)/loss on disposal of property, plant and equipment | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Write-off on property, plant and equipment | 8(c),10(e) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Write-off on inventories | 18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gain on disposal of a subsidiary | 34 | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gain on partial disposal of an equity-accounted investee | 16 | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share of loss of equity-accounted investees, net of tax | 16 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Equity-settled share-based payment expenses | 30 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based payment expenses in relation to issuance of shares to advisors | 30(d) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income tax expense/(credit) | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Changes in: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Decrease/(increase) in deferred expenses | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Increase)/decrease in inventories | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Increase)/decrease in trade receivables | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Increase)/decrease in deposits, prepayments and other receivables | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Increase)/decrease in amounts due from related companies | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Decrease/(increase) in amounts due from equity-accounted investees | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Decrease/(increase) in other non-current assets | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increase/(decrease) in trade payables | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increase/(decrease) in accrued expenses and other current liabilities | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (Decrease)/increase in contract liabilities | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Increase/(decrease) in other non-current liabilities | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cash used in operating activities | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Income taxes paid | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Net cash used in operating activities | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||||||||||||||
F-8
Consolidated statements of cash flows for each of the three years in the period ended December 31, 2025 (continued)
(All amounts in thousands of U.S. dollars (“$”))
| Notes | 2025 | 2024 | 2023 | ||||||||||||||||||||
| Cash flows from investing activities | |||||||||||||||||||||||
| Payment for purchase of property, plant and equipment | ( | ( | ( | ||||||||||||||||||||
| Proceeds from disposal of property, plant and equipment | |||||||||||||||||||||||
| Payment for purchase of digital assets | ( | ||||||||||||||||||||||
| Payment for purchase of intangible assets | ( | ( | |||||||||||||||||||||
| Payment for purchase of short-term deposits | ( | ||||||||||||||||||||||
| Payment for purchase of financial assets at fair value through profit or loss | 20 | ( | ( | ( | |||||||||||||||||||
| Proceeds from partial disposal of an equity-accounted investee | |||||||||||||||||||||||
| Proceeds from redemption of short-term deposits | |||||||||||||||||||||||
| Net cash inflow from disposal of a subsidiary | 34 | ||||||||||||||||||||||
| Payment for acquisition of a subsidiary, net of cash acquired | 33 | ( | |||||||||||||||||||||
| Investment in an equity-accounted investee | 16 | ( | |||||||||||||||||||||
| Interest received | 7,10(b) | ||||||||||||||||||||||
| Net cash (used in)/from investing activities | ( | ( | |||||||||||||||||||||
| Cash flows from financing activities | |||||||||||||||||||||||
| Capital element of lease rentals paid | 13(b),26 | ( | ( | ( | |||||||||||||||||||
| Interest element of lease rentals paid | 13(b),26 | ( | ( | ( | |||||||||||||||||||
| Proceeds from issuance of shares | |||||||||||||||||||||||
| Proceeds from public placement | 27 | ||||||||||||||||||||||
| Payment for purchase of treasury shares | ( | ( | |||||||||||||||||||||
| Net cash from/(used in) financing activities | ( | ( | |||||||||||||||||||||
| Net (decrease)/increase in cash and cash equivalents | ( | ( | |||||||||||||||||||||
| Cash and cash equivalents at the beginning of the year | |||||||||||||||||||||||
| Effect of foreign exchange rate changes | ( | ||||||||||||||||||||||
| Cash and cash equivalents at the end of the year | $ | $ | $ | ||||||||||||||||||||
The accompanying notes are an integral part of these consolidated financial statements.
F-9
Notes to the consolidated financial statements
(All amounts in thousands of U.S. dollars (“US$”), except for share, per share data and otherwise stated)
1 General Information
Prenetics Global Limited (the “Company”), is a company limited by shares incorporated in Cayman Islands.
The Company and its subsidiaries (collectively, the “Group”) is a leading health sciences company redefining the future of health and longevity.
On July 20, 2023, the Group acquired 50 % shareholdings of an equity-accounted investee, Insighta Holdings Limited ("Insighta"). On October 14, 2024, the Group’s shareholding in Insighta was reduced to 35 % following the disposal of 15 % shareholdings with consideration of $30 million to a third party. Details of the interests in equity-accounted investees are included in note 16.
On August 9, 2024, the Group fully acquired the issued units of Europa Partners Holdings Limited (“Europa”), and obtained 100 % shareholdings in Europa. Europa is a consumer health and wellness products distributor, which specializing in sports distribution, and qualifies as a business as defined in IFRS 3 in note 33.
On October 1, 2025, the Group fully disposed 73.27 % of the issued share capital of ACT Genomics Holdings Company Limited (“ACT Genomics”) with considerations of $46 million from a third party. Accordingly, the operations of ACT Genomics have been classified as discontinued operations for all periods presented. The results of the discontinued operations for the years ended December 31, 2024 and 2023 have been restated in the consolidated statement of profit or loss to conform to the current period’s presentation. Details of the disposal of a subsidiary are included in note 34.
On October 28, 2025, the Group completed a public placement and issued equity securities to investors, raising net proceeds of approximately $40 million. In connection with the placement, the Group issued 2,722,642 Class A ordinary shares together with warrants, including 2,722,642 Class A warrants, 2,722,642 Class B warrants, and 131,829 placement agent warrants. On December 23, 2025, the Group entered into warrant exchange agreements with certain warrant holders, pursuant to which each pair of one Class A warrant and one Class B warrant was exchanged for one Class C warrant for Class A ordinary shares of the Company. The exchange resulted in a reduction in the number of outstanding Class A and Class B warrants and the issuance of new Class C warrants with modified terms. Details of the warrants are included in note 27.
These consolidated financial statements are presented in United States dollars (“USD”), which is the Company’s functional currency.
2 Adoption of new and revised standards
Amendments to IFRS Accounting Standards that are mandatorily effective for the current year
In the current year, the Group has applied the following amendments to IFRS Accounting Standards issued by the IASB for the first time, which are mandatorily effective for the Group's annual period beginning on January 1, 2025 for the preparation of the consolidated financial statements:
| Amendments to IAS 21 | Lack of Exchangeability | ||||
The application of the amendments to IFRS Accounting Standards in the current year has had no material impact on the Group's financial positions and performance for the current and prior years and/or on the disclosures set out in these consolidated financial statements.
F-10
2Adoption of new and revised standards (continued)
New and amendments to IFRS Accounting Standards in issue but not yet effective
At the date of authorization of these consolidated financial statements, the Group has not applied the following new and amended IFRS Accounting Standards that have been issued but are not yet effective that are relevant to the Group.
| Amendments to IFRS 9 and IFRS 7 | Amendments to the Classification and Measurement of Financial Instruments2 | ||||
| Amendments to IFRS 10 and IAS 28 | Sale or Contribution of Assets between an Investor and its Associate or Joint Venture1 | ||||
| Amendments to IFRS Accounting Standards | Annual Improvements to IFRS Accounting Standards - Volume 112 | ||||
| IFRS 18 | Presentation and Disclosure in Financial Statements3 | ||||
1 Effective for annual periods beginning on or after a date to be determined
2 Effective for annual periods beginning on or after January 1, 2026
3 Effective for annual periods beginning on or after January 1, 2027
Except for the new and amendments to IFRS Accounting Standards mentioned below, the directors of the Company anticipate that the application of all other new and amendments to IFRS Accounting Standards will have no material impact on the consolidated financial statements in the foreseeable future.
IFRS 18 Presentation and Disclosures in Financial Statements ("IFRS 18")
IFRS 18, which sets out requirements on presentation and disclosures in financial statements, will replace IAS 1 Presentation of Financial Statements ("IAS 1"). This new IFRS Accounting Standard, while carrying forward many of the requirements in IAS 1, introduces new requirements to present specified categories and defined subtotals in the statement of profit or loss; provide disclosures on management-defined performance measures ("MPMs) in the notes to the financial statements and improve aggregation and disaggregation of information to be disclosed in the financial statements. In addition, some IAS 1 paragraphs have been moved to IAS 8 Accounting Policies, Changes in Accounting Estimates and Errors (the title of which will be changed to Basis of Preparation of Financial Statements upon effective of IFRS 18) and IFRS 7. Minor amendments to IAS 7 Statement of Cash Flows and IAS 33 Earnings per Share are also made.
IFRS 18, and amendments to other standards, will be effective for annual periods beginning on or after January 1, 2027, with early application permitted. IFRS 18 requires retrospective application with specific transition provisions. The application of the new standard is not expected to have significant impact on the financial performance and positions of the Group in terms of recognition and measurement. However, it is expected to affect the structure and presentation of the consolidated statement of profit or loss.
F-11
3 Basis of preparation of consolidated financial statements and material accounting policy information
Basis of preparation of consolidated financial statements
The consolidated financial statements have been prepared in accordance with IFRS Accounting Standards as issued by the IASB. For the purpose of preparation of the consolidated financial statements, information is considered material if such information is reasonably expected to influence decisions made by primary users.
The material accounting policies adopted are set out below.
Going concern
The directors of the Company have, at the time of approving the consolidated financial statements, a reasonable expectation that the Group has adequate resources to continue in operational existence for the foreseeable future. Thus, they continue to adopt the going concern basis of accounting in preparing the consolidated financial statements.
Material accounting policy information
Basis of consolidation
The consolidated financial statements incorporate the financial statements of the Company and entities controlled by the Group. Control is achieved when the Company:
•has power over the investee;
•is exposed, or has rights, to variable returns from its involvement with the investee; and
•has the ability to use its power to affect its returns.
The Group reassesses whether or not it controls an investee if facts and circumstances indicate that there are changes to one or more of the three elements of control listed above.
When the Group has less than a majority of the voting rights of an investee, it considers that it has power over the investee when the voting rights are sufficient to give it the practical ability to direct the relevant activities of the investee unilaterally. The Group considers all relevant facts and circumstances in assessing whether or not the group’s voting rights in an investee are sufficient to give it power, including:
•the size of the group’s holding of voting rights relative to the size and dispersion of holdings of the other vote holders
•potential voting rights held by the group, other vote holders or other parties
•rights arising from other contractual arrangements
•any additional facts and circumstances that indicate that the group has, or does not have, the current ability to direct the relevant activities at the time that decisions need to be made, including voting patterns at previous shareholders’ meetings.
Consolidation of a subsidiary begins when the Group obtains control over the subsidiary and ceases when the Group loses control of the subsidiary. Specifically, the results of subsidiaries acquired or disposed of during the year are included in profit or loss from the date the Group gains control until the date when the Group ceases to control the subsidiary.
Profit or loss and each component of other comprehensive income are attributed to the owners of the Company and to the non-controlling interests. Total comprehensive income of the subsidiaries is attributed to the owners of the Company and to the non-controlling interests even if this results in the non-controlling interests having a deficit balance.
Where necessary, adjustments are made to the financial statements of subsidiaries to bring the accounting policies used into line with the Group’s accounting policies.
All intra-group assets and liabilities, equity, income, expenses and cash flows relating to transactions between members of the Group are eliminated in full on consolidation.
F-12
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Non-controlling interests in subsidiaries are presented separately from the Group’s equity therein, which represent present ownership interests entitling their holders to a proportionate share of net assets of net assets of relevant subsidiaries upon liquidation.
Changes in the Group's interests in existing subsidiaries
Changes in the Group’s interests in subsidiaries that do not result in the Group losing control over the subsidiaries are accounted for as equity transactions. The carrying amounts of the Group’s relevant components of equity and the non-controlling interests are adjusted to reflect the changes in their relative interests in the subsidiaries.
Any difference between the amount by which the non-controlling interests are adjusted, and the fair value of the consideration paid or received is recognized directly in equity and attributed to the owners of the Company.
When the group loses control of a subsidiary, the assets and liabilities of that subsidiary and non-controlling interests (if any) are derecognized. A gain or loss is recognized in profit or loss and is calculated as the difference between (i) the aggregate of the fair value of the consideration received and the fair value of any retained interest and (ii) the carrying amount of the assets (including goodwill), and liabilities of the subsidiary attributable to the owners of the Company. All amounts previously recognized in other comprehensive income in relation to that subsidiary are accounted for as if the Group had directly disposed of the related assets or liabilities of the subsidiary (i.e. reclassified to profit or loss or transferred to another category of equity as specified/permitted by applicable IFRS Accounting Standards). The fair value of any investment retained in the former subsidiary at the date when control is lost is regarded as the fair value on initial recognition for subsequent accounting under IFRS 9 Financial Instruments or, when applicable, the cost on initial recognition of an investment in an associate or a joint venture.
Business combinations
A business is an integrated set of activities and assets which includes an input and a substantive process that together significantly contribute to the ability to create outputs. The acquired processes are considered substantive if they are critical to the ability to continue producing outputs, including an organized workforce with the necessary skills, knowledge, or experience to perform the related processes or they significantly contribute to the ability to continue producing outputs and are considered unique or scarce or cannot be replaced without significant cost, effort, or delay in the ability to continue producing outputs.
Acquisitions of businesses are accounted for using the acquisition method. The consideration transferred in a business combination is measured at fair value, which is calculated as the sum of the acquisition-date fair values of assets transferred by the Group, liabilities incurred by the Group to the former owners of the acquiree and the equity interest issued by the Group in exchange for control of the acquiree. Acquisition-related costs are generally recognized in profit or loss as incurred.
At the acquisition date, the identifiable assets acquired and the liabilities assumed are recognized at their fair value at the acquisition date.
Goodwill is measured as the excess of the sum of the consideration transferred, the amount of any non-controlling interests in the acquiree, and the fair value of the acquirer's previously held equity interest in the acquiree (if any) over the net amount of the identifiable assets acquired and the liabilities assumed as at acquisition date.
Goodwill
Goodwill arising on an acquisition of a business is carried at cost as established at the date of acquisition of the business (see the accounting policy above) less accumulated impairment losses, if any.
For the purpose of impairment testing, goodwill is allocated to each of the Group’s cash-generating units (or groups of cash-generating units) that is expected to benefit from the synergies of the combination, which represent the lowest level at which the goodwill is monitored for internal management purposes and is not larger than an operating segment .
A cash-generating units (or groups of cash-generating units) to which goodwill has been allocated is tested for impairment annually or more frequently when there is indication that the unit may be impaired. For goodwill arising on an acquisition in an annual period, the cash-generating unit (or group of cash-generating units) to which goodwill has been allocated is tested for impairment before the end of that annual period. If the recoverable amount is less than its carrying amount, the impairment loss is allocated first to reduce the carrying amount of any goodwill and then to the other assets on a pro-rata basis based on the carrying amount of each asset in the unit (or group of cash-generating units).
F-13
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
On disposal of the relevant cash-generating unit or any of the cash-generating unit within the group of cash generating units, the attributable amount of goodwill is included in the determination of the amount of the profit or loss on disposal.
Interests in equity-accounted investees
An associate is an entity over which the Group has significant influence. Significant influence is the power to participate in the financial and operating policy decisions of the investee but is not control or joint control over those policies.
The results and assets and liabilities of associates are incorporated in these consolidated financial statements using the equity method of accounting. Under the equity method, an investment in an associate is initially recognized in the consolidated statement of financial position at cost and adjusted thereafter to recognize the Group’s share of the profit or loss and other comprehensive income of the associate. When the Group’s share of losses of an associate exceeds the Group’s interest in that associate (which includes any long-term interests that, in substance, form part of the Group’s net investment in the associate), the Group discontinues recognizing its share of further losses. Additional losses are provided for, and a liability is recognized only to the extent that the Group has incurred legal or constructive obligations or made payments on behalf of the associate.
An investment in an associate is accounted for using the equity method from the date on which the investee becomes an associate. On acquisition of the investment in an associate, any excess of the cost of the investment over the Group’s share of the net fair value of the identifiable assets and liabilities of the investee is recognized as goodwill, which is included within the carrying amount of the investment.
The Group assesses whether there is an objective evidence that the interest in an associate or a joint venture may be impaired. When any objective evidence exists, the entire carrying amount of the investment (including goodwill) is tested for impairment in accordance with IAS 36 Impairment of Assets ("IAS 36") as a single asset by comparing its recoverable amount (higher of value in use and fair value less costs of disposal) with its carrying amount. Any impairment loss recognized is not allocated to any asset, including goodwill, that forms part of the carrying amount of the investment. Any reversal of that impairment loss is recognized in accordance with IAS 36 to the extent that the recoverable amount of the investment subsequently increases.
Revenue from contracts with customers
The Group recognizes revenue from the following major products:
•sale of consumer health products;
•provision of genetic testing services;
•provision of fulfillment and distribution services of sports nutrition products; and
•provision of precision oncology services.
Revenue is measured based on the consideration to which the Group expects to be entitled in a contract with a customer and excludes amounts collected on behalf of third parties. The Group recognizes revenue when it transfers control over a product or service to customer. An asset is transferred when the customer obtains control of the asset.
The Group transfers control of a good or service at a point in time unless one of the following overtime criteria is met:
(a)the customer simultaneously receives and consumes the benefits provided as the Group performs;
(b)the Group’s performance creates or enhances an asset that the customer controls as the asset is created or enhanced; or
(c)the Group’s performance does not create an asset with an alternative use and the Group has an enforceable right to payment for performance completed to date.
F-14
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Sales of consumer health products
The Group sells consumer health products directly to customers. Revenue is recognized at a point in time when control of the goods transfers to the customer, which occurs when the goods are handed over to the courier for delivery (i.e. shipment). Revenue is recorded net of discount. When the customer initially purchases the goods online the transaction price received by the Group is recognized as a contract liability until the goods have been delivered to the customer. There exists a 30-day right of return on orders and a refund liability is recognized for expected refunds to customers.
Provision of fulfillment and distribution services
The Group sells sports nutrition products typically satisfies its performance obligations for sales of goods as goods are delivered and control of the goods transfers to the customer. Sales of products by the Group may include fees for shipping and handling.
The Group satisfies its performance obligations for fulfillment and distribution services at a point in time typically when the customer's goods are delivered to the named destination. The Group provides services for storage, packaging, receiving, and in-bound freight management and satisfies those performance obligations over-time.
Provision of genetic testing and precision oncology services
The Group provides i) genetic testing services to individuals and corporates for their employees and customers; and ii) diagnostic services which are precision oncology services.
The Group collects consideration for genetic testing services to individuals and corporations upfront, and such consideration received usually becomes non-refundable up to 30 days from the date of delivery of the kits to individuals and corporations, or the date of purchase. Revenue is recorded net of discount. The upfront consideration received is initially recognized as deposit liabilities and subsequently reclassified to contract liabilities when the amount becomes non-refundable. Such amount does not include any variable consideration.
The Group collects consideration for precision oncology services to patient from public hospitals upfront, and such consideration received usually becomes non-refundable from the date of receipt of patients' specimen, or the date of purchase. The upfront consideration received is recognized as contract liabilities when the amount becomes non-refundable. Such amount does not include any variable consideration.
The Group determines that its sales contracts do not
On October 1, 2025, the Group completed the disposal of ACT Genomics. Following the disposal, the Group no longer has exposure to the operation of precision oncology services business and is classified as discontinued operations. Details of discontinued operations are disclosed in note 10.
Performance obligations
Generally, the Group fulfilled its performance obligations for genetic testing and precision oncology services at a point in time upon delivery of the testing results or reports to customers or patients.
Revenue breakage
Provision of genetic testing services require individuals to provide specimen samples to the Group before it can proceed with the necessary laboratory procedures. Sales contracts relating to testing kits sold directly to individuals normally require specimen samples to be sent back to the Group within one year (the “sample return period”) from the date of purchase depending on the jurisdictions in which the kits are purchased by customers. If these customers do not return their specimen samples within the sample return period, the Group has no further obligation to provide the service. Sales contracts relating to kits sold to corporations normally do not include specified sample return periods.
The Group generally has sufficient and relevant historical experience for other sales contracts such that the Group expects to be entitled to a breakage amount in relation to non-refundable and unexercised rights. For these sales contracts, the Group estimates and recognizes the expected breakage amount as revenue in proportion to the pattern of rights exercised by customers on a portfolio basis to the extent that it is considered highly probable that a significant reversal will not occur in the future.
F-15
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
The Group updates its breakage estimate regularly and if necessary, adjusts the deferred revenue balance accordingly. If actual return patterns vary from the estimate, actual breakage revenue may differ from the amounts recorded.
Leases
The Group assesses whether a contract is or contains a lease based on the definition under IFRS 16 at inception of the contract. Such contract will not be reassessed unless the terms and conditions of the contract are subsequently changed.
The Group as lessee
Short-term leases and leases of low-value assets
The Group applies the short-term lease recognition exemption to leases that have a lease term of 12 months or less from the commencement date and do not contain a purchase option. It also applies the recognition exemption for lease of low-value assets (such as IT equipment). Lease payments on short-term leases and leases of low-value assets are recognized as expense on a straight-line basis unless another systematic basis is more representative of the time pattern in which economic benefits from the leased assets are consumed.
Right-of-use assets
The cost of right-of-use assets include:
•the amounts of the initial measurement of the lease liabilities;
•any lease payments made at or before the commencement date, less any lease incentives received;
•any initial direct costs incurred by the Group; and
•an estimate of costs to be incurred by the Group in dismantling and removing the underlying assets, restoring the site on which it is located or restoring the underlying asset to the condition required by the terms and conditions of the lease.
Right-of-use assets are measured at cost, less any accumulated depreciation and impairment losses, and adjusted for any remeasurement of lease liabilities.
Right-of-use assets in which the Group is reasonably certain to obtain ownership of the underlying leased assets at the end of the lease term are depreciated from commencement date to the end of the useful life. Otherwise, right-of-use assets are depreciated on a straight-line basis over the shorter of its estimated useful life and the lease term.
The Group presents right-of-use assets that do not meet the definition of investment property in "property, plant and equipment", the same line item within which the corresponding underlying assets would be presented if they were owned.
Refundable rental deposits
Refundable rental deposits paid are accounted under IFRS 9 and initially measured at fair value. Adjustments to fair value at initial recognition are considered as additional lease payments and included in the cost of right-of-use assets.
Lease liabilities
At the commencement date of a lease, the Group recognizes and measures the lease liability at the present value of lease payments that are unpaid at that date. In calculating the present value of lease payments, the Group uses the incremental borrowing rate at the lease commencement date if the interest rate implicit in the lease is not readily determinable. The incremental borrowing rate depends on the term, currency and start date of the lease.
The Group determines its incremental borrowing rate by obtaining interest rates from various external financing sources and makes certain adjustments to reflect the terms of the lease and type of the asset leased and the lease does not benefit from a guarantee from the Group.
The lease payments include fixed payments (including in-substance fixed payments).
After the commencement date, lease liabilities are adjusted by interest accretion and lease payments.
The Group presents lease liabilities as a separate line item on the consolidated statement of financial position.
F-16
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Foreign currencies
In preparing the financial statements of each individual group entity, transactions in currencies other than the functional currency of that entity (foreign currencies) are recognized at the rates of exchange prevailing on the dates of the transactions. At the end of the reporting period, monetary items denominated in foreign currencies are retranslated at the rates prevailing at that date. Non-monetary items carried at fair value that are denominated in foreign currencies are retranslated at the rates prevailing on the date when the fair value was determined. Non-monetary items that are measured in terms of historical cost in a foreign currency are not retranslated.
Exchange differences arising on the settlement of monetary items, and on the retranslation of monetary items, are recognized in profit or loss in the period in which they arise.
For the purpose of presenting consolidated financial statements, the assets and liabilities of the Group’s operations are translated into the presentation currency of the Group (i.e. US$) using exchange rates prevailing at the end of each reporting period unless exchange rates fluctuate significantly during that period, in which case the exchange rates at the date of transactions are used. Exchange differences arising, if any, are recognized in other comprehensive income and accumulated in equity under the heading of translation reserve.
On the disposal of a foreign operation (that is, a disposal of the Group’s entire interest in a foreign operation), all of the exchange differences accumulated in equity in respect of that operation attributable to the owners of the Company are reclassified to profit or loss.
Goodwill and fair value adjustments on identifiable assets acquired arising on an acquisition of a foreign operation are treated as assets and liabilities of the foreign operation and translated at the rate exchange prevailing at the end of each reporting period. Exchange differences arising are recognized in other comprehensive income.
Employee benefits
Retirement benefit costs
Payments to defined contribution retirement benefit plans are recognized as an expense when employees have rendered service entitling them to the contributions.
Short-term employee benefits
Short-term employee benefits are recognized at the undiscounted amount of the benefits expected to be paid as and when employees rendered the services. All short-term employee benefits are recognized as an expense unless another IFRS Accounting Standard requires or permits the inclusion of the benefit in the cost of an asset.
A liability is recognized for benefits accruing to employees (such as wages and salaries and annual leave) after deducting any amount already paid.
Equity-settled share-based payment transactions
Share awards granted to employees and others providing similar services
Equity-settled share-based payments to employees and others providing similar services are measured at the fair value of the equity instruments at the grant date.
The fair value of the equity-settled share-based payments determined at the grant date without taking into consideration all non-market vesting conditions is expensed on a straight-line basis over the vesting period, based on the Group's estimate of equity instruments that will eventually vest, with a corresponding increase in equity (capital reserve). At the end of each reporting period, the Group revises its estimate of the number of equity instruments expected to vest based on assessment of all relevant non-market vesting conditions. The impact of the revision of the original estimates, if any, is recognized in profit or loss such that the cumulative expense reflects the revised estimate, with a corresponding adjustment to the capital reserve. For shares that vest immediately at the date of grant, the fair value of the shares granted is expensed immediately to profit or loss.
Shares granted to non-employees
Equity-settled share-based payment transactions with parties other than employees are measured at the fair value of the services received, except where that fair value cannot be estimated reliably, in which case they are measured at the fair value of the equity instruments granted, measured at the date the counterparty renders the service. The fair values of the services received are recognized as expenses (unless the goods or services qualify for recognition as assets).
F-17
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Modification to the terms and conditions of the share-based payment arrangements
When the terms and conditions of an equity-settled share-based payment arrangement are modified, the Group recognizes, as a minimum, the services received measured at the grant date fair value of the equity instruments granted, unless those equity instruments do not vest because of failure to satisfy a vesting condition (other than a market condition) that was specified at grant date. In addition, if the Group modifies the vesting conditions (other than a market condition) in a manner that is beneficial to the employees, for example, by reducing the vesting period, the Group takes the modified vesting conditions into consideration over the remaining vesting period.
Taxation
The income tax expense represents the sum of current and deferred income tax expense.
The tax currently payable is based on taxable profit for the year. Taxable profit differs from loss before taxation because of income or expense that are taxable or deductible in other years and items that are never taxable or deductible. The Group’s liability for current tax is calculated using tax rates that have been enacted or substantively enacted by the end of the reporting period.
Deferred tax is recognized on temporary differences between the carrying amounts of assets and liabilities in the consolidated financial statements and the corresponding tax bases used in the computation of taxable profit. Deferred tax liabilities are generally recognized for all taxable temporary differences. Deferred tax assets are generally recognized for all deductible temporary differences to the extent that it is probable that taxable profits will be available against which those deductible temporary differences can be utilized. Such deferred tax assets and liabilities are not recognized if the temporary difference arises from the initial recognition (other than in a business combination) of assets and liabilities in a transaction that affects neither the taxable profit nor the accounting profit and at the time of the transaction does not give rise to equal taxable and deductible temporary differences. In addition, deferred tax liabilities are not recognized if the temporary difference arises from the initial recognition of goodwill.
The carrying amount of deferred tax assets is reviewed at end of each reporting period and reduced to the extent that it is no longer probable that sufficient taxable profits will be available to allow all or part of the asset to be recovered.
Deferred tax assets and liabilities are measured at the tax rates that are expected to apply in the period in which the liability is settled or the asset is realized, based on tax rates (and tax laws) that have been enacted or substantively enacted by the end of the reporting period.
The measurement of deferred tax liabilities and assets reflects the tax consequences that would follow from the manner in which the Group expects, at the end of the reporting period, to recover or settle the carrying amount of its assets and liabilities.
Deferred tax assets and liabilities are offset when there is a legally enforceable right to set off current tax assets against current tax liabilities and when they relate to income taxes levied to the same taxable entity by the same taxation authority.
Current and deferred tax are recognized in profit or loss.
Loss per share
Basic loss per share is computed by dividing net loss attributable to ordinary shareholders by the weighted average number of ordinary shares outstanding during the year. Diluted loss per share reflect the potential dilution that could occur if securities or other contracts to issue ordinary shares were exercised or converted into ordinary shares.
Property, plant and equipment
Property, plant and equipment are tangible assets that are held for use in the production or supply of goods or services, or for administrative purposes. Property, plant and equipment are stated in the consolidated statement of financial position at cost less subsequent accumulated depreciation and subsequent accumulated impairment losses, if any.
Depreciation is recognized so as to write off the cost of assets less their residual values over their estimated useful lives, using the straight-line method. The estimated useful lives, residual values and depreciation method are reviewed at the end of each reporting period, with the effect of any changes in estimate accounted for on a prospective basis.
F-18
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
The estimated useful lives of property, plant and equipment for current and comparative periods are as follows:
| – Properties leased for own use | Over the unexpired lease period | ||||
| – Office equipment leased for own use | Over the unexpired lease period | ||||
| – Leasehold improvements | Shorter period of the lease term or the useful life | ||||
| – Fixtures and furniture | |||||
| – Office and lab equipment | |||||
| – Computer equipment | |||||
| – Motor vehicles | |||||
| – Manufacturing equipment | |||||
An item of property, plant and equipment is derecognized upon disposal or when no future economic benefits are expected to arise from the continued use of the asset. Any gain or loss arising on the disposal or retirement of an item of property, plant and equipment is determined as the difference between the sales proceeds and the carrying amount of the asset and is recognized in profit or loss.
Intangible assets
Intangible assets acquired separately
Intangible assets with finite useful lives that are acquired separately are carried at costs less accumulated amortization and any accumulated impairment losses amounts. Amortization for intangible assets with finite useful lives is recognized on a straight-line basis over their estimated useful lives. The estimated useful life and amortization method are reviewed at the end of each reporting period, with the effect of any changes in estimate being accounted for on a prospective basis. Intangible assets with indefinite useful lives that are acquired separately are carried at cost less any subsequent accumulated impairment losses.
The estimated useful lives for current and comparative periods are as follows:
| – Website and mobile apps | |||||
| – Trademark and technology | |||||
| – Products development cost | |||||
| – Computer software | |||||
| – Customer relationship | |||||
Research and development expenditure
Expenditure on research activities is recognized as an expense in the period which it is incurred.
Intangible assets acquired in a business combination
Intangible assets acquired in a business combination are recognized separately from goodwill and are initially recognized at their fair value at the acquisition date (which is regarded as their cost).
Subsequent to initial recognition, intangible assets acquired in a business combination with finite useful lives are reported at costs less accumulated amortization and any accumulated impairment losses, on the same basis as intangible assets that are acquired separately. Intangible assets acquired in a business combination with indefinite useful lives are carried at cost less any subsequent accumulated impairment losses.
An intangible asset is derecognized on disposal, or when no future economic benefits are expected from use or disposal. Gains or losses arising from derecognition of an intangible asset, measured as the difference between the net disposal proceeds and the carrying amount of the asset, are recognized in profit or loss when the asset is derecognized.
F-19
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Digital assets
Digital assets on hand at the end of a reporting period, are accounted for under IAS 38 Intangible Assets, as an intangible asset with an indefinite useful life initially measured at cost, deemed to be the fair value upon receipt and subsequently measured under the revaluation model. Under the revaluation model, increases in the digital assets’ carrying amount, determined as the excess of fair value on revaluation over the weighted average cost, are recognized in other comprehensive income, except to the extent that they reverse a revaluation decrease previously recognized in profit or loss. Decreases are recognized in profit or loss, except to the extent that they reverse a revaluation increase previously recognized in other comprehensive income. Once the digital asset is sold, the revaluation increase related to it is transferred from revaluation surplus to retained earnings. The Company revalues its digital assets on hand on a monthly basis and following any significant fair value fluctuations. The fair value of digital assets on hand at the end of the reporting period is determined as the quantity of digital assets on hand multiplied by the quoted market price on an active exchange at the reporting date.
Impairment on property, plant and equipment, right-of-use assets and intangible assets
At the end of each reporting period, the Group reviews the carrying amounts of its property, plant and equipment, right-of-use assets and intangible assets with finite useful lives to determine whether there is any indication that these assets have suffered an impairment loss. If any such indication exists, the recoverable amount of the relevant asset is estimated in order to determine the extent of the impairment loss (if any).
The recoverable amount of property, plant and equipment, right-of-use assets and intangible assets are estimated individually. When it is not possible to estimate the recoverable amount individually, the Group estimates the recoverable amount of the cash-generating unit to which the asset belongs.
In testing a cash-generating unit for impairment, corporate assets are allocated to the relevant cash-generating unit when a reasonable and consistent basis of allocation can be established, or otherwise they are allocated to the smallest group of cash generating units for which a reasonable and consistent allocation basis can be established. The recoverable amount is determined for the cash-generating unit or group of cash-generating units to which the corporate asset belongs, and is compared with the carrying amount of the relevant cash-generating unit or group of cash-generating units.
Recoverable amount is the higher of fair value less costs of disposal and value in use. In assessing value in use, the estimated future cash flows are discounted to their present value using a pre-tax discount rate that reflects current market assessments of the time value of money and the risks specific to the asset (or a cash-generating unit) for which the estimates of future cash flows have not been adjusted.
If the recoverable amount of an asset (or a cash-generating unit) is estimated to be less than its carrying amount, the carrying amount of the asset (or a cash-generating unit) is reduced to its recoverable amount. For corporate assets or portion of corporate assets which cannot be allocated on a reasonable and consistent basis to a cash-generating unit, the Group compares the carrying amount of a group of cash-generating units, including the carrying amounts of the corporate assets or portion of corporate assets allocated to that group of cash-generating units, with the recoverable amount of the group of cash-generating units. In allocating the impairment loss, the impairment loss is allocated first to reduce the carrying amount of any goodwill (if applicable) and then to the other assets on a pro-rata basis based on the carrying amount of each asset in the unit or the group of cash-generating units. The carrying amount of an asset is not reduced below the highest of its fair value less costs of disposal (if measurable), its value in use (if determinable) and zero. The amount of the impairment loss that would otherwise have been allocated to the asset is allocated pro rata to the other assets of the unit or the group of cash-generating units. An impairment loss is recognized immediately in profit or loss.
Where an impairment loss subsequently reverses, the carrying amount of the asset (or cash-generating unit or a group of cash-generating units) is increased to the revised estimate of its recoverable amount, but so that the increased carrying amount does not exceed the carrying amount that would have been determined had no impairment loss been recognized for the asset (or a cash-generating unit or a group of cash-generating units) in prior years. A reversal of an impairment loss is recognized immediately in profit or loss.
F-20
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Cash and cash equivalents
Cash and cash equivalents presented on the consolidated statement of financial position include:
a.cash, which comprises of cash on hand and demand deposits; and
b.cash equivalents, which comprises of short-term investments (generally with original maturity of three months or less).
For the purposes of the consolidated statement of cash flows, cash and cash equivalents consist of cash and cash equivalents as defined above.
Inventories
Inventories are stated at the lower of cost and net realizable value. Cost of inventories are determined on a first-in, first-out allocation method. Net realizable value represents the estimated selling price for inventories less all estimated costs of completion and costs necessary to make the sale. Costs necessary to make the sale include incremental costs directly attributable to the sale and non-incremental costs which the Group must incur to make the sale.
Financial instruments
Financial assets and financial liabilities are recognized when a group entity becomes a party to the contractual provisions of the instrument.
Financial assets and financial liabilities are initially measured at fair value, except for trade receivables arising from contracts with customers which are initially measured in accordance with IFRS 15. Transaction costs that are directly attributable to the acquisition or issue of financial assets and financial liabilities (other than financial assets and financial liabilities at fair value through profit or loss ("FVTPL")) are added to or deducted from the fair value of the financial assets or financial liabilities, as appropriate, on initial recognition. Transaction costs directly attributable to the acquisition of financial assets or financial liabilities at FVTPL are recognized immediately in profit or loss.
The effective interest method is a method of calculating the amortized cost of a financial asset or financial liability and of allocating interest income and interest expense over the relevant period. The effective interest rate is the rate that exactly discounts estimated future cash receipts and payments (including all fees and points paid or received that form an integral part of the effective interest rate, transaction costs and other premiums or discounts) through the expected life of the financial asset or financial liability, or, where appropriate, a shorter period, to the net carrying amount on initial recognition.
Financial assets
All regular way purchases or sales of financial assets are recognized and derecognized on a trade date basis. Regular way purchases or sales are purchases or sales of financial assets that require delivery of assets within the time frame established generally by regulation or convention in the market place concerned.
All recognized financial assets are measured subsequently in their entirety at either amortized cost or fair value, depending on the classification of the financial assets.
Classification of financial assets
Financial assets that meet the following conditions are subsequently measured at amortized cost:
•the financial asset is held within a business model whose objective is to collect contractual cash flows; and
•the contractual terms give rise on specified dates to cash flows that are solely payments of principal and interest on the principal amount outstanding.
All other financial assets are subsequently measured at FVTPL.
F-21
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
(i)Amortized cost and interest income
Interest income is recognized using the effective interest method for debt instruments measured subsequently at amortized cost. Interest income is calculated by applying the effective interest rate to the gross carrying amount of a financial asset, except for financial assets that have subsequently become credit-impaired (see below). For financial assets that have subsequently become credit-impaired, interest income is recognized by applying the effective interest rate to the amortized cost of the financial asset from the next reporting period. If the credit risk on the credit-impaired financial instrument improves so that the financial asset is no longer credit-impaired, interest income is recognized by applying the effective interest rate to the gross carrying amount of the financial asset from the beginning of the reporting period following the determination that the asset is no longer credit-impaired.
(ii)Financial assets at FVTPL
Financial assets that do not meet the criteria for being measured at amortized cost are measured at FVTPL.
Financial assets at FVTPL are measured at fair value at the end of each reporting period, with any fair value gains or losses recognized in profit or loss. The net gain or loss recognized in profit or loss includes any dividend or interest earned on the financial asset and is included in the ‘other income and other net gains’ line item.
Business model assessment of financial assets
The business models of the Group are as follows.
Held to collect
The Group holds financial assets which arise from its principal operations. The objective of the business model for these financial instruments is to collect the amounts due from the Group’s receivables and to earn contractual interest income on the amounts collected.
Assessment whether contractual cash flows are solely payments of principal and interest of financial assets
In assessing whether the contractual cash flows are solely payments of principal and interest ("SPPI"), the Group considers the contractual terms of the instrument. This includes assessing whether the financial asset contains a contractual term that could change the timing or amount of contractual cash flows such that it would not meet this condition. In making this assessment, the Group considers:
•contingent events that would change the amount or timing of cash flows;
•terms that may adjust the contractual coupon rate, including variable-rate features;
•prepayment and extension features; and
•terms that limit the Group’s claim to cash flows from specified assets (e.g. non-recourse features).
A prepayment feature is consistent with the SPPI criterion if the prepayment amount substantially represents unpaid amounts of principal and interest on the principal amount outstanding.
Impairment of financial assets subject to impairment assessment under IFRS 9
The Group performs impairment assessment under expected credit losses ("ECL") model on financial assets (including trade receivables, other receivables, amount due from a related company and cash and cash equivalent), which are subject to impairment assessment under IFRS 9. The amount of ECL is updated at each reporting date to reflect changes in credit risk since initial recognition.
The Group always recognizes lifetime ECL for trade receivables. The expected credit losses on these financial assets are estimated using a provision matrix based on the Group’s historical credit loss experience, adjusted for factors that are specific to the debtors, general economic conditions and an assessment of both the current as well as the forecast direction of conditions at the reporting date, including time value of money where appropriate.
For all other instruments, the Group measures the loss allowance equal to 12-month ECL ("12m ECL"), unless there has been a significant increase in credit risk since initial recognition, in which case the Group recognizes lifetime ECL. The assessment of whether lifetime ECL should be recognized is based on significant increases in the likelihood or risk of a default occurring since initial recognition.
F-22
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Lifetime ECL represents the ECL that will result from all possible default events over the expected life of the relevant instrument. In contrast, 12m ECL represents the portion of lifetime ECL that is expected to result from default events that are possible within 12 months after the reporting date. Assessments are done based on the Group’s historical credit loss experience, adjusted for factors that are specific to the debtors, general economic conditions and an assessment of past events and current conditions at the reporting date as well as the forecast of future economic conditions.
(i)Significant increase in credit risk
In assessing whether the credit risk on a financial instrument has increased significantly since initial recognition, the Group compares the risk of a default occurring on the financial instrument as at the reporting date with the risk of a default occurring on the financial instrument as at the date of initial recognition. In making this assessment, the Group considers both quantitative and qualitative information that is reasonable and supportable, including historical experience and forward-looking information that is available without undue cost or effort. Forward-looking information considered includes the future prospects of the industries in which the Group’s debtors operate, obtained from economic expert reports, financial analysts, governmental bodies, relevant think-tanks and other similar organizations, as well as consideration of various external sources of actual and forecast economic information that relate to the Group’s core operations.
In particular, the following information is taken into account when assessing whether credit risk has increased significantly:
•an actual or expected significant deterioration in the financial instrument’s external (if available) or internal credit rating;
•significant deterioration in external market indicators of credit risk, e.g. a significant increase in the credit spread, the credit default swap prices for the debtor;
•existing or forecast adverse changes in business, financial or economic conditions that are expected to cause a significant decrease in the debtor’s ability to meet its debt obligations;
•an actual or expected significant deterioration in the operating results of the debtor;
•an actual or expected significant adverse change in the regulatory, economic, or technological environment of the debtor that results in a significant decrease in the debtor’s ability to meet its debt obligations.
Irrespective of the outcome of the above assessment, the Group has reasonable and supportable information to rebut the presumption the credit risk has increased significantly since initial recognition when contractual payments are more than 30 days past due.
Despite the foregoing, the Group assumes that the credit risk on a debt instrument has not increased significantly since initial recognition if the debt instrument is determined to have low credit risk at the reporting date. A debt instrument is determined to have low credit risk if (i) it has a low risk of default, (ii) the borrower has a strong capacity to meet its contractual cash flow obligations in the near term and (iii) adverse changes in economic and business conditions in the longer term may, but will not necessarily, reduce the ability of the borrower to fulfil its contractual cash flow obligations. The Group considers a debt instrument to have low credit risk when it has and internal or external credit rating of ‘investment grade’ as per globally understood definitions.
The Group regularly monitors the effectiveness of the criteria used to identify whether there has been a significant increase in credit risk and revises them as appropriate to ensure that the criteria are capable of identifying significant increase in credit risk before the amount becomes past due.
F-23
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
(ii)Definition of default
The Group considers the following as constituting an event of default for internal credit risk management purposes as historical experience indicates that financial assets that meet either of the following criteria are generally not recoverable:
•when there is a breach of financial covenants by the debtor; or
•information developed internally or obtained from external sources indicates that the debtor is unlikely to pay its creditors, including the Group, in full (without taking into account any collateral held by the Group).
Irrespective of the above analysis, the Group considers that default has occurred when a financial asset is more than 90 days past due unless the Group has reasonable and supportable information to demonstrate that a more lagging default criterion is more appropriate.
(iii)Credit-impaired financial assets
A financial asset is credit-impaired when one or more events that have a detrimental impact on the estimated future cash flows of that financial asset have occurred. Evidence that a financial asset is credit-impaired includes observable data about the following events:
•significant financial difficulty of the issuer or the borrower;
•a breach of contract, such as a default or past due event;
•the lender(s) of the borrower, for economic or contractual reasons relating to the borrower’s financial difficulty, having granted to the borrower a concession(s) that the lender(s) would not otherwise consider;
•it is becoming probable that the borrower will enter bankruptcy or other financial reorganization; or
•the disappearance of an active market for that financial asset because of financial difficulties.
(iv)Write-off policy
The Group writes off a financial asset when there is information indicating that the counterparty is in severe financial difficulty and there is no realistic prospect of recovery, for example, when the counterparty has been placed under liquidation or has entered into bankruptcy proceedings, or in the case of trade receivables, when the amounts are over 180 days past due, whichever occurs sooner. Financial assets written off may still be subject to enforcement activities under the Group’s recovery procedures, taking into account legal advice where appropriate. A write-off constitutes a derecognition event. Any subsequent recoveries are recognized in profit or loss.
(v)Measurement and recognition of expected credit losses
The measurement of ECL is a function of the probability of default, loss given default (i.e. the magnitude of the loss if there is a default) and the exposure at default. The assessment of the probability of default and loss given default is based on historical data and forward-looking information. Estimation of ECL reflects an unbiased and probability-weighted amount that is determined with the respective risks of default occurring as the weights. The Group uses a practical expedient in estimating ECL on trade receivables using a provision matrix taking into consideration historical credit loss experience, adjusted for factors that are specific to the debtors, general economic conditions and forward-looking information, including time value of money where appropriate, that is available without undue cost or effort.
Generally, the ECL is the difference between all contractual cash flows that are due to the Group in accordance with the contract and the cash flows that the Group expects to receive, discounted at the original effective interest rate determined at initial recognition.
Interest income is calculated based on the gross carrying amount of the financial asset unless the financial asset is credit-impaired, in which case interest income is calculated based on amortized cost of the financial asset.
F-24
3 Basis of preparation of consolidated financial statements and material accounting policy information (continued)
Derecognition of financial assets
The Group derecognizes a financial asset only when the contractual rights to the cash flows from the asset expire, or when it transfers the financial asset and substantially all the risks and rewards of ownership of the asset to another entity. If the Group neither transfers nor retains substantially all the risks and rewards of ownership and continues to control the transferred asset, the Group recognizes its retained interest in the asset and an associated liability for amounts it may have to pay. If the Group retains substantially all the risks and rewards of ownership of a transferred financial asset, the Group continues to recognize the financial asset and also recognizes a collateralized borrowing for the proceeds received.
On derecognition of a financial asset measured at amortized cost, the difference between the asset’s carrying amount and the sum of the consideration received and receivable is recognized in profit or loss.
Financial liabilities and equity
Classification as debt or equity
Debt and equity instruments are classified as either financial liabilities or as equity in accordance with the substance of the contractual arrangements and the definitions of a financial liability and an equity instrument.
Financial liabilities
All financial liabilities are subsequently measured at amortized cost using the effective interest method or at FVTPL.
Derecognition of financial liabilities
The Group derecognizes financial liabilities when, and only when, the Group’s obligations are discharged, cancelled or have expired. The difference between the carrying amount of the financial liability derecognized and the consideration paid and payable is recognized in profit or loss.
Liabilities for puttable financial instrument
Liabilities for puttable financial instrument are an obligation arising from put options written to non-controlling shareholders of subsidiaries, which will be settled based on the fair value of the shares held by the non-controlling shareholders, results in a gross financial liability. The gross financial liability is initially recognized and measured at amortize cost with the corresponding debit to the “other reserve”. In subsequent periods, the changes of fair value is recognized in ‘other reserve’.
Discontinued operations
A discontinued operation is a component of the Group’s business, the operations and cash flows of which can be clearly distinguished from the rest of the Group and which:
•represents a separate major line of business or geographic area of operations;
•is a part of a single co-ordinated plan to dispose of a separate major line of business or geographic area of operations; or
•is a subsidiary acquired exclusively with a view to resale.
Classification as a discontinued operation occurs at the earlier of disposal or when the operation meets the criteria to be classified as held-for-sale.
When an operation is classified as a discontinued operation, the comparative statement of profit or loss and other comprehensive income is re-presented as if the operation had been discontinued from the start of the comparative year.
4 Key sources of estimation uncertainty
The key assumptions concerning the future, and other key sources of estimation uncertainty at the reporting period that may have a significant risk of causing a material adjustment to the carrying amounts of assets and liabilities within the next financial year, are discussed below.
F-25
4Key sources of estimation uncertainty (continued)
Fair value measurements and valuation processes
Certain Group’s accounting policies and disclosures require the measurement of fair values, for both financial and non-financial assets and liabilities.
The Group assigned its finance team to oversee all significant fair value measurements, including Level 3 fair values, and reports directly to the chief financial officer.
The finance team regularly reviews significant unobservable inputs and valuation adjustments. If third party information, such as broker quotes or pricing services, is used to measure fair values, then the finance team assesses the evidence obtained from the third parties to support the conclusion that these valuations meet the requirements of the IFRS Accounting Standards, including the level in the fair value hierarchy in which the valuations should be classified.
Significant valuation issues are reported to the Group’s audit committee.
When measuring the fair value of an asset or a liability, the Group uses observable market data as far as possible. Fair values are categorized into different levels in a fair value hierarchy based on the inputs used in the valuation techniques as follows.
•Level 1: quoted prices (unadjusted) in active markets for identical assets or liabilities.
•Level 2: inputs other than quoted prices included in Level 1 that are observable for the asset or liability, either directly (i.e. as prices) or indirectly (i.e. derived from prices).
•Level 3: inputs for the asset or liability that are not based on observable market data (unobservable inputs).
If the inputs used to measure the fair value of an asset or a liability fall into different levels of the fair value hierarchy, then the fair value measurement is categorized in its entirety in the same level of the fair value hierarchy as the lowest level input that is significant to the entire measurement.
The Group recognizes transfers between levels of the fair value hierarchy at the end of the reporting period during which the change has occurred.
Further information about the assumptions made in measuring fair values is included in the following notes:
•Note 15: digital assets;
•Note 27: warrant liabilities;
•Note 30: equity-settled share-based transactions;
•Note 31(B): financial instruments; and
•Note 33: acquisition of Europa.
Estimated impairment of goodwill
Determining whether goodwill is impaired requires an estimation of the recoverable amount of the cash-generating unit (or group of cash-generating units) to which goodwill has been allocated, which is the higher of the value in use or fair value less costs of disposal. For the year ended December 31, 2025, the recoverable amount was determined by management based on internally developed valuation models. For the year ended December 31, 2024, the Group engaged an independent qualified professional valuer to support the determination of the recoverable amount; however, management remained responsible for the assumptions and conclusions applied in the impairment assessment.
The value in use calculation requires the Group to estimate the future cash flows with key assumption including budgeted sales of the "cash generating units ("CGU") expected to arise from the CGU and a suitable discount rate in order to calculate the present value.
Key assumptions include forecast revenue growth, operating margins, terminal growth rates and discount rates. Where the actual future cash flows are less than expected, or changes in facts and circumstances result in a downward revision of future cash flows or an upward revision of discount rates, a material impairment loss or further impairment loss may arise.
The fair value less costs of disposal is determined using valuation techniques based on market participant assumptions, including reference to recent transaction prices for comparable assets where available.
F-26
4Key sources of estimation uncertainty (continued)
The determination of the recoverable amount involves significant judgement, particularly in assessing the appropriate valuation methodology and estimating key assumptions. The recoverable amount is sensitive to changes in these assumptions, and a reasonably possible change in key inputs, including discount rates and projected cash flows, could result in a material change in the impairment assessment.
At December 31, 2025 and 2024, the carrying amounts of goodwill were approximately $1,379 and $37,364 , respectively. During the year ended December 31, 2025, an impairment loss of $6,815 was recognized in respect of goodwill. Details of significant judgements and assumptions applied in the impairment assessment are disclosed in note 12.
Fair value measurement of warrant liabilities
Warrant liabilities are stated at fair value based on the valuation performed by independent professional valuers. The determination of the fair value involves the use of valuation techniques and key assumptions, which are set out in note 27.
In relying on the valuation reports, the directors of the Company have exercised their judgement and are satisfied that the valuation methodologies, including binomial option pricing models and Monte Carlo simulation models, are appropriate and reflective of the contractual terms of the warrants and current market conditions.
The valuation of warrant liabilities involves significant unobservable inputs, including expected volatility, expected term and other model assumptions. These inputs are inherently uncertain and subject to management judgement. In light of the prevailing macroeconomic environment, including volatility in equity markets, interest rates and broader financial market conditions, there is a higher degree of estimation uncertainty associated with the valuation of these financial instruments.
Whilst the Group considers the fair value of warrant liabilities to be the best estimate, changes in the underlying assumptions, including fluctuations in share price, expected volatility, risk-free interest rates or changes in market conditions, could result in material changes to the fair value of the warrant liabilities and the corresponding gain or loss recognized in the consolidated statement of profit or loss.
As at December 31, 2025, the carrying amount of warrant liabilities which classified within Level 3 of the fair value hierarchy was $19,652 . A reasonably possible change in key unobservable inputs, in particular expected volatility, would result in a corresponding increase or decrease in the fair value of the warrant liabilities.
F-27
5 Revenue
The Group derives its revenue from contracts with customers for the transfer of goods and services at a point in time and over time in the following major product lines. Following the classification of Diagnostics - sale of precision oncology services as a discontinued operation during the year ended December 31, 2025, the following disclosure of revenue has been retrospectively recast to represent results from continuing operations. The disclosure of revenue by product line is consistent with the revenue information that is disclosed for each reportable segment under IFRS 8 Operating Segments ("IFRS 8") (see note 6).
Disaggregation of revenue
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| External revenue by product line | |||||||||||||||||
| Prevention - genetic testing | $ | $ | $ | ||||||||||||||
| Consumer health - health and wellness solutions | |||||||||||||||||
| Consumer health - sports distribution | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| External revenue by timing of revenue | |||||||||||||||||
| Goods transferred at a point in time | $ | $ | $ | ||||||||||||||
| Services transferred at a point in time | |||||||||||||||||
| Services transferred overtime | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Revenue expected to be recognized in the future arising from contracts with customers in existence at the report date
At December 31, 2025, 2024 and 2023, the amount of service fee income allocated to the remaining performance obligations under the Group’s existing contracts that are non-refundable is $3,086 , $6,491 and $6,111 , respectively. The Group will recognize the expected revenue in the future when the performance obligations are fulfilled, which may be after one year from the end of the reporting period. Such amount does not include any variable consideration.
F-28
6 Segment Information
Products and services from which reportable segments derive their revenues
The Group manages its businesses by divisions, which are organized based on the nature of the products and services offered. For management purposes, the Group's operations are reviewed by the Group’s Chief Operating Decision Maker (the "CODM") in accordance with IFRS 8, and resources are allocated based on this structure.
In the prior years, the Group operated three operating segments, namely the Prevention, Diagnostics, and Consumer Health segments.
During the year ended December 31, 2025, the Group disposed of its Diagnostics business, which is presented as discontinued operations in accordance with IFRS 5. As a result of this disposal, the CODM revised the internal reporting structure.
Accordingly, the Group currently has two reportable segments from continuing operations: (i) Prevention segment, and (ii) Consumer Health segment. The Group’s reportable segments are as follows:
1.Prevention being the design and sale of genetic testing; and
2.Consumer Health being provision of consumer health products, including health and wellness products, and fulfillment and distribution services of sports nutrition products.
The following is an analysis of the Group’s revenue and gross profit from continuing operations by reportable segments. Performance is measured based on gross profit from continuing operations, as included in the internal management reports that are reviewed by the CODM:
| Prevention | Consumer Health | Unallocated | Total from continuing operations | ||||||||||||||||||||
| 2025 | |||||||||||||||||||||||
| Revenue | $ | $ | $ | $ | |||||||||||||||||||
| Gross profit | |||||||||||||||||||||||
| 2024 (Restated) | |||||||||||||||||||||||
| Revenue | |||||||||||||||||||||||
| Gross profit | |||||||||||||||||||||||
| 2023 (Restated) | |||||||||||||||||||||||
| Revenue | n/a | ||||||||||||||||||||||
| Gross profit/(loss) | n/a | ( | |||||||||||||||||||||
F-29
6 Segment Information (continued)
During the years ended December 31, 2025, 2024 and 2023, the Prevention segment within continuing operations incurred processing service fees of approximately $2,312 , $2,968 , and $1,498 , respectively, in respect of processing services provided by the Diagnostics business presented within discontinued operations (see note 10). In preparing the Group’s consolidated financial statements, the processing service revenue recognized by the Diagnostics business and the corresponding processing service expense recognized by the Prevention segment were eliminated in full on consolidation. As the Diagnostics business is presented within discontinued operations while the Prevention segment remains within continuing operations in the re-presented comparative information, this elimination reduces revenue in discontinued operations and reduces costs in continuing operations. Accordingly, the effect of such intragroup transactions is not reflected on a gross basis within continuing operations in the re-presented comparative information. Such processing service arrangement is expected to continue after the disposal of the Diagnostics business pursuant to ongoing commercial arrangements. Including the impact of such processing service fees, the adjusted gross profit of the Prevention segment from continuing operations would have been approximately $8,686 , $5,154 and $1,617 , respectively.
Unallocated direct costs represent the unallocated depreciation and amortization related to property, plant and equipment and intangible assets commonly used across segments, and certain staff costs commonly worked across segments.
Segment revenue from continuing operations reported above represents revenue generated from external customers. There were no
No analysis of the Group’s assets and liabilities by reportable segment is disclosed as they are not regularly provided to the CODM for review.
Segment gross profit represents the gross profit earned by each segment without allocation of certain direct costs associated with depreciation and amortization and certain staff costs commonly used across segments. This is the measure reported to the CODM for the purposes of resource allocation and performance assessment.
The following is the reconciliation of the reportable segments' gross profit from continuing operation to the loss before taxation for the years ended December 31, 2025, 2024 and 2023.
F-30
6 Segment Information (continued)
| 2025 | 2024 | 2023 | ||||||||||||||||||
| (Restated) | (Restated) | |||||||||||||||||||
| Gross profit | $ | $ | $ | |||||||||||||||||
| Other income and other net gains | ||||||||||||||||||||
| Selling and marketing expenses | ( | ( | ( | |||||||||||||||||
| Research and development expenses | ( | ( | ( | |||||||||||||||||
| Impairment of goodwill | ( | |||||||||||||||||||
| Administrative and other operating expenses | ( | ( | ( | |||||||||||||||||
| Fair value gain/(loss) on financial assets at fair value through profit or loss | ( | ( | ||||||||||||||||||
| Gain on warrant exchange | ||||||||||||||||||||
| Fair value (loss)/gain on warrant liabilities | ( | |||||||||||||||||||
| Unrealized fair value loss on digital assets | ( | |||||||||||||||||||
| Gain on partial disposal of an equity-accounted investee | ||||||||||||||||||||
| Share of loss of equity-accounted investees, net of tax | ( | ( | ( | |||||||||||||||||
| Other finance costs | ( | ( | ( | |||||||||||||||||
| Loss before taxation | $ | ( | $ | ( | $ | ( | ||||||||||||||
Revenues from major products and services
The Group’s revenues from continuing operations from its major products and services are disclosed in note 5.
F-31
6 Segment Information (continued)
Geographical information
Information about the Group's revenue from continuing operations from external customers is presented based on the location of the region of domicile of entities of the Group.
| Revenue from external customers | |||||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Hong Kong | $ | $ | $ | ||||||||||||||
| The United States | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Information about the Group's non-current assets is presented based on the geographical location of the assets.
| Non-current assets | |||||||||||
| 2025 | 2024 | ||||||||||
| Hong Kong | $ | $ | |||||||||
| The United States | |||||||||||
| United Kingdom | |||||||||||
| Taiwan | |||||||||||
| Rest of the world | |||||||||||
| $ | $ | ||||||||||
Non-current assets exclude those relating to financial assets at fair value through profit or loss.
Information about major customers
No single customer from continuing operations contributed 10% or more to the Group's revenue from continuing operations in 2025, 2024 and 2023.
F-32
7 Other income and other net gains
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Government subsidies (notes) | $ | $ | $ | ||||||||||||||
| Bank interest income | |||||||||||||||||
| Dividend income | |||||||||||||||||
| Net foreign exchange (loss)/gain | ( | ||||||||||||||||
| Sundry income | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Notes: The Group has recognized various subsidies granted by the governments in different jurisdictions, including:
(i)funding support of $151 under the Research Talent Hub program of Innovation and Technology Fund, administered by the Innovation and Technology Commission of The Government of Hong Kong Special Administrative Region during the year ended December 31, 2025. The Research Talent Hub aims to provide funding support for companies undertaking research and development projects funded by the Innovation and Technology Fund to engage research talent to conduct research and development work.
(ii)funding support of $72 and $36 from the Employment Support Scheme (the “ESS”) and the Job Creation Scheme from the Hong Kong Institute of Human Resource Management (the “HKIHRM”) under the Anti-epidemic Fund set up by The Government of Hong Kong Special Administrative Region during the year ended December 31, 2023, respectively. The purpose of the funding was to provide financial support to enterprises to retain their employees who would otherwise be made redundant. Under the ESS, the Group was required not to make redundancies during the subsidy period and to spend all the funding on paying wages to the employees; and Under the HKIHRM, employers who are HKIHRM members which created job positions are eligible to apply for salary government grants.
8 Loss for the year
Loss for the year has been arrived at after charging:
(a)Other finance costs
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Interest expenses on lease liabilities | $ | $ | $ | ||||||||||||||
F-33
8 Loss for the year (continued)
(b)Staff costs
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Salaries, wages and other benefits | $ | $ | $ | ||||||||||||||
| Contributions to defined contribution retirement plan | |||||||||||||||||
| Equity-settled share-based payment expenses | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| Represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Selling and marketing expenses | |||||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total staff costs | $ | $ | $ | ||||||||||||||
(c)Other items
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Cost of inventories | $ | $ | $ | ||||||||||||||
| Depreciation of | |||||||||||||||||
| - property, plant and equipment | |||||||||||||||||
| - right-of-use assets | |||||||||||||||||
| Amortization of intangible assets | |||||||||||||||||
| Write-off on property, plant and equipment | |||||||||||||||||
| Auditor’s remuneration | |||||||||||||||||
| Freight and delivery charges | |||||||||||||||||
Total depreciation and amortization charges represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total depreciation and amortization charges | $ | $ | $ | ||||||||||||||
F-34
9 Income tax expense/(credit)
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Current tax - Hong Kong Profits Tax | |||||||||||||||||
| Overprovisions related to prior years | $ | $ | ( | $ | ( | ||||||||||||
| Current tax - Overseas | |||||||||||||||||
| Underprovisions related to prior years | |||||||||||||||||
| Current year | ( | ||||||||||||||||
| Deferred tax | |||||||||||||||||
| Origination and reversal of temporary differences | ( | ( | |||||||||||||||
| Tax expense/(credit) | $ | $ | ( | $ | |||||||||||||
The provision for Hong Kong Profits Tax is calculated by applying the estimated annual effective tax rate of 16.5 one
The U.S. federal statutory corporate income tax rate is calculated at 21 % for the year ended December 31, 2025.
Taxation for other overseas subsidiaries and branch is charged at the appropriate current rates of taxation ruling in the relevant countries.
The credit for the year can be reconciled to the loss before taxation as follows:
| 2025 | 2024 | 2023 | |||||||||||||||
| (Restated) | (Restated) | ||||||||||||||||
| Continuing operations | |||||||||||||||||
| Loss before taxation | $ | ( | $ | ( | $ | ( | |||||||||||
| Tax at average income tax rate (Note) | $ | ( | $ | ( | $ | ( | |||||||||||
| Tax effect of non-deductible expenses | |||||||||||||||||
| Tax effect of non-taxable income | ( | ( | ( | ||||||||||||||
| Tax effect on utilization of previously unrecognized tax losses | ( | ||||||||||||||||
| Under/(over) provision in respect of prior years | ( | ||||||||||||||||
| Tax effect of tax losses not recognized | |||||||||||||||||
| Tax effect of deductible temporary differences not recognized | |||||||||||||||||
| Tax effect of previously unrecognized temporary differences recognized in current year | ( | ( | |||||||||||||||
| Others | |||||||||||||||||
| $ | $ | ( | $ | ||||||||||||||
Note: The average income tax rates for the years ended December 31, 2025, 2024 and 2023 represent the weighted average tax rate of the operations in different jurisdictions on the basis of the relative amounts of loss before taxation and the relevant statutory rates.
F-35
10 Discontinued operations
In 2023, the Group discontinued its 2019 novel coronavirus (“COVID-19”) related diagnostic services globally and other DNA testing operations in the EMEA region. This strategic decision was influenced by the World Health Organization's latest pronouncements and the diminishing demand for COVID-19 diagnostic services, primarily attributable to the relaxation of testing mandates for international travelers and local citizens worldwide. Consequently, these developments led to a noticeable decline in the number of testing centers operating in Hong Kong and the United Kingdom for testing samples from the EMEA region.
On October 1, 2025, the Group completed the disposal of ACT Genomics. The disposal was part of a strategic shift to streamline operations and exit the clinical diagnostics sector, enabling the Group to focus its resources and capital on the high-growth consumer health market and its core flagship brands. Following the disposal, the Group no longer has exposure to the operations of ACT Genomics. Consequently, the results of ACT Genomics have been classified as discontinued operations in the consolidated financial statements for all periods presented. The results of discontinued operations for the years ended December 31, 2024, 2023, and related disclosures in notes 5, 6, 7, 8 and 9 have also been restated accordingly.
Together with the earlier cessation of COVID-19 related diagnostic services and other DNA testing operations in the EMEA region, these actions reflect management’s continued strategic realignment to streamline operations and reallocate resources toward its core businesses in response to evolving market conditions.
In accordance with IFRS 5, the above cessation and disposal represent the discontinuation and disposal of components of the Group that constitute separate major lines of business and/or geographical areas of operations, and therefore met the criteria to be classified as discontinued operations as of the respective dates of cessation and disposal.
Results of discontinued operations
The following is a reconciliation of the amounts of major classes of loss from operations classified as discontinued operations in the consolidated statements of comprehensive loss for the years ended December 31, 2025, 2024, and 2023:
| 2025 | 2024 | 2023 | |||||||||||||||
| Revenue | $ | $ | $ | ||||||||||||||
| Direct costs | ( | ( | ( | ||||||||||||||
| Gross profit | |||||||||||||||||
| Other income and other net gains | |||||||||||||||||
| Selling and marketing expenses | ( | ( | ( | ||||||||||||||
| Research and development expenses | ( | ( | ( | ||||||||||||||
| Impairment loss of goodwill | ( | ||||||||||||||||
| Administrative and other operating expenses | ( | ( | ( | ||||||||||||||
| Loss from operating activities | ( | ( | ( | ||||||||||||||
| Share of (loss)/profit of equity-accounted investees, net of tax | ( | ( | |||||||||||||||
| Gain on disposal of a subsidiary (note 34) | |||||||||||||||||
| Other finance costs | ( | ( | ( | ||||||||||||||
| Loss from discontinued operations before taxation | ( | ( | ( | ||||||||||||||
| Income tax credit | |||||||||||||||||
| Loss from discontinued operations, net of tax | $ | ( | $ | ( | $ | ( | |||||||||||
| Loss per share | |||||||||||||||||
| Basic | ( | ( | ( | ||||||||||||||
| Diluted | ( | ( | ( | ||||||||||||||
F-36
10 Discontinued operations (continued)
During the years ended December 31, 2025, 2024 and 2023, the Diagnostics business within discontinued operations recognized processing service income of approximately $2,312 , $2,968 , and $1,498 , respectively, in respect of processing services provided to the Prevention segment presented within continuing operations (see note 6). In preparing the Group’s consolidated financial statements, the processing service revenue recognized by the Diagnostics business and the corresponding processing service expense recognized by the Prevention segment were eliminated in full on consolidation. As the Diagnostics business is presented within discontinued operations while the Prevention segment remains within continuing operations in the re-presented comparative information, this elimination reduces revenue in discontinued operations and reduces costs in continuing operations. Accordingly, the effect of such intragroup transactions is not reflected on a gross basis within discontinued operations in the re-presented comparative information. Such processing service arrangement is expected to continue after the disposal of the Diagnostics business pursuant to ongoing commercial arrangements. Including the impact of such processing service income, the adjusted loss from discontinued operations, net of tax, would have been approximately $1,419 , $8,452 and $20,281 , respectively.
The loss from discontinued operations attributable to the equity shareholders of the Company amounted to $3,173 , $7,086 , and $20,155 , while loss attributable to non-controlling interests amounted to $558 , $4,334 , and $1,624 for the years ended December 31, 2025, 2024 and 2023, respectively.
The total comprehensive expense from continuing operations of $34,169 , $39,045 , and $41,493 was attributable entirely to the equity shareholders of the Company for the years ended December 31, 2025, 2024 and 2023, respectively.
(a)Revenue
Disaggregation of revenue
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by product line | |||||||||||||||||
| Prevention and diagnostics - COVID-19 related and EMEA operations | $ | $ | $ | ||||||||||||||
| Diagnostics - sale of precision oncology services | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by timing of revenue | |||||||||||||||||
| Goods transferred at a point in time | $ | $ | $ | ||||||||||||||
| Services transferred at a point in time | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Geographical information
Information about the Group's revenue from discontinued operations from external customers is presented based on the location of the region of domicile of entities of the Group.
| 2025 | 2024 | 2023 | |||||||||||||||
| Hong Kong | $ | $ | $ | ||||||||||||||
| United Kingdom | |||||||||||||||||
| Taiwan | |||||||||||||||||
| Rest of the world | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
F-37
10 Discontinued operations (continued)
(b)Other income and other net gains/(losses)
| 2025 | 2024 | 2023 | |||||||||||||||
| Bank interest income | $ | $ | $ | ||||||||||||||
| Net foreign exchange gains/(losses) | ( | ||||||||||||||||
| Sundry income | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
(c)Other finance costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Interest expenses on lease liabilities | $ | $ | $ | ||||||||||||||
(d)Staff costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Salaries, wages and other benefits | $ | $ | $ | ||||||||||||||
| Contributions to defined contribution retirement plan | |||||||||||||||||
| Equity-settled share-based payment expenses | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| Represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Selling and marketing expenses | |||||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total staff costs | $ | $ | $ | ||||||||||||||
(e)Other items
| 2025 | 2024 | 2023 | |||||||||||||||
| Cost of inventories | $ | $ | $ | ||||||||||||||
| Depreciation of | |||||||||||||||||
| - property, plant and equipment | |||||||||||||||||
| - right-of-use assets | |||||||||||||||||
| Amortization of intangible assets | |||||||||||||||||
| Write-off on property, plant and equipment | |||||||||||||||||
| Auditor’s remuneration | |||||||||||||||||
| Freight and delivery charges | |||||||||||||||||
Total depreciation and amortization charges represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total depreciation and amortization charges | $ | $ | $ | ||||||||||||||
F-38
10 Discontinued operations (continued)
Cash flows from/(used in) discontinued operations
| 2025 | 2024 | 2023 | |||||||||||||||
| Net cash from/(used in) operating activities | $ | $ | $ | ( | |||||||||||||
| Net cash used in investing activities | ( | ( | ( | ||||||||||||||
| Net cash used in financing activities | ( | ( | ( | ||||||||||||||
| Net cash (outflows)/inflows for the year | $ | ( | $ | $ | ( | ||||||||||||
A.COVID-19 diagnostic services and other DNA testing operations in the EMEA region
| 2025 | 2024 | 2023 | |||||||||||||||
| Revenue | $ | $ | $ | ||||||||||||||
| Direct costs | ( | ( | |||||||||||||||
| Gross profit | |||||||||||||||||
| Other income and other net gains | |||||||||||||||||
| Selling and marketing expenses | ( | ( | |||||||||||||||
| Research and development expenses | ( | ||||||||||||||||
| Administrative and other operating expenses | ( | ( | ( | ||||||||||||||
| Profit/(loss) from operating activities | ( | ||||||||||||||||
| Other finance costs | ( | ||||||||||||||||
| Profit/(loss) from discontinued operations before taxation | ( | ||||||||||||||||
| Income tax credit | |||||||||||||||||
| Profit/(loss) from discontinued operations, net of tax | $ | $ | $ | ( | |||||||||||||
| Earnings/(losses) per share | |||||||||||||||||
| Basic | $ | $ | $ | ( | |||||||||||||
| Diluted | ( | ||||||||||||||||
F-39
10 Discontinued operations (continued)
(a)Revenue
Disaggregation of revenue
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by product line | |||||||||||||||||
| Prevention and diagnostics - COVID-19 related and EMEA operations | $ | $ | $ | ||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by timing of revenue | |||||||||||||||||
| Goods transferred at a point in time | $ | $ | $ | ||||||||||||||
| Services transferred at a point in time | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Geographical information
Information about the Group's revenue from discontinued operations from external customers is presented based on the location of the region of domicile of entities of the Group.
| 2025 | 2024 | 2023 | |||||||||||||||
| Hong Kong | $ | $ | $ | ||||||||||||||
| United Kingdom | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
(b)Other income and other net gains
| 2025 | 2024 | 2023 | |||||||||||||||
| Bank interest income | $ | $ | $ | ||||||||||||||
| Net foreign exchange (losses)/gains | ( | ( | |||||||||||||||
| Sundry income | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
(c)Other finance costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Interest expenses on lease liabilities | $ | $ | $ | ||||||||||||||
(d)Staff costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Salaries, wages and other benefits | $ | $ | $ | ||||||||||||||
| Contributions to defined contribution retirement plan | |||||||||||||||||
| Equity-settled share-based payment expenses | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| Represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Selling and marketing expenses | |||||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total staff costs | $ | $ | $ | ||||||||||||||
F-40
10 Discontinued operations (continued)
(e)Other items
| 2025 | 2024 | 2023 | |||||||||||||||
| Cost of inventories | $ | $ | $ | ||||||||||||||
| Depreciation of | |||||||||||||||||
| - property, plant and equipment | |||||||||||||||||
| - right-of-use assets | |||||||||||||||||
| Amortization of intangible assets | |||||||||||||||||
| Write-off on property, plant and equipment | |||||||||||||||||
| Auditor’s remuneration | |||||||||||||||||
| Freight and delivery charges | |||||||||||||||||
Total depreciation and amortization charges represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total depreciation and amortization charges | $ | $ | $ | ||||||||||||||
Cash flows from/(used in) discontinued operations
| 2025 | 2024 | 2023 | |||||||||||||||
| Net cash from/(used in) operating activities | $ | $ | $ | ( | |||||||||||||
| Net cash (used in)/from investing activities | ( | ||||||||||||||||
| Net cash used in financing activities | ( | ||||||||||||||||
| Net cash inflows/(outflows) for the year | $ | $ | $ | ( | |||||||||||||
F-41
10 Discontinued operations (continued)
B. ACT Genomics
| 2025 | 2024 | 2023 | |||||||||||||||
| Revenue | $ | $ | $ | ||||||||||||||
| Direct costs | ( | ( | ( | ||||||||||||||
| Gross profit | |||||||||||||||||
| Other income and other net gains | |||||||||||||||||
| Selling and marketing expenses | ( | ( | ( | ||||||||||||||
| Research and development expenses | ( | ( | ( | ||||||||||||||
| Impairment loss of goodwill | ( | ||||||||||||||||
| Administrative and other operating expenses | ( | ( | ( | ||||||||||||||
| Loss from operating activities | ( | ( | ( | ||||||||||||||
| Share of (loss)/profit of equity-accounted investees, net of tax | ( | ( | |||||||||||||||
| Gain on disposal of a subsidiary (note 34) | |||||||||||||||||
| Other finance costs | ( | ( | ( | ||||||||||||||
| Loss from discontinued operations before taxation | ( | ( | ( | ||||||||||||||
| Income tax credit | |||||||||||||||||
| Loss from discontinued operations, net of tax | $ | ( | $ | ( | $ | ( | |||||||||||
| Loss per share | |||||||||||||||||
| Basic | ( | ( | ( | ||||||||||||||
| Diluted | ( | ( | ( | ||||||||||||||
(a)Revenue
Disaggregation of revenue
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by product line | |||||||||||||||||
| Diagnostics - sale of precision oncology services | $ | $ | $ | ||||||||||||||
| 2025 | 2024 | 2023 | |||||||||||||||
| External revenue by timing of revenue | |||||||||||||||||
| Services transferred at a point in time | $ | $ | $ | ||||||||||||||
Geographical information
Information about the Group's revenue from discontinued operations from external customers is presented based on the location of the region of domicile of entities of the Group.
| 2025 | 2024 | 2023 | |||||||||||||||
| Hong Kong | $ | $ | $ | ||||||||||||||
| United Kingdom | |||||||||||||||||
| Taiwan | |||||||||||||||||
| Rest of the world | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
F-42
10 Discontinued operations (continued)
(b)Other income and other net gains
| 2025 | 2024 | 2023 | |||||||||||||||
| Bank interest income | $ | $ | $ | ||||||||||||||
| Net foreign exchange gains/(losses) | ( | ||||||||||||||||
| Sundry income | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
(c)Other finance costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Interest expenses on lease liabilities | $ | $ | $ | ||||||||||||||
(d)Staff costs
| 2025 | 2024 | 2023 | |||||||||||||||
| Salaries, wages and other benefits | $ | $ | $ | ||||||||||||||
| Contributions to defined contribution retirement plan | |||||||||||||||||
| Equity-settled share-based payment expenses | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| Represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Selling and marketing expenses | |||||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total staff costs | $ | $ | $ | ||||||||||||||
(e)Other items
| 2025 | 2024 | 2023 | |||||||||||||||
| Cost of inventories | $ | $ | $ | ||||||||||||||
| Depreciation of | |||||||||||||||||
| - property, plant and equipment | |||||||||||||||||
| - right-of-use assets | |||||||||||||||||
| Amortization of intangible assets | |||||||||||||||||
| Write-off on property, plant and equipment | |||||||||||||||||
| Auditor’s remuneration | |||||||||||||||||
| Freight and delivery charges | |||||||||||||||||
Total depreciation and amortization charges represented by: | |||||||||||||||||
| Direct costs | $ | $ | $ | ||||||||||||||
| Research and development expenses | |||||||||||||||||
| Administrative and other operating expenses | |||||||||||||||||
| Total depreciation and amortization charges | $ | $ | $ | ||||||||||||||
F-43
10 Discontinued operations (continued)
Cash flows (used in)/from discontinued operations
| 2025 | 2024 | 2023 | |||||||||||||||
| Net cash (used in)/from operating activities | $ | ( | $ | $ | ( | ||||||||||||
| Net cash used in investing activities | ( | ( | ( | ||||||||||||||
| Net cash used in financing activities | ( | ( | ( | ||||||||||||||
| Net cash (outflows)/inflows for the year | $ | ( | $ | $ | ( | ||||||||||||
11 Loss per share
From continuing and discontinued operations
The calculation of the basic and diluted loss per share is based on the following data:
| 2025 | 2024 | 2023 | |||||||||||||||
| Loss | |||||||||||||||||
| Loss for the purpose of basic and diluted loss per share being net loss attributable to equity shareholders of the Company | |||||||||||||||||
| - from continuing operations | $ | ( | $ | ( | $ | ( | |||||||||||
| - from discontinued operations | ( | ( | ( | ||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||
| Number of shares | |||||||||||||||||
| Weighted-average number of ordinary shares for the purposes of basic and diluted loss per share | |||||||||||||||||
At December 31, 2025, 489,088 shares underlying restricted share units (RSUs), 1,492,307 shares underlying warrants, and 3,216,617 shares underlying private placement warrants were excluded from the diluted weighted-average number of ordinary shares calculation because their effect would have been anti-dilutive. At December 31, 2024, 1,214,951 shares underlying RSUs and 1,492,307 shares underlying warrants were excluded from the diluted weighted-average number of ordinary shares calculation because their effect would have been anti-dilutive. At December 31, 2023, 859,331 shares underlying RSUs and 1,492,307 shares underlying warrants were excluded from the diluted weighted-average number of ordinary shares calculation because their effect would have been anti-dilutive.
F-44
12 Goodwill
| At January 1, 2024 | $ | ||||
| Recognized on acquisition of a subsidiary | |||||
| At December 31, 2024 and January 1, 2025 | |||||
| Eliminated on disposal of a subsidiary | ( | ||||
| Impairment loss recognized | ( | ||||
| At December 31, 2025 | $ | ||||
Impairment tests for cash-generating units (“CGU”) containing goodwill
The Group tests goodwill annually for impairment, or more frequently if there are indications that goodwill might be impaired.
E-commerce and retail logistics for sports distribution business within the consumer health segment
The goodwill associated with e-commerce and retail logistics for sports distribution business within the consumer health segment arose when that business was acquired by the Group on August 9, 2024. It comprises of a CGU responsible for the related operations based in the United States.
Cancer genetic testing services within the diagnostics segment
The goodwill associated with cancer genetic testing services within the diagnostics segment arose when that business was acquired by the Group on December 30, 2022. It comprises of a CGU responsible for the related operations based in Hong Kong, Taiwan and Thailand.
Sales of medical diagnostics products within the diagnostics segment
The goodwill associated with sales of medical diagnostics products within the diagnostics segment arose when that business was acquired by the Group on December 30, 2022. It represents a CGU responsible for the related operations based in the United Kingdom. Following the disposal of the medical diagnostics product business in the United Kingdom during 2023, the associated goodwill has been derecognized in its entirety as of the disposal date.
The carrying amount of goodwill has been allocated to CGUs as follows:
| 2025 | 2024 | ||||||||||
| E-commerce and retail logistics for sports distribution business within the consumer health segment | $ | $ | |||||||||
| Cancer genetic testing services within the diagnostics segment | |||||||||||
| Sales of medical diagnostics products within the diagnostics segment | |||||||||||
| $ | $ | ||||||||||
CGUs of e-commerce and retail logistics for sports distribution business
At December 31, 2024, the recoverable amount of the CGU of e-commerce and retail logistics for sports distribution business was determined based on value-in-use calculations. These calculations used cash flow projections based on financial budgets approved by management covering five years period and pre-tax discount rate of 18.1 %. Cash flows beyond the forecast period were extrapolated using a steady 3.0 % growth rate. The values assigned to the key assumptions represent management’s assessment of future trends in the relevant industries and were based on historical data from external and internal sources. The recoverable amounts of the CGUs of e-commerce and retail logistics for sports distribution based on the estimated value-in-use calculations was higher than the carrying amounts of the respective CGUs. Accordingly, the management of the Group considered no impairment for goodwill.
At December 31, 2025, the CGU of e-commerce and retail logistics for sports distribution business was determined to be impaired and goodwill of $6,815 was recognized in profit or loss. The recoverable amount of the CGU of e-commerce and retail logistics for sports distribution business based on fair value less costs of disposal, reflecting management’s strategic decision during the year to divest the business as set out in note 39.
F-45
12 Goodwill (continued)
Fair value less costs of disposal was determined using valuation techniques based on market participant assumptions. The valuation incorporated a probability-weighted assessment of expected future consideration, including both fixed and contingent share consideration linked to the achievement of specified performance milestones. The contingent share consideration is subject to multiple operational and commercial performance targets, including shipment volume–based targets and referral revenue generated under post-closing arrangements. The valuation therefore required management to estimate the likelihood of achieving the relevant milestones and the associated expected value of consideration.
Management also considered value in use based on cash flow projections derived from financial budgets approved by management covering five years period, using a pre-tax discount rate of 18.1 % and a terminal growth rate of 3.0 %. However, the value in use calculation resulted in a lower recoverable amount compared to fair value less costs of disposal, and accordingly fair value less costs of disposal was used as the recoverable amount.
The determination of the recoverable amount involved significant judgement, particularly in assessing the appropriate valuation methodology and estimating key assumptions, including the probability of achieving performance milestones and the expected timing and amount of consideration. Changes in these assumptions could result in a material change in the impairment assessment.
Based on the assessment, the recoverable amount of the CGU was determined to be lower than its carrying amount, and accordingly an impairment loss was recognized.
CGUs of cancer genetic testing services and sales of medical diagnostics products
At December 31, 2024, the recoverable amounts of the CGU of cancer genetic testing services and the CGU of sales of medical diagnostics products were determined based on value-in-use calculations. These calculations used cash flow projections based on financial budgets approved by management covering a five years period and pre-tax discount rate of 20.0 % and 17.0 %. The projections for periods greater than five years have been used on the basis that a longer projection period facilitates a comprehensive evaluation of the future prospects and stability of both CGUs, allowing for a thorough consideration of potential technological advancements that could impact the long-term performance. Cash flows beyond the period of five years are extrapolated using a steady growth rate. The values assigned to the key assumptions represent management’s assessment of future trends in the relevant industries and are based on historical data from external and internal sources.
The recoverable amounts of the CGUs of cancer genetic testing services and sales of medical diagnostics products based on the estimated value-in-use calculations were higher than the carrying amounts of the respective CGUs. Accordingly, the management of the Group considered no impairment for goodwill as of December 31, 2024. The management of the Group determines that there is no reasonable possible change in the key parameters that could cause the carrying amount of the groups of CGUs to exceed the recoverable amount.
On October 1, 2025, the Group disposed of the CGUs of cancer genetic testing services and and sales of medical diagnostics products (the “Disposed CGUs”). As a result of the disposal, the Disposed CGUs ceased to be included in the Group’s cash-generating units for impairment assessment purposes subsequent to that date. Up to the disposal date, no impairment loss was recognized prior to disposal as the recoverable amount exceeded the carrying amount of the Disposed CGUs.
F-46
13 Property, plant and equipment
Right-of-use assets (note (a)) | Leasehold improvements | Fixtures and furniture | Office and lab equipment | Computer equipment | Motor vehicles | Manufacturing equipment | Total | ||||||||||||||||||||||||||||||||||||||||
| Cost: | |||||||||||||||||||||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| Additions | |||||||||||||||||||||||||||||||||||||||||||||||
| Additions from acquisition (note 33(III)) | |||||||||||||||||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||
| Exchange differences | ( | ( | ( | ( | ( | ( | |||||||||||||||||||||||||||||||||||||||||
| At December 31, 2024 and January 1, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||
| Additions | |||||||||||||||||||||||||||||||||||||||||||||||
| Disposal of a subsidiary (note 34) | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Disposals | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Exchange differences | |||||||||||||||||||||||||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| Accumulated depreciation: | |||||||||||||||||||||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| Charge for the year | |||||||||||||||||||||||||||||||||||||||||||||||
| Eliminated on disposal | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Exchange differences | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| At December 31, 2024 and January 1, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||
| Charge for the year | |||||||||||||||||||||||||||||||||||||||||||||||
| Eliminated on disposal of a subsidiary (note 34) | ( | ( | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||
| Eliminated on disposal | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Written off | ( | ( | ( | ||||||||||||||||||||||||||||||||||||||||||||
| Exchange differences | |||||||||||||||||||||||||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| Carrying amounts: | |||||||||||||||||||||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| At December 31, 2024 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||||||||||
F-47
13 Property, plant and equipment (continued)
(a)Right-of-use assets
The analysis of the carrying amount of right-of-use assets by class of underlying asset is as follows:
| Note | 2025 | 2024 | ||||||||||||
| Properties leased for own use, carried at depreciated cost | (i) | $ | $ | |||||||||||
| Office equipment, carried at depreciated cost | (ii) | |||||||||||||
| $ | $ | |||||||||||||
The analysis of expense items in relation to leases recognized in profit or loss is as follows:
| 2025 | 2024 | 2023 | |||||||||||||||
| Depreciation charge of right-of-use assets by class of underlying asset: | |||||||||||||||||
| - Properties leased for own use | $ | $ | $ | ||||||||||||||
| - Office equipment | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
| Interest on lease liabilities (notes 8(a) and 10(c)) | $ | $ | $ | ||||||||||||||
| Expense relating to short-term leases or leases of low-value assets | |||||||||||||||||
During the years ended December 31, 2025, 2024 and 2023, additions to right-of-use assets of $37 , $3,094 and $197 , respectively, mainly resulted from the capitalization of lease payments payable under new tenancy agreements.
Details of the maturity analysis of lease liabilities are set out in note 25.
(i)Properties leased for own use
The Group has obtained the right to use some properties as its warehouses and offices through tenancy agreements. The leases typically run for an initial period of 2 to 5 years (2024: 2 to 5 years) with only fixed lease payments. The lease agreements do not impose any covenants other than the security interests in the leased assets that are held by the lessor. Leased assets may not be used as security for borrowing purposes.
Some leases include an option to renew the lease for an additional period after the end of the contract term. Where practicable, the Group seeks to include such extension options exercisable by the Group to provide operational flexibility. The Group assesses at lease commencement date whether it is reasonably certain to exercise the extension options, and reassesses whether it is reasonably certain to exercise the options if there is a significant event or significant changes in circumstances within its control. If the Group is not reasonably certain to exercise the extension options, the future lease payments during the extension periods are not included in the measurement of lease liabilities. The potential exposure to future lease payments in relation to such leases are assessed as insignificant.
(ii)Office equipment
The Group leases office equipment under a lease expiring in 5 years. The lease does not include an option to renew the lease or purchase the leased equipment at the end of the lease term at a price deemed to be a bargain purchase option. The lease does not include variable lease payments.
F-48
13 Property, plant and equipment (continued)
(b)Amounts recognized in consolidated statement of cash flows
Amounts included in the consolidated statement of cash flows for leases comprise the following:
| 2025 | 2024 | 2023 | |||||||||||||||
| Within operating cash flows | $ | ( | $ | ( | $ | ( | |||||||||||
| Within financing cash flows | ( | ( | ( | ||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||
F-49
14 Intangible assets
Website and mobile apps | Trademark and technology | Computer software | Customer relationship | Total | |||||||||||||||||||||||||
| Cost: | |||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Additions | |||||||||||||||||||||||||||||
| Additions from acquisition (note 33(III)) | |||||||||||||||||||||||||||||
| Disposals | ( | ( | |||||||||||||||||||||||||||
| Written off | ( | ( | |||||||||||||||||||||||||||
| Exchange differences | ( | ||||||||||||||||||||||||||||
| At December 31, 2024 and January 1, 2025 | |||||||||||||||||||||||||||||
| Disposal of a subsidiary (note 34) | ( | ( | ( | ( | |||||||||||||||||||||||||
| Exchange differences | ( | ||||||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Accumulated amortization: | |||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Charge for the year | |||||||||||||||||||||||||||||
| Eliminated on disposal | ( | ( | |||||||||||||||||||||||||||
| Written off | ( | ( | |||||||||||||||||||||||||||
| Exchange differences | ( | ||||||||||||||||||||||||||||
| At December 31, 2024 and January 1, 2025 | |||||||||||||||||||||||||||||
| Charge for the year | |||||||||||||||||||||||||||||
| Eliminated on disposal of a subsidiary (note 34) | ( | ( | ( | ( | |||||||||||||||||||||||||
| Exchange differences | ( | ||||||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Carrying amounts: | |||||||||||||||||||||||||||||
| At January 1, 2024 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| At December 31, 2024 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
F-50
15 Digital assets
The Group's holdings of digital assets consist of the following:
| 2025 | 2024 | ||||||||||
| Digital assets - Bitcoin | $ | $ | |||||||||
The movement in the Group's digital assets holdings during the year ended December 31, 2025 was as follows:
| 2025 | |||||||||||
| Number of Bitcoin | Amount | ||||||||||
| Balance at January 1 | $ | ||||||||||
| Acquisition of digital assets | |||||||||||
| Revaluation adjustments | ( | ||||||||||
| Balance at December 31 | $ | ||||||||||
Digital assets held are revalued each reporting period based on the fair market value being the trading price of Bitcoin on the reporting date. At December 31, 2025, the market price used for Bitcoin was $88 , resulting in total revaluation loss of $9,725 .
Digital assets were revalued at fair value using the quoted prices on Coinbase, which the Group used as its principal market for determining fair value during the year ended December 31, 2025. The Group believes the price difference between the principal market and other comparative active exchanges to be immaterial.
16 Interests in equity-accounted investees
| 2025 | 2024 | ||||||||||
| Interests in associates | $ | $ | |||||||||
Particulars of equity-accounted investees of the Group are as follows:
| Name of an equity-accounted investee | Place of incorporation/ operation | Particular of issued and paid-up capital | Proportion of nominal value of issue capital held by the Company | Principal activity | |||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | ||||||||||||||||||||||||||||||||||||||||
| Directly | Indirectly | Directly | Indirectly | ||||||||||||||||||||||||||||||||||||||
| % | % | % | % | ||||||||||||||||||||||||||||||||||||||
| Insighta | Cayman Islands | Multi-cancer genetic testing services | |||||||||||||||||||||||||||||||||||||||
| CERBACT Asia Holdings Pte. Ltd. (“CERBACT”) | Singapore | Investment holdings | |||||||||||||||||||||||||||||||||||||||
Insighta
Insighta represents an associate in which the Group has a 35 percent ownership interest and joint control under a transactional agreement. Insighta is not publicly listed.
F-51
16 Interests in equity-accounted investees (continued)
On July 20, 2023, the Group acquired 50 % shareholdings of an equity-accounted investee, Insighta. This involved a total consideration of $80,000 contribution in cash to the joint venture and an issuance of 1,481,481 Class A ordinary shares. The fair value of the ordinary shares issued was based on the listed share price of the Company at July 20, 2023 of $0.83 per share (equivalent to $12.51 per share after reverse stock split). The joint venture partner entered into a license agreement with the joint venture that will be automatically terminated upon the occurrence of certain events, including the cessation of business and liquidation of the joint venture.
On October 14, 2024, the Group’s shareholding in Insighta was reduced to 35 % following the disposal of 15 % equity interest in Insighta to a third party with a cash consideration of $30,000 . As such, investment in Insighta was reclassified as interests in associate in 2024. Subsequent to the reporting period, the Group further disposed of its interest in Insighta. Details are set out in note 39.
The following table summarizes the financial information of Insighta, for the years ended December 31, 2025 and 2024, as included in its own financial statements, adjusted for fair value adjustments at acquisition and differences in accounting policies. The table also reconciles the summarized financial information to the carrying amount of the Group's interest in Insighta recognized in the consolidated financial statements.
| 2025 | 2024 | ||||||||||
| Non-current assets | $ | $ | |||||||||
Current assets (including cash and cash equivalents $ | |||||||||||
| Non-current liabilities | ( | ( | |||||||||
| Other payables, accruals and contract liabilities | ( | ( | |||||||||
| Amount due to a related company | ( | ( | |||||||||
| Equity | $ | $ | |||||||||
| Proportion of the Group’s interest in the associate | % | % | |||||||||
| Carrying amount of the Group’s interest in the associate | $ | $ | |||||||||
| 2025 | 2024 | ||||||||||
| Other income | $ | $ | |||||||||
| Other gains or losses | |||||||||||
| Interest income | |||||||||||
| Depreciation and amortization | ( | ( | |||||||||
| Other expenses | ( | ( | |||||||||
| Income tax expense | |||||||||||
| Loss for the year | $ | ( | $ | ( | |||||||
| Item that may be reclassified subsequently to profit or loss: | |||||||||||
| Exchange differences on translation of foreign operations | ( | ||||||||||
| Total comprehensive expense for the year | $ | ( | $ | ( | |||||||
| Group's share of loss and total comprehensive expense | $ | ( | $ | ( | |||||||
| Dividends received by the Group | $ | $ | |||||||||
F-52
16 Interests in equity-accounted investees (continued)
Change in the group’s ownership interest in Insighta
In 2024, the Group disposed of 15 % in Insighta to a third party for proceeds of $30,000 . The Group has accounted for the remaining 35 % as interests in associates. This transaction has resulted in the recognition of a gain in profit or loss, calculated as follows:
| 2024 | |||||
| Proceeds of disposal | $ | ||||
Fair value of interest retained - | |||||
| Carrying amount of interest on the date of disposal | ( | ||||
| Gain recognized | $ | ||||
CERBACT
CERBACT represents an associate and was incorporated on July 12, 2021 as a limited liability company in Singapore. CERBACT is not individually material to the Group and is not publicly listed. The Group’s interest in CERBACT was held indirectly through a subsidiary. Following the disposal of that subsidiary on October 1, 2025, the Group ceased to have an indirect interest in CERBACT, and therefore, the Group’s share of (loss)/profit and comprehensive (expense)/income are disclosed in note 10.
Aggregate information of associates that are not individually material:
| 2025 | 2024 | ||||||||||
| Carrying amount of interest in CERBACT | $ | $ | |||||||||
| Group's share of (loss)/profit and total comprehensive (expense)/income | ( | ||||||||||
17 Other non-current assets
| 2025 | 2024 | ||||||||||
| Deposits and other receivables - non-current | $ | $ | |||||||||
The balances are classified as non-current assets as they are either expected to be (i) recovered or recognized as expense after one year, or (ii) capitalized as property, plant and equipment after the end of the reporting period.
F-53
18 Inventories
| 2025 | 2024 | ||||||||||
| Consumables and reagent | $ | $ | |||||||||
| Work in progress | |||||||||||
| Finished goods | |||||||||||
| $ | $ | ||||||||||
In 2025, 2024 and 2023, inventories of $23,747 , $10,462 and $9,082 , respectively, were recognized as an expense during the year and included in ‘direct costs’.
In addition, inventories have been reduced by $721 and $736 as a result of the write-down to net realizable value for years ended December 31, 2025 and 2024, respectively. Of these amounts, $44 and $75 , respectively, related to discontinued operations. This write-down was recognized as an expense during 2025 and 2024 included in ‘direct costs’.
All inventories are expected to be recovered within one year.
19 Trade and other receivables and deferred expenses
| 2025 | 2024 | ||||||||||
| Current | |||||||||||
| Trade receivables, net of loss allowance | $ | $ | |||||||||
| Deposits, prepayments and other receivables | |||||||||||
| - deposits | |||||||||||
| - prepayments | |||||||||||
| - other receivables - current | |||||||||||
| Deferred expenses (note) | |||||||||||
| $ | $ | ||||||||||
Note: Deferred expenses represent the advanced bonus payment to a director and certain employees for retention purpose under various arrangements. These balances are amortized over the period as stated in the employment agreements and recognized as an expense when the Group consumes the benefit arising from the services provided by the director and those employees in exchange for employee benefits. The amounts expected to be amortized within one year were recognized under current assets.
All trade receivables, deposits, prepayments and other receivables are expected to be recovered or recognized as expense within one year. Trade receivables are due within 30 to 90 days from the date of billing.
Information about the Group’s exposure to credit and market risks, and expected credit loss for trade receivables is included in note 31(C).
F-54
20 Financial assets at fair value through profit or loss
| 2025 | 2024 | ||||||||||
| Financial assets at FVTPL | |||||||||||
| - Investment funds | $ | $ | |||||||||
| - Unlisted securities | |||||||||||
| $ | $ | ||||||||||
Movement of the balance during the years ended December 31, 2025 and 2024 is as follow:
| 2025 | 2024 | ||||||||||
| Balance at January 1 | $ | $ | |||||||||
| Additions | |||||||||||
| Redemptions | ( | ||||||||||
| Changes in fair value recognized in profit or loss | ( | ||||||||||
| Balance at December 31 | $ | $ | |||||||||
| 2025 | 2024 | ||||||||||
| Represented by: | |||||||||||
| Non-current | $ | $ | |||||||||
| Current | |||||||||||
| $ | $ | ||||||||||
21 Short-term deposits and cash and cash equivalents
(a)Short-term deposits
In 2024, the short-term deposits of the Group carried weighted average interest rates of 5.15 % per annum. No
(b)Cash and cash equivalents
| 2025 | 2024 | ||||||||||
| Bank balances | $ | $ | |||||||||
| Cash on hand | |||||||||||
| Cash and cash equivalents | $ | $ | |||||||||
F-55
22 Accrued expenses and other liabilities
| 2025 | 2024 | ||||||||||
| Current | |||||||||||
| Accrued staff costs | $ | $ | |||||||||
| Accrued expenses | |||||||||||
| Accrued professional fee | |||||||||||
| Value added tax payable | |||||||||||
| Deposit liabilities | |||||||||||
| Other payables and accruals | |||||||||||
| Non-current | |||||||||||
| Other non-current liabilities | |||||||||||
| $ | $ | ||||||||||
All of the accrued expenses and other current liabilities are expected to be settled within one year or repayable on demand.
23 Contract liabilities
Contract liabilities represent non-refundable consideration received from customers before the Group recognizes the related revenue. Such consideration is recognized as contract liabilities until the performance obligation is fulfilled or the likelihood of having to fulfil the performance obligation is remote and it is highly probable that a significant reversal of revenue will not occur.
| 2025 | 2024 | ||||||||||
| Contract liabilities | $ | $ | |||||||||
Movement in contract liabilities is as follows:
| 2025 | 2024 | ||||||||||
| Balance at January 1 | $ | $ | |||||||||
| Revenue recognized (Note) | ( | ( | |||||||||
| Receipt from customers upon entering sales contracts | |||||||||||
| Eliminated on disposal of a subsidiary | ( | ||||||||||
| Balance at December 31 | $ | $ | |||||||||
Note: At December 31, 2025 and 2024, the amount of $4,832 and $1,126 , respectively, recognized as revenue from the expiration of unused test kits, which had previously been recorded as contract liabilities.
At December 31, 2025 and 2024, the amount of $2,220 and $2,757 , respectively, are expected to be recognized as revenue within one year .
F-56
24 Deferred tax
(a)Movement in deferred tax balances:
The following are the major deferred tax liabilities and assets recognized by the Group and the movements thereon during the years ended December 31, 2023, 2024 and 2025.
Depreciation allowances in excess of the related depreciation | Tax losses recognized | Intangible assets arising from business combination | Total | ||||||||||||||||||||
| At January 1, 2024 | $ | $ | ( | $ | $ | ||||||||||||||||||
| (Credited)/charged to profit or loss | ( | ( | ( | ||||||||||||||||||||
| Exchange differences | |||||||||||||||||||||||
| At December 31, 2024 and January 1, 2025 | ( | ( | |||||||||||||||||||||
| Credited to profit or loss | ( | ( | ( | ||||||||||||||||||||
| Eliminated on disposal of a subsidiary (note 34) | $ | ( | ( | ||||||||||||||||||||
| Exchange differences | ( | ||||||||||||||||||||||
| At December 31, 2025 | $ | $ | $ | $ | |||||||||||||||||||
(b)Unrecognized deferred tax assets
The Group has not recognized deferred tax assets in respect of cumulative estimated tax losses of $83,265 (2024: $118,477 ) as it is not probable that future taxable profits against which the losses can be utilized will be available in the relevant tax jurisdictions and entities.
The expiry dates of the cumulative tax losses are as follows:
| 2025 | 2024 | ||||||||||
| Within 1 year | $ | $ | |||||||||
| Over 1 year but within 5 years | |||||||||||
| Over 5 years but within 10 years | |||||||||||
| Do not expire under the relevant tax legislation | |||||||||||
| $ | $ | ||||||||||
25 Lease liabilities
The following table shows the remaining contractual maturities of the Group’s lease liabilities at the end of the reporting periods:
| 2025 | 2024 | ||||||||||
| Within 1 year | $ | $ | |||||||||
| After 1 year but within 2 years | |||||||||||
| After 2 years but within 5 years | |||||||||||
| Total | $ | $ | |||||||||
F-57
26 Reconciliation of movements of liabilities to cash flows arising from financing activities
The table below details changes in the Group’s liabilities from financing activities, including both cash and non-cash changes. Liabilities arising from financing activities are liabilities for which cash flows were, or future cash flows will be, classified in the Group’s consolidated statement of cash flows as cash flows from financing activities.
| Lease liabilities | Warrant liabilities | Total | |||||||||||||||
| (note 25) | (note 27) | ||||||||||||||||
| Balance at January 1, 2024 | $ | $ | $ | ||||||||||||||
| Changes from financing cash flows: | |||||||||||||||||
| Capital element of lease rentals paid | ( | ( | |||||||||||||||
| Interest element of lease rentals paid | ( | ( | |||||||||||||||
| Total changes from financing cash flows | ( | ( | |||||||||||||||
| Other changes: | |||||||||||||||||
| Increase in lease liabilities from entering into new leases | |||||||||||||||||
| Additions from acquisition (note 33(III)) | |||||||||||||||||
| Lease modification | ( | ( | |||||||||||||||
| Interest expenses (notes 8(a) and 10(c)) | |||||||||||||||||
| Fair value gain on warrant liabilities | ( | ( | |||||||||||||||
| Total other changes | ( | ||||||||||||||||
| Balance at December 31, 2024 and January 1, 2025 | |||||||||||||||||
| Changes from financing cash flows: | |||||||||||||||||
| Capital element of lease rentals paid | ( | ( | |||||||||||||||
| Interest element of lease rentals paid | ( | ( | |||||||||||||||
| Total changes from financing cash flows | ( | ( | |||||||||||||||
| Other changes: | |||||||||||||||||
| Increase in lease liabilities from entering into new leases | |||||||||||||||||
| Eliminated on disposal of a subsidiary (note 34) | ( | ( | |||||||||||||||
| Interest expenses (notes 8(a) and 10(c)) | |||||||||||||||||
| Issuance of warrants | |||||||||||||||||
| Gain on warrant exchange | ( | ( | |||||||||||||||
| Fair value loss on warrant liabilities | |||||||||||||||||
| Total other changes | ( | ||||||||||||||||
| Balance at December 31, 2025 | $ | $ | $ | ||||||||||||||
F-58
27 Warrant liabilities
The reverse recapitalization included the issuance of 1,492,307 public warrants, each entitling the holder to purchase one Class A ordinary share of the Company at an exercise price of $8.91 per whole share (equivalent to $133.65 per whole share after reverse stock split). The warrants became exercisable on May 18, 2022 and will expire on May 18, 2027. The warrants are listed on Nasdaq under the trading symbol “PRENW” and are measured at fair value based on quoted market price, using Level 1 inputs.
On October 28, 2025, the Group issued private placement warrants, including 2,722,642 Class A warrants, 2,722,642 Class B warrants, and 131,829 placement agent warrants, in connection with financing arrangements. These warrants are not publicly traded and are subject to certain transfer restrictions. The private placement warrants have similar economic characteristics to the public warrants, but differ in exercise price, transferability and certain contractual terms.
On December 23, 2025, the Group entered into warrant exchange agreements with certain warrant holders, pursuant to which each pair of one Class A warrant and one Class B warrant were exchanged for one Class C warrant for Class A ordinary shares of the Company. The exchange resulted in a reduction in the number of outstanding Class A and Class B warrants and the issuance of new Class C warrants with modified terms.
Upon completion of the exchange, the Group derecognized the carrying amount of the warrant liabilities associated with the exchanged warrants and recognized new warrant liabilities at fair value at the exchange date. The exchange was accounted for as an extinguishment of the original warrant liabilities and the recognition of new financial liabilities with modified terms. The difference between the fair value of the new warrant liabilities and the carrying amount of the warrant liabilities derecognized was recognized in profit or loss. The financial impact of the warrant exchange is reflected as a gain on warrant exchange in the consolidated statement of profit or loss for the year ended December 31, 2025.
Private placement warrants, including placement agent warrants, are not traded in an active market and are therefore measured using valuation techniques. The Group applies a binomial option pricing model to estimate their fair value, incorporating key assumptions such as share price, expected volatility, risk-free rate, expected term and exercise price. Class C warrants issued as part of the warrant exchange are measured using a Monte Carlo simulation model due to their more complex contractual features. These private placement and placement agent warrants are classified within Level 3 of the fair value hierarchy.
The following table summarizes the key assumptions used in the valuation of warrant liabilities classified within Level 3 of the fair value hierarchy:
| 2025 | |||||
| Fair value of private placement warrants and key assumptions | |||||
| Valuation technique | Binomial / Monte Carlo | ||||
| Exercise price | $ | ||||
| Expected volatility | |||||
| Risk-free interest rate | |||||
| Expected term (years) | |||||
| Dividend yield | % | ||||
All warrants are classified as financial liabilities through profit or loss and changes in the fair value of warrant liabilities are recognized in profit or loss in the period in which they arise.
Movement of the carrying amount of warrant liabilities measured using Level 1 inputs during the years ended December 31, 2025 and 2024 are as follow:
| 2025 | 2024 | ||||||||||
| Balance at January 1 | $ | $ | |||||||||
| Change in fair value recognized in profit or loss | ( | ||||||||||
| Balance at December 31 | $ | $ | |||||||||
F-59
27 Warrant liabilities (continued)
Movement of the carrying amount of warrant liabilities measured using Level 3 inputs during the years ended December 31, 2025 is as follow:
| Class A warrants | Class B warrants | Class C warrants | Placement agent warrants | Total | |||||||||||||||||||||||||
| Balance at January 1 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
| Issuance of warrants | |||||||||||||||||||||||||||||
| Warrants exchange | ( | ( | ( | ||||||||||||||||||||||||||
| Change in fair value recognized in profit or loss | ( | ||||||||||||||||||||||||||||
| Balance at December 31 | $ | $ | $ | $ | $ | ||||||||||||||||||||||||
28 Liabilities for puttable financial instrument
On December 30, 2022, the Group acquired 74.39 % of the issued share capital of ACT Genomics. In connection with the ACT Acquisition, a puttable financial instrument had been granted under the shareholders’ agreements to the remaining shareholders of ACT (the “NCI of ACT”), which the Group has an obligation to buy the remaining shares from the NCI of ACT at specified price if the NCI of ACT exercises the option before the contract’s expiry date.
The puttable financial instrument was presented as a current financial liability in the consolidated financial statements due to a potential event could trigger within twelve months from the end of the reporting period as of December 31, 2024. Following the disposal of a subsidiary on October 1, 2025, the shareholders’ agreements were amended and the relevant provisions governing the put option were terminated. Accordingly, upon completion of the disposal, the Group no longer has any contractual obligation to purchase the remaining shares from the NCI of ACT, and the puttable financial instrument ceased to exist.
The movement of the liabilities for puttable financial instrument during years ended December 31, 2025 and 2024 are analyzed as follows:
| 2025 | 2024 | ||||||||||
| Balance at January 1 | $ | $ | |||||||||
| Change in fair value recognized in equity | ( | ||||||||||
| Eliminated on disposal of a subsidiary | ( | ||||||||||
| Balance at December 31 | $ | $ | |||||||||
F-60
29 Capital and reserves
(a)Movement in ordinary shares of the Company
Authorized and issued share capital
| 2025 | 2024 | ||||||||||||||||||||||||||||
| Note | No. of shares | No. of shares | |||||||||||||||||||||||||||
Authorized Class A ordinary shares of $ | (i) | $ | $ | ||||||||||||||||||||||||||
Authorized Class B ordinary shares of $ | (i) | ||||||||||||||||||||||||||||
| $ | $ | ||||||||||||||||||||||||||||
| Class A ordinary shares, issued and fully paid: | |||||||||||||||||||||||||||||
| Balance at January 1 | $ | $ | |||||||||||||||||||||||||||
| Shares issued for advisors | (iv) | ||||||||||||||||||||||||||||
| Shares issued for vesting of RSUs | |||||||||||||||||||||||||||||
| Shares issued for public placement | (v) | ||||||||||||||||||||||||||||
| Balance at December 31 | (ii) | $ | $ | ||||||||||||||||||||||||||
| Class B ordinary shares, issued and fully paid: | |||||||||||||||||||||||||||||
| Balance at January 1 and December 31 | (iii) | $ | $ | ||||||||||||||||||||||||||
| Total share capital | $ | $ | |||||||||||||||||||||||||||
Notes:
(i)The authorized share capital of the Company is $320 and $50 as at December 31, 2025 and 2024, respectively divided into 213,333,334 and 33,333,334 shares, respectively, with a par value of $0.0015 198,000,001 shares (2024: 30,000,001 shares) are designated as Class A Ordinary Shares, and (ii) 15,333,333 shares (2024: 3,333,333 shares) are designated as convertible Class B Ordinary Shares. The share capital would reflect the par value with the excess recorded as share premium.
(ii)Class A ordinary shareholders are entitled to receive dividends as declared from time to time and are entitled to one vote per share at meetings of the Company. All ordinary shares rank equally with regard to the Group’s residual assets.
(iii)Class B ordinary shareholders are entitled to receive dividends as declared from time to time and are entitled to twenty vote per share at meetings of the Company. All ordinary shares rank equally with regard to the Group’s residual assets.
(iv)The Company has issued shares to advisors during December 31, 2025 and 2024 (see note 30(d)).
(v)On October 28, 2025, the Company issued ordinary shares pursuant to a public placement.
F-61
29 Capital and reserves (continued)
(b)Nature and purpose of reserves
(i)Capital reserve
The capital reserve represents restricted shares granted to shareholders but are subjected to certain restrictions and portion of the grant date fair value of unexercised share options granted to employees of the Company that has been recognized.
(ii)Translation reserve
The translation reserve comprises all foreign exchange differences arising from the translation of the financial statements of foreign operations.
(iii)Other reserves
The other reserves comprise (i) the fair value of shares to be issued of $5,061 in connection with the ACT Acquisition in 2022, which were issued in January 2023; (ii) the amortized cost of puttable financial instrument in connection with the ACT Acquisition; (iii) the then shareholders of Oxsed Limited exchanged GBP5,865,450 (equivalent to $7,549 ) into 110,150 ordinary shares in connection with the acquisition of Oxsed Limited; (iv) the remaining balance of the unconverted portion of the exchange loan notes recognized as equity instrument in accordance with the accounting policy adopted for convertible securities; and (v) the change in fair value of liabilities for puttable financial instrument, which had been granted under the shareholders’ agreements to the NCI of ACT.
(iv)Share premium
Under the Companies Law of the Cayman Islands, the funds in the share premium account of the Company are distributable to the shareholders of the Company provided that immediately following the date on which the dividend is proposed to be distributed, the Company will be in a position to pay off its debts as they fall due in the ordinary course of business.
(v)Treasury stock
At December 31, 2025 and 2024, the Company holds 143,072 639 .
(c)Capital management
The Group’s primary objectives when managing capital are to safeguard the Group’s ability to continue as a going concern, so that it can continue to provide returns for shareholders and benefits for other stakeholders, and to support the Group’s stability and growth, by pricing products and services commensurately with the level of risk.
The Group actively and regularly reviews and manages its capital structure to ensure optimal capital structure and shareholders return, taking into consideration the future of the Company and capital efficiency, prevailing and projected profitability, projected operating cash flows, projected capital expenditures and projected strategic investment opportunities.
The Group manages its capital structure and makes adjustments to it, in light of changes in economic conditions. To maintain or adjust the capital structure, the Group may adjust the dividend payment to shareholders, return capital to shareholders or issue new shares.
The Group's overall capital management objectives remained unchanged during the years ended December 31, 2023, 2024 and 2025. During the year ended December 31, 2025, the Group expanded its treasury management approach to include digital assets (primarily Bitcoin) as part of its capital resources. This resulted in enhanced monitoring procedures over market risk, liquidity and capital preservation in respect of such holdings. Digital assets are considered alongside cash and other financial resources in assessing the Group’s capital position and financial flexibility.
Neither the Company nor any of its subsidiaries are subject to externally imposed capital requirements.
30 Equity-settled share-based transactions
The Group’s equity incentive arrangements comprise the PHCL 2021 Share Incentive Plan (the “PHCL 2021 Plan”) and the Prenetics 2022 Share Incentive Plan (the “Prenetics 2022 Plan”).
F-62
30 Equity-settled share-based transactions (continued)
No further awards are granted under the PHCL 2021 Plan. All RSUs with respect to PHCL ordinary shares that were outstanding under the PHCL 2021 Plan have been replaced by awards under the Prenetics 2022 Plan. The replacement of awards did not result in any incremental fair value over and above the original grant-date fair value of the awards cancelled under the PHCL 2021 Plan.
(a)Prenetics 2022 Plan
The number of RSUs and aggregate fair value of RSUs granted to certain employees, directors and third parties under Prenetics 2022 Plan were as follows,
| Grant date | Number of RSUs | Closing price per ordinary share less subscription price per ordinary share | Aggregate fair value of the RSUs | ||||||||
| On May 18, 2022 | $ | $ | |||||||||
| On June 30, 2022 | |||||||||||
| On December 31, 2022 | |||||||||||
| On February 1, 2023 | |||||||||||
| On June 23, 2023 | |||||||||||
| On June 30, 2023 | |||||||||||
| On December 31, 2023 | |||||||||||
| On June 3, 2024 | |||||||||||
| On June 28, 2024 | |||||||||||
| On June 30, 2024 | |||||||||||
| On July 2, 2024 | |||||||||||
| On July 15, 2024 | |||||||||||
| On August 1, 2024 | |||||||||||
| On August 22, 2024 | |||||||||||
| On September 9, 2024 | |||||||||||
| On October 7, 2024 | |||||||||||
| On December 1, 2024 | |||||||||||
| On December 31, 2024 | |||||||||||
| On January 30, 2025 | |||||||||||
| On March 1, 2025 | |||||||||||
| On April 8, 2025 | |||||||||||
| On May 23, 2025 | |||||||||||
| On June 1, 2025 | |||||||||||
| On June 16, 2025 | |||||||||||
| On June 30, 2025 | |||||||||||
| On July 1, 2025 | |||||||||||
| On July 15, 2025 | |||||||||||
| On August 1, 2025 | |||||||||||
| On September 1, 2025 | |||||||||||
| On September 15, 2025 | |||||||||||
| On October 1, 2025 | |||||||||||
| On October 15, 2025 | |||||||||||
| On October 17, 2025 | |||||||||||
| On November 1, 2025 | |||||||||||
| On November 15, 2025 | |||||||||||
| On December 1, 2025 | |||||||||||
| On December 31, 2025 | |||||||||||
The RSUs granted were measured at the closing price per ordinary share less subscription price per ordinary share on grant date. The Company recognized employee share-based compensation benefits over the vesting period.
The RSUs outstanding at December 31, 2025 had an exercise price ranged from $0.001 to $2.96 per ordinary share (2024: from $0.001 to $3.00 per ordinary share), and a range of vesting period up to 3 years (2024: up to 3 years).
F-63
30 Equity-settled share-based transactions (continued)
The number and weighted average exercise prices of the RSUs are as follows:
| 2025 | 2024 | ||||||||||||||||||||||
| Weighted average exercise price | Number of RSUs | Weighted average exercise price | Number of RSUs | ||||||||||||||||||||
| Balance at January 1 | $ | $ | |||||||||||||||||||||
| Granted | |||||||||||||||||||||||
| Cancelled | ( | ( | |||||||||||||||||||||
| Exercised | ( | ( | |||||||||||||||||||||
| Outstanding balance at December 31 | $ | $ | |||||||||||||||||||||
| Exercisable balance at December 31 | $ | $ | |||||||||||||||||||||
During the year ended December 31, 2025, equity-settled share-based payment expenses in respect of the Prenetics 2022 Plan of $6,381 (2024: $5,416 ) was recognized in profit or loss, respectively. The remaining balance is recognized in profit or loss over the remaining vesting period.
(b)PHCL 2021 Plan
Details of the RSUs outstanding at December 31, 2025 and 2024 are as follows:
| Number of instruments | |||||||||||
| 2025 | 2024 | ||||||||||
| RSUs granted to directors | |||||||||||
| RSUs granted to employees | |||||||||||
| RSUs granted to third parties | |||||||||||
Under the PHCL 2021 Plan, PHCL granted 3,933,063 RSUs to certain employees, directors and third parties on June 16, 2021 and 63,934 RSUs in December 2021 to certain directors, employees and third parties, respectively.
The fair value of services received in return for the RSUs granted was measured by reference to the fair value of share options granted in 2021 and 2022. The estimate of the fair value of the share options granted was measured based on Black-Scholes Model. The contractual life of RSUs was used as an input into this model.
| 2021 | |||||
| Fair value of RSUs and key assumptions | |||||
| Fair value at measurement date | $ | ||||
| Share price | $ | ||||
| Exercise price | $ | ||||
| Expected volatility | |||||
| Expected option life | |||||
| Expected dividends | |||||
| Risk-free interest rate | |||||
| Likelihood of achieving a redemption event | % | ||||
| Likelihood of achieving a liquidity event | % | ||||
F-64
30 Equity-settled share-based transactions (continued)
The number and weighted average exercise prices of the RSUs are as follows:
| 2025 | 2024 | ||||||||||||||||||||||
| Weighted average exercise price | Number of RSUs | Weighted average exercise price | Number of RSUs | ||||||||||||||||||||
| Balance at January 1 | $ | $ | |||||||||||||||||||||
| Exercised | ( | n/a | |||||||||||||||||||||
| Outstanding balance at December 31 | $ | $ | |||||||||||||||||||||
| Exercisable balance at December 31 | $ | $ | |||||||||||||||||||||
The RSUs outstanding at December 31, 2025 had a weighted average exercise price of $0.01 per ordinary share (2024: $0.01 per ordinary share). At December 31, 2025, these RSUs had remaining contractual life as all outstanding awards were fully vested (2024: 0.4 years).
The aggregate fair value of the RSUs granted to the selected employees on the dates of grants on June 30, 2021 and December 31, 2021 was $54,646 ($13.89 per share) and $1,209 ($18.91 per share) respectively. The Company recognized employee share-based compensation benefits according to the restriction conditions.
During the year ended December 31, 2024, equity-settled share-based payment expenses in respect of the PHCL 2021 Plan of $557 was recognized in profit or loss. At December 31, 2025, no balance of equity-settled share-based payment expenses was recognized, as all awards granted under the PHCL 2021 Plan were fully vested.
(c)Share options issued by ACT Genomics
The range of vesting period is from 2 to 3 years from the date of share options granted.
At December 31, 2025, no share options were outstanding following the disposal of ACT Genomics on October 1, 2025. At December 31, 2024, the share options outstanding had a weighted average exercise price of $0.01 per ordinary share and a weighted average remaining contractual life of 9.0 years and weighted average fair value at grant date of $0.33 dollar.
The fair value of share options granted by ACT Genomics was estimated on the date of grant using binomial lattice model. The assumptions that the Group used to determine the grant-date fair value of share options granted during the year end December 31, 2024 was as follows:
| 2024 | |||||
| Fair value at measurement date ($ cents) | |||||
| Share price ($ cents) | |||||
| Exercise price ($) | |||||
| Expected option life | |||||
| Risk-free interest rate | % | ||||
| Expected volatility | |||||
| Expected dividend yield | % | ||||
| Expected probability of a liquidation event | % | ||||
During the year ended December 31, 2025, equity-settled share-based payment expenses in respect of the share options granted ACT Genomics of $1,226 (2024: $1,873 ) was recognized in profit or loss up to the date of disposal of ACT Genomics on October 1, 2025. Following the disposal, no further share-based payment expense will be recognized, and no remaining balance to be recognized in future periods.
(d)Ordinary shares of the Company issued to parties other than employees for service rendered
During the year ended December 31, 2024 and 2025, equity-settled share-based payment expenses of $4,132 and $— were recognized in profit or loss in relation to 721,106 ordinary shares and 6,148 ordinary shares of the Company issued to parties other than employees for service rendered, respectively. Such shares granted are measured at the market closing price of the shares of the Company granted at the date the counterparty renders the service.
F-65
31 Financial instruments - Fair values and risk management
A.Accounting classification and fair values
The following table shows the carrying amounts and fair values of financial assets and financial liabilities, including their levels in the fair value hierarchy. It does not include fair value information for financial assets and financial liabilities not measured at fair value if the carrying amount is a reasonable approximation of fair value.
| Carrying amount | Fair value | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes | Mandatorily at FVTPL - others | Financial assets at amortized cost | Other financial liabilities | Total | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2025 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial asset measured at fair value | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial assets at FVTPL | 20 | $ | $ | — | $ | — | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
| Financial assets at amortized cost | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trade receivables | 19 | $ | — | $ | $ | — | $ | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||
| Deposits and other receivables | 17, 19 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| Amount due from a related company | — | — | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | 21 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| $ | — | $ | $ | — | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
| Financial liabilities measured at fair value | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Warrant liabilities | 27 | $ | $ | — | $ | — | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Financial liabilities at amortized cost | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trade payables | $ | — | $ | — | $ | $ | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||||||||||||||||||||
| Other liabilities | 22 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| $ | — | $ | — | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
F-66
31 Financial instruments - Fair values and risk management (continued)
| Carrying amount | Fair value | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Notes | Mandatorily at FVTPL - others | Financial assets at amortized cost | Other financial liabilities | Total | Level 1 | Level 2 | Level 3 | Total | |||||||||||||||||||||||||||||||||||||||||||||
| Balance at December 31, 2024 | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial asset measured at fair value | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Financial assets at FVTPL | 20 | $ | $ | — | $ | — | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||||||||||||||
| Financial assets at amortized cost | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trade receivables | 19 | $ | — | $ | $ | — | $ | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||
| Deposits and other receivables | 17, 19 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| Amount due from a related company | — | — | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | 21 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| $ | — | $ | $ | — | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
| Financial liabilities measured at fair value | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Warrant liabilities | 27 | $ | $ | — | $ | — | $ | ||||||||||||||||||||||||||||||||||||||||||||||
| Financial liabilities at amortized cost | |||||||||||||||||||||||||||||||||||||||||||||||||||||
| Trade payables | $ | — | $ | — | $ | $ | n/a | n/a | n/a | n/a | |||||||||||||||||||||||||||||||||||||||||||
| Other liabilities | 22 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| Liabilities for puttable financial instrument | 28 | — | — | n/a | n/a | n/a | n/a | ||||||||||||||||||||||||||||||||||||||||||||||
| $ | — | $ | — | $ | $ | ||||||||||||||||||||||||||||||||||||||||||||||||
The Group’s finance team is responsible for overseeing the valuation of the financial instruments of the unlisted securities which are categorized into Level 3 of the fair value hierarchy. The team reports directly to the chief financial officer. Valuation results with analysis of changes in fair value measurement are prepared by the team with the assistance from external valuers where necessary and reviewed by the chief financial officer at each quarter end and annual reporting date. The valuation process is documented and updated where appropriate by the team and reviewed by the chief financial officer quarterly that coincides with the reporting dates.
F-67
31 Financial instruments - Fair values and risk management (continued)
B.Measurement of fair values
(i)Valuation techniques and significant unobservable inputs
The following tables show the valuation techniques used in measuring Level 1 and Level 3 fair values for financial instruments in the statement of financial position, as well as the significant unobservable inputs used.
Financial instruments measured at fair value
| Type | Fair value hierarchy | Valuation technique | Significant unobservable inputs | Inter-relationship between significant unobservable inputs and fair value measurement (Note) | ||||||||||||||||||||||
| Financial assets at FVTPL | Level 2 | The fair value is based on the published net asset value per share which is determined using observable market data for the underlying assets | n/a | n/a | ||||||||||||||||||||||
| Level 3 | Adjusted net asset value | Underlying assets’ value | The estimated fair value would increase if the underlying assets’ value is higher. | |||||||||||||||||||||||
| Level 3 | Asset-based approach with equity allocation using option pricing method (2024: Discounted cash flow method) | Volatility: | The estimated fair value would increase if: - the underlying assets’ value is higher; - the volatility is higher; - the time to liquidity event is longer; or - the risk-free rate is higher. (2024: The estimated fair value would increase if: - the risk-adjusted discount rate was lower; or - the discount for lack of marketability was lower.) | |||||||||||||||||||||||
Note: There are often interrelationships between significant unobservable inputs. For instance, the higher underlying assets’ value tends to increase with higher volatility, a longer time to liquidity event, or a higher risk-free rate. No sensitivity analysis is performed for the Level 3 financial assets as the directors of the Company consider that the exposure is insignificant.
(ii)Transfers between Levels 1 and 2
There were no transfers from Level 2 to Level 1 for the years ended December 31, 2025 and 2024.
F-68
31 Financial instruments - Fair values and risk management (continued)
(iii)Level 3 recurring fair values
Reconciliation of Level 3 fair values
The following table shows a reconciliation of financial assets at FVTPL and warrant liabilities from the opening balances to the closing balances for Level 3 fair values.
| Financial assets at FVTPL | Warrant liabilities | ||||||||||
| Balance at January 1, 2024 | $ | $ | |||||||||
| Additions | |||||||||||
| Changes in fair value recognized in profit or loss | ( | ||||||||||
| Balance at December 31, 2024 and January 1, 2025 | |||||||||||
| Additions | |||||||||||
| Redemptions | ( | ||||||||||
| Warrants exchange | ( | ||||||||||
| Changes in fair value recognized in profit or loss | |||||||||||
| Balance at December 31, 2025 | $ | $ | |||||||||
C.Financial risk management
The Group has exposure to the following risks arising from financial instruments:
-credit risk (see (C)(ii));
-liquidity risk (see (C)(iii)); and
-currency risk (see (C)(iv)).
(i)Risk management framework
The Company's board of directors has overall responsibility for the establishment and oversight of the Group's risk management framework. The management of the Group establishes policies and procedures around risk identification, measurement and management; and setting and monitoring risk limits and controls, in accordance with the objectives and underlying principles in the risk management framework approved by the board of directors. Risk management policies and procedures are reviewed regularly to reflect changes in market conditions and the Group's activities.
(ii)Credit risk
Credit risk is the risk of financial loss to the Group if a customer or counterparty to a financial instrument fails to meet its contractual obligations and arises principally from the Group's receivables from customers.
The carrying amounts of financial assets represent the maximum credit exposure.
F-69
31 Financial instruments - Fair values and risk management (continued)
Trade receivables
The Group’s exposure to credit risk is influenced mainly by the individual characteristics of each customer. At December 31, 2025 and 2024, 37 % and 5 % of the total trade receivables were due from the Group’s largest counterparty, respectively, and 81 % and 16 % of the total trade receivables were due from the Group’s five largest counterparties, respectively.
Expected credit loss assessment
During the year ended December 31, 2025, the Group determines the expected credit losses on trade receivables by using a provision matrix, estimated based on historical credit loss experience based on the past due status of the debtors, adjusted as appropriate to reflect current conditions and estimates of future economic conditions at the reporting date.
Internal credit scoring is performed on all customers for the year ended December 31, 2025. These take into account the customer’s past payment history, financial position and other factors. Trade receivables are due within 30 to 90 days from the billing date. The Group does not obtain collateral in respect of trade and other receivables. The Group does not have trade receivable for which no loss allowance is recognized because of collateral.
The Group’s current credit risk grading framework comprises the following categories:
| Category | Description | Basis for recognizing expected credit losses | ||||||
| Performing | The counterparty has a low risk of default and does not have any past-due amounts. | Lifetime ECL – not credit-impaired | ||||||
| Doubtful | Amount is >30 days past due or there has been a significant increase in credit risk since initial recognition. | Lifetime ECL – not credit-impaired | ||||||
| In default | Amount is >90 days past due or there is evidence indicating the asset is credit-impaired. | Lifetime ECL – credit-impaired | ||||||
| Write-off | There is evidence indicating that the debtor is in severe financial difficulty and the Group has no realistic prospect of recovery. | Amount is written off | ||||||
The Group has applied the simplified approach in IFRS 9 Financial Instruments to measure the loss allowance at lifetime ECL. The Group determines the expected credit losses on trade receivables by using an internal credit rating for its customers. The following table provides information about the exposure to credit risk for trade receivables which are assessed on a collective basis within lifetime ECL (not credit-impaired) and the estimated loss rates are estimated based on historical observed default rates over the expected life of the debtors.
Gross carrying amount
| 2025 | |||||||||||
| Internal credit rating | Average loss rate | Trade receivables | |||||||||
| Performing | % | $ | |||||||||
| Doubtful | % | ||||||||||
| In default | % | ||||||||||
| Write-off | % | ||||||||||
| $ | |||||||||||
F-70
31 Financial instruments - Fair values and risk management (continued)
Movement in the allowance for impairment in respect of trade receivables
Movement in the loss allowance account in respect of trade receivable during the years ended December 31, 2025 and 2024 is as follows:
| 2025 | 2024 | ||||||||||
| Balance at January 1 | $ | $ | |||||||||
| Additions from acquisition (note 33(III)) | |||||||||||
| Net remeasurement of loss allowance | |||||||||||
| Amounts written off | ( | ( | |||||||||
| Eliminated on disposal of a subsidiary (note 34) | ( | ||||||||||
| Exchange differences | |||||||||||
| Balance at December 31 | $ | $ | |||||||||
Cash and cash equivalents
The Group held cash and cash equivalents of $32,131 at December 31, 2025 (2024: $52,251 ). The cash and cash equivalents are held with bank and financial institution counterparties with high credit-ratings assigned by international credit-rating agencies.
Impairment on cash and cash equivalents has been measured on a 12-month expected loss basis and reflects the short maturities of the exposures. The Group considers that its cash and cash equivalents have low credit risk based on the external credit ratings of the counterparties.
(iii)Liquidity risk
Liquidity risk is the risk that the Group will encounter difficulty in meeting the obligations associated with its financial liabilities that are settled by delivering cash or another financial asset. The Group's objective when managing liquidity is to ensure, as far as possible, that it will have sufficient liquidity to meet its liabilities when they are due, under both normal and stressed conditions, without incurring unacceptable losses or risking damage to the Group's reputation.
F-71
31 Financial instruments - Fair values and risk management (continued)
The following are the remaining contractual maturities of financial liabilities at the reporting date. The amounts are gross and undiscounted, and include contractual interest payments.
| Contractual undiscounted cash flows | |||||||||||||||||||||||||||||||||||
| Weighted average effective interest rate | Carrying amount | Total | Within 1 year or on demand | 1 - 2 years | More than 2 years | ||||||||||||||||||||||||||||||
| Balance at December 31, 2025 | |||||||||||||||||||||||||||||||||||
| Trade payables | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||
| Other liabilities | |||||||||||||||||||||||||||||||||||
| Lease liabilities | % | ||||||||||||||||||||||||||||||||||
| $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||
| Balance at December 31, 2024 | |||||||||||||||||||||||||||||||||||
| Trade payables | $ | $ | $ | $ | $ | ||||||||||||||||||||||||||||||
| Other liabilities | |||||||||||||||||||||||||||||||||||
| Lease liabilities | % | ||||||||||||||||||||||||||||||||||
| Liabilities for puttable financial instrument | % | ||||||||||||||||||||||||||||||||||
| $ | $ | $ | $ | $ | |||||||||||||||||||||||||||||||
(iv)Market risk
Market risk is the risk that changes in market prices – e.g. foreign exchange rates – will affect the Group’s income or the value of its holdings of financial instruments. The objective of market risk management is to manage and control market risk exposures within acceptable parameters, while optimizing the return.
Currency risk
The Group is exposed to transactional foreign currency risk to the extent that there is a mismatch between the currencies in which sales, purchases and receivables are denominated and the respective functional currencies of Group companies. The functional currencies of Group companies are primarily USD and Hong Kong dollar ("HKD"). The currencies in which these transactions are primarily denominated are USD and HKD.
As the HKD is pegged to the USD, the Group considers the risk of movements in exchange rates between the HKD and the USD to be insignificant.
F-72
32 List of subsidiaries
The following list contains the material subsidiaries of the Group at December 31, 2025 and 2024 are as follows:
| Name of subsidiaries | Place of incorporation/ operation | Issued and fully paid share capital | Proportion of nominal value of issue capital held by the Company | Principal activities | ||||||||||||||||||||||||||||||||||||||||
| 2025 | 2024 | |||||||||||||||||||||||||||||||||||||||||||
| Directly | Indirectly | Directly | Indirectly | |||||||||||||||||||||||||||||||||||||||||
| % | % | % | % | |||||||||||||||||||||||||||||||||||||||||
| Prenetics Limited | Hong Kong | HK$ | Genetic and diagnostic health testing | |||||||||||||||||||||||||||||||||||||||||
| Europa Partners Holdings, LLC (note 33) | The United States | GBP | Provision of fulfillment and distribution services of sports nutrition products | |||||||||||||||||||||||||||||||||||||||||
| IM8 Group Holding Company | Cayman Islands | $ | Sales of consumer health products | |||||||||||||||||||||||||||||||||||||||||
| IM8 (US) LLC | The United States | n/a | Sales of consumer health products | |||||||||||||||||||||||||||||||||||||||||
| IM8 Limited | Hong Kong | HK$ | Sales of consumer health products | |||||||||||||||||||||||||||||||||||||||||
| ACT Genomics Holdings Company Limited | Cayman Islands | $ | Precise cancer genetic testing services | |||||||||||||||||||||||||||||||||||||||||
| ACT Genomics Co., Ltd. | Taiwan | TWD | Precise cancer genetic testing - services | |||||||||||||||||||||||||||||||||||||||||
| ACT Genomics (Hong Kong) Limited | Hong Kong | HK$ | Precise cancer genetic testing - services | |||||||||||||||||||||||||||||||||||||||||
| Sanomics Limited | Hong Kong | HK$ | Precise cancer genetic testing - services | |||||||||||||||||||||||||||||||||||||||||
| MC Diagnostics Limited | United Kingdom | GBP | Sales of medical diagnostics products | |||||||||||||||||||||||||||||||||||||||||
33 Acquisition of a subsidiary
On August 9, 2024, the Group acquired 100 % interest in Europa with cash consideration of $8,509 . Europa is principally engaged in the sales and distribution of consumer health and wellness products in the United States and was acquired with the objective of expanding the Group into the United States consumer health and wellness industry. The acquisition has been accounted for as acquisition of business using the acquisition method.
I.Consideration transferred
The following table summarizes the acquisition date fair value of each major class of consideration transferred.
| Total cash consideration transferred | $ | ||||
| Net cash outflow arising on acquisition: | |||||
| Cash consideration | $ | ||||
| Less: cash and cash equivalent balances acquired (note 33(III)) | |||||
| Total net cash inflow arising on acquisition | $ | ||||
II.Acquisition-related costs
The Group incurred acquisition-related costs of $613 on legal fees and due diligence costs. These costs have been included in ‘administrative and other operating expenses’.
F-73
33 Acquisition of a subsidiary (continued)
III.Identifiable assets acquired and liabilities assumed
The following table summarizes the recognized amounts of assets acquired and liabilities based on fair value at the date of acquisition.
| Property, plant and equipment and intangible assets | $ | ||||
| Other non-current assets | |||||
| Inventories | |||||
Trade receivables (including loss allowance $ | |||||
| Deposits, prepayments and other receivables | |||||
| Cash and cash equivalents | |||||
| Trade payables | ( | ||||
| Accrued expenses | ( | ||||
| Lease liabilities | ( | ||||
| Total identifiable net assets acquired at fair value | $ | ||||
The receivables acquired (which principally comprised trade receivables) with a fair value of $619 at the date of acquisition had gross contractual amounts of $699 . The best estimate at acquisition date of the contractual cash flows not expected to be collected amounted to $80 .
Measurement of fair values
The valuation technique used for measuring the fair value of material assets acquired was as follow.
| Assets acquired | Valuation technique | ||||
| Property, plant and equipment | Cost technique: The valuation model considers market prices for depreciated replacement cost when appropriate. Depreciated replacement cost reflects functional and economic obsolescence. | ||||
If the acquisition had occurred on January 1, 2024, management estimates that the Group’s consolidated revenue would have been increased by $13,094 , and consolidated loss for the year would have been increased by $3,624 . The pro forma information is for illustrative purposes only and is not necessarily an indication of revenue and results of operations of the Group that actually would have been achieved had the acquisition been completed on January 1, 2024, nor is it intended to be a projection of future results.
IV.Goodwill
Goodwill arising from the acquisition has been recognized as follows.
| 2024 | |||||
| Consideration transferred (note 33(I)) | $ | ||||
| Fair value of identifiable net assets (note 33(III)) | ( | ||||
| Goodwill | $ | ||||
The goodwill is attributable mainly to the skills and technical talent of Europa’s work force and the synergies expected to be achieved from integrating the company into the Group’s existing business. None of the goodwill recognized is expected to be deductible for tax purposes.
F-74
34 Disposal of a subsidiary
As referred to in note 10, on October 1, 2025, the Group discontinued its precision oncology services at the time of disposal of its subsidiary, ACT Genomics. The net assets of ACT Genomics at the date of disposal were as follows:
Analysis of assets and liabilities over which control was lost:
| Property, plant and equipment | $ | ||||
| Intangible assets | |||||
| Goodwill | |||||
| Other non-current assets | |||||
| Inventories | |||||
| Trade receivables | |||||
| Deposits, prepayments and other receivables | |||||
| Cash and cash equivalents | |||||
| Deferred tax liabilities | ( | ||||
| Lease liabilities - non-current | ( | ||||
| Other non-current liabilities | ( | ||||
| Trade payables | ( | ||||
| Accrued expenses and other current liabilities | ( | ||||
| Contract liabilities | ( | ||||
| Lease liabilities - current | ( | ||||
| Net assets disposed of | $ | ||||
Consideration received:
| Cash received | $ | ||||
| Deferred cash consideration (Note) | |||||
| Total consideration | $ | ||||
Gain on disposal of a subsidiary:
| Consideration received and receivable | $ | ||||
| Net assets disposed of | ( | ||||
| Non-controlling interests | ( | ||||
| Reclassification of cumulative translation reserve upon disposal of ACT Genomics to profit or loss | |||||
| Gain on disposal (note 10) | $ | ||||
Net cash inflow arising on disposal:
| Cash consideration | $ | ||||
| Less: cash and cash equivalents disposed of | ( | ||||
| $ | |||||
F-75
35 Non-controlling interests
From continuing and discontinued operations
The non-controlling interests were based on the following data:
| 2025 | 2024 | 2023 | |||||||||||||||
| (Loss)/profit attributable to non-controlling interests | |||||||||||||||||
| - from continuing operations | $ | ( | $ | $ | ( | ||||||||||||
| - from discontinued operations | ( | ( | ( | ||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||
| Total comprehensive (expenses)/income attributable to non-controlling interests | |||||||||||||||||
| - from continuing operations | $ | ( | $ | $ | ( | ||||||||||||
| - from discontinued operations | ( | ( | ( | ||||||||||||||
| $ | ( | $ | ( | $ | ( | ||||||||||||
The following table summaries the information relating each of the Group's subsidiaries that has material non-controlling interests, before any intra-group eliminations.
| As at October 1, 2025 | As at December 31, 2024 | ||||||||||
| ACT Genomics | |||||||||||
| Non-controlling interests percentage | % | % | |||||||||
| As at October 1, 2025 | As at December 31, 2024 | ||||||||||
| Non-current assets | $ | $ | |||||||||
| Current assets | |||||||||||
| Non-current liabilities | ( | ( | |||||||||
| Current liabilities | ( | ( | |||||||||
| Equity attributable to equity shareholder of the parent Company | |||||||||||
| Equity attributable to non-controlling interests | ( | ||||||||||
F-76
| January 1, 2025 to October 1, 2025 | January 1, 2024 to December 31, 2024 | ||||||||||
| Revenue | $ | $ | |||||||||
| Expenses | ( | ( | |||||||||
| Loss for the year | $ | ( | $ | ( | |||||||
| Loss attributable to equity shareholder of the parent Company | $ | ( | $ | ( | |||||||
| Loss attributable to non-controlling interests | ( | ( | |||||||||
| Loss for the year | $ | ( | $ | ( | |||||||
| Other comprehensive income/(expense) attributable to equity shareholder of the parent Company | $ | $ | ( | ||||||||
| Other comprehensive income/(expense) attributable to non-controlling interests | ( | ||||||||||
| Other comprehensive income/(expense) for the year | $ | $ | ( | ||||||||
| Total comprehensive expense attributable to equity shareholder of the parent Company | $ | ( | $ | ( | |||||||
| Total comprehensive expense attributable to non-controlling interests | ( | ( | |||||||||
| Total comprehensive expense for the year | $ | ( | $ | ( | |||||||
| Net cash (outflow)/inflow from operating activities | $ | ( | $ | ||||||||
| Net cash outflow from investing activities | ( | ( | |||||||||
| Net cash outflow from financing activities | ( | ( | |||||||||
| Net cash (outflow)/inflow | $ | ( | $ | ||||||||
| Dividends paid to non-controlling interests | $ | $ | |||||||||
F-77
36 Related parties
Apart from balances and transactions disclosed elsewhere in these consolidated financial statements, the Group has also entered into the following related party transactions under the normal course of the Group’s business:
(a)Transactions with other related parties
| 2025 | 2024 | 2023 | |||||||||||||||
| Sales to an equity-accounted investee (Note(a)) | $ | $ | $ | ||||||||||||||
| Services recharge received from an equity-accounted investee (Note(b)) | |||||||||||||||||
| Services recharge received from a related party (Note(c)) | |||||||||||||||||
Notes:
(a)Sales to an equity-accounted investee represents cancer treatment services provided. There is no outstanding balance at December 31, 2025, while there was $127 outstanding balance at December 31, 2024 in relation to these transactions.
(b)Service recharged received from an equity-accounted investee represents the office rental services. There is $4 outstanding balance at December 31, 2025 and no outstanding balance at December 31, 2024 in relation to these transactions.
(c)Service recharged received from a related party represents the office supporting services. The related party is considered related as one of the directors of the Company is a key management personnel of that entity. There is no outstanding balance at at December 31, 2025, while there is $3 outstanding balance at December 31, 2024 in relation to these transactions.
(d)Key management personnel compensation
Key management personnel compensation comprised as following.
| 2025 | 2024 | 2023 | |||||||||||||||
| Directors’ fees | $ | $ | $ | ||||||||||||||
| Salaries, wages and other benefits | |||||||||||||||||
| Contributions to defined contribution retirement plan | |||||||||||||||||
| Equity-settled share-based payment expenses (note) | |||||||||||||||||
| $ | $ | $ | |||||||||||||||
Note: The balances are non-cash transactions for the reporting period. Details of the recognition and the fair value determination are included in note 30.
37 Major non-cash transaction
During the year ended December 31, 2025, the Group redeemed financial assets at fair value through profit or loss with a carrying amount of $1,000 , which were reclassified to other receivables. As this transaction did not involve cash consideration, it has been excluded from the consolidated statement of cash flows.
During the year ended December 31, 2025, the Group recognized the issuance of private placement warrants with an aggregate fair value of approximately $38,858 in connection with its financing arrangements. As this transaction did not involve cash consideration, it has been excluded from the consolidated statement of cash flows.
F-78
38 Pending Litigation
The Group is subject to periodic legal or administrative proceedings in the ordinary course of business. Any situation will be reviewed in conjunction with the Group’s legal advisors. The Group does not believe that any currently pending legal proceeding to which the Group is a party will have a material effect on its business, results of operations or cash flows.
39 Subsequent Events
On January 1, 2026, the Group entered into an Asset Purchase Agreement with an independent buyer pursuant to which the Group divested substantially all of the assets of its logistics, fulfillment, warehousing and distribution business operated under Europa Sports Partners, LLC (the “3PL Business”). The aggregate consideration comprises share consideration in the buyer’s parent, consisting of (i) closing shares with an aggregate value of approximately $3,000 , subject to a vesting condition linked to specified performance milestones, and (ii) contingent share consideration of up to $10,000 , payable in tranches upon achievement of additional performance targets during the earn-out period. The performance milestones are primarily linked to the operational and commercial performance of the disposed business, including shipment volume–based targets and referral revenue generated under post-closing commercial arrangements. The buyer also assumed certain liabilities associated with the transferred business relating to the assigned contracts and operations subsequent to the closing date.
On February 17, 2026, the Group completed the sale of its 35 % equity interest in Insighta to a third party for total cash consideration of $70,000 , including $1,000 held in escrow pending customary release conditions.
The Group has evaluated events subsequent to December 31, 2025 through April 30, 2026, the date on which the financial statements are available to be issued. Other than the event disclosed above, no further subsequent events have been identified that would require adjustment to or disclosure in the consolidated financial statements.
40 Approval of Consolidated Financial Statements
The consolidated financial statements were approved and authorized for issue by the Board of Directors on April 30, 2026.
F-79

8054020/81436035/2 COMPANIES ACT (AS AMENDED) COMPANY LIMITED BY SHARES AMENDED AND RESTATED MEMORANDUM AND ARTICLES OF ASSOCIATION OF PRENETICS GLOBAL LIMITED (adopted by a special resolution passed on 15 September 2021 and effective at the Initial Merger Effective Time) Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

1 COMPANIES ACT (AS AMENDED) COMPANY LIMITED BY SHARES AMENDED AND RESTATED MEMORANDUM OF ASSOCIATION OF PRENETICS GLOBAL LIMITED (adopted by a special resolution passed on 15 September 2021 and effective at the Initial Merger Effective Time) 1. The name of the Company is Prenetics Global Limited. 2. The registered office of the Company shall be at the offices of Mourant Governance Services (Cayman) Limited, 94 Solaris Avenue, Camana Bay, PO Box 1348, Grand Cayman KY1-1108, Cayman Islands or at such other place as the Directors may from time to time decide. 3. The objects for which the Company is established are unrestricted and the Company shall have full power and authority to carry out any object not prohibited by law as provided by Section 7(4) of the Companies Act. 4. The Company shall have and be capable of exercising all the functions of a natural person of full capacity irrespective of any question of corporate benefit as provided by Section 27(2) of the Companies Act. 5. Nothing in the preceding paragraphs shall be deemed to permit the Company to carry on the business of a bank or trust company without being licensed in that behalf under the provisions of the Banks and Trust Companies Act (as amended) or to carry on insurance business from within the Cayman Islands or the business of an insurance manager, agent, sub-agent or broker without being licensed in that behalf under the provisions of the Insurance Act (as amended), Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

2 or to carry on the business of company management without being licensed in that behalf under the provisions of the Companies Management Act (as amended). 6. The Company will not trade in the Cayman Islands with any person, firm or corporation except in furtherance of the business of the Company carried on outside the Cayman Islands, provided that nothing in this Memorandum of Association shall be construed as to prevent the Company from effecting and concluding contracts in the Cayman Islands, and exercising in the Cayman Islands all of its powers necessary for the carrying on of business outside the Cayman Islands. 7. The liability of each member is limited to the amount from time to time unpaid on such member's shares. 8. The authorised share capital of the Company is US$50,000 divided into 500,000,000 Shares of US$0.0001 par value each, of which (i) 400,000,000 shall be designated as Class A Ordinary Shares, (ii) 50,000,000 shall be designated as convertible Class B Ordinary Shares and (iii) 50,000,000 shall be designated as shares of such class or classes (however designated) as the Board of Directors may determine in accordance with Article 10 of the Articles, with the power for the Company, insofar as is permitted by law and the Articles, to redeem, purchase or redesignate any of its shares and to increase or reduce the said share capital subject to the Companies Act and the Articles and to issue any part of its capital, whether original, redeemed or increased with or without any preference, priority or special privilege or subject to any postponement of rights or to any conditions or restrictions and so that unless the conditions of issue shall otherwise expressly declare every issue of shares whether declared to be preference or otherwise shall be subject to the powers hereinbefore contained. 9. The Company may exercise the power contained in Section 206 of the Companies Act to deregister in the Cayman Islands and be registered by way of continuation in another jurisdiction. 10. Capitalised terms that are not defined in this Memorandum bear the same meanings given to those terms in the Articles. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

i COMPANIES ACT (AS AMENDED) COMPANY LIMITED BY SHARES AMENDED AND RESTATED ARTICLES OF ASSOCIATION OF PRENETICS GLOBAL LIMITED (adopted by a special resolution passed on 15 September 2021 and effective at the Initial Merger Effective Time) Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

ii TABLE OF CONTENTS ARTICLE PAGE TABLE A ............................................................................................................................. 1 DEFINITIONS AND INTERPRETATION..................................................................................... 1 COMMENCEMENT OF BUSINESS ............................................................................................ 6 SITUATION OF REGISTERED OFFICE ..................................................................................... 7 SHARES ............................................................................................................................. 7 REDEMPTION, PURCHASE AND SURRENDER OF SHARES ......................................................... 12 TREASURY SHARES ............................................................................................................ 13 MODIFICATION OF RIGHTS ................................................................................................. 13 SHARE CERTIFICATES ........................................................................................................ 14 TRANSFER AND TRANSMISSION OF SHARES ......................................................................... 14 LIEN ................................................................................................................................. 16 CALL ON SHARES ............................................................................................................... 16 FORFEITURE OF SHARES .................................................................................................... 17 ALTERATION OF SHARE CAPITAL ......................................................................................... 18 GENERAL MEETINGS .......................................................................................................... 18 NOTICE OF GENERAL MEETINGS .......................................................................................... 19 PROCEEDINGS AT GENERAL MEETINGS ................................................................................ 19 VOTES OF SHAREHOLDERS ................................................................................................. 21 WRITTEN RESOLUTIONS OF SHAREHOLDERS ........................................................................ 23 DIRECTORS ....................................................................................................................... 23 TRANSACTIONS WITH DIRECTORS ....................................................................................... 25 POWERS AND DUTIES OF DIRECTORS .................................................................................. 26 PROCEEDINGS OF DIRECTORS ............................................................................................ 26 WRITTEN RESOLUTIONS OF DIRECTORS .............................................................................. 28 PRESUMPTION OF ASSENT .................................................................................................. 28 BORROWING POWERS ........................................................................................................ 29 SECRETARY ....................................................................................................................... 29 THE SEAL .......................................................................................................................... 29 DIVIDENDS, DISTRIBUTIONS AND RESERVES ....................................................................... 29 SHARE PREMIUM ACCOUNT ................................................................................................. 30 ACCOUNTS ........................................................................................................................ 31 AUDIT .............................................................................................................................. 31 NOTICES AND INFORMATION .............................................................................................. 32 WINDING UP AND FINAL DISTRIBUTION OF ASSETS .............................................................. 34 INDEMNITY ....................................................................................................................... 34 DISCLOSURE ..................................................................................................................... 35 CLOSING REGISTER OF MEMBERS OR FIXING RECORD DATE .................................................. 35 REGISTRATION BY WAY OF CONTINUATION .......................................................................... 35 FINANCIAL YEAR ................................................................................................................ 36 AMENDMENTS TO MEMORANDUM AND ARTICLES OF ASSOCIATION ......................................... 36 MERGERS AND CONSOLIDATION ......................................................................................... 36 Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

1 COMPANIES ACT (AS AMENDED) COMPANY LIMITED BY SHARES AMENDED AND RESTATED ARTICLES OF ASSOCIATION OF PRENETICS GLOBAL LIMITED (adopted by a special resolution passed on 15 September 2021 and effective at the Initial Merger Effective Time) TABLE A 1. In these Articles, the regulations contained in Table A in the First Schedule to the Companies Act (as defined below) do not apply except insofar as they are repeated or contained in these Articles. DEFINITIONS AND INTERPRETATION 2. In these Articles, the following words and expressions shall have the meanings set out below save where the context otherwise requires: Acquisition Effective Time has the meaning ascribed to such term in the Business Combination Agreement; Affiliate means, in respect of a person, any other person that, directly or indirectly, through one or more intermediaries, Controls, is Controlled by, or is under common Control with, such person; provided, that in the case of a Key Executive, the term Affiliate shall include such Key Executive’s Permitted Entities, notwithstanding anything to the contrary contained herein; Articles means these Articles of Association of the Company, as amended from time to time by Special Resolution; Auditors means the auditor or auditors for the time being of the Company; Board of Directors means the Directors assembled as a board; Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

2 Business Combination Agreement means that certain Business Combination Agreement among the Company, Artisan Acquisition Corp., AAC Merger Limited, PGL Merger Limited and Prenetics Group Limited dated 15 September 2021 (as the same may be amended, restated or supplemented); Business Day means any day, excluding Saturdays, Sundays, and any other day on which commercial banks are authorized or required by law to close in New York, U.S., the Cayman Islands, or Hong Kong; Chairperson means the chairperson of the Board of Directors; Class A Ordinary Share means a Class A Ordinary Share in the capital of the Company of a par value of US$0.0001 having the rights, benefits and privileges set out in these Articles; Class B Ordinary Share means a Class B Ordinary Share in the capital of the Company of a par value of US$0.0001 having the rights, benefits and privileges set out in these Articles; Class B Ordinary Shareholder means a holder of Class B Ordinary Shares; Communication Facilities means video, video-conferencing, internet or online conferencing applications, telephone or tele-conferencing and/or any other video-communications, internet or online conferencing application or telecommunications facilities by means of which all persons participating in a meeting are capable of hearing and being heard by each other; Companies Act means the Companies Act (as amended); Company means the above-named company; Control, Controlling, under common Control with means directly or indirectly: (i) the ownership or control of a majority of the outstanding voting securities of such person; (ii) the right to control the exercise of a majority of the votes at a meeting of the board of directors (or equivalent governing body) of such person; or (iii) the ability to direct or cause the direction of the management and policies of such person (whether by contract, through other legally enforceable rights or howsoever arising); Designated Stock Exchange means NASDAQ or any other internationally recognized stock exchange on which the Company’s securities are traded; Designated Stock Exchange Rules means the relevant code, rules and regulations, as amended, from time to time, applicable as a result of the original and continued listing of any Shares on the Designated Stock Exchange; Directors means the directors of the Company for the time being; Electronic Record has the same meaning as in the Electronic Transactions Act; Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

3 Electronic Transactions Act means the Electronic Transactions Act (as amended); Family Members means the following individuals: the applicable individual, the spouse of the applicable individual (including former spouses), the parents of the applicable individual, the lineal descendants of the applicable individual, the siblings of the applicable individual, and the lineal descendants of a sibling of the applicable individual. For purposes of the preceding sentence, the descendants of any individual shall include adopted individuals and their issue but only if the adopted individual was adopted prior to attaining age 18; Incapacity means with respect to an individual, the permanent and total disability of such individual so that such individual is unable to engage in any substantial gainful activity by reason of any medically determinable mental impairment which can be expected to result in death or which has lasted or can be expected to last for a continuous period of not less than twelve (12) months as determined by a licensed medical practitioner. In the event of a dispute regarding whether an individual has suffered an Incapacity, no Incapacity of such individual will be deemed to have occurred unless and until an affirmative ruling regarding such Incapacity has been made by a court or arbitral panel of competent jurisdiction, and such ruling has become final and non-appealable; Indemnified Person has the meaning set out in Article 173; Initial Merger Effective Time has the meaning ascribed to such term in the Business Combination Agreement; Key Executive means Danny Yeung and his Permitted Entities and Permitted Transferees of each of them; Memorandum means the Memorandum of Association of the Company, as amended and restated from time to time by Special Resolution; Notice Period has the meaning set out in Article 123; Ordinary Resolution means a resolution: (a) passed by a simple majority of the votes of such Shareholders as, being entitled to do so, vote in person or, where proxies are allowed, by proxy, at a general meeting of the Company and where a poll is taken regard shall be had in computing a majority to the number of votes to which each Shareholder is entitled; or (b) approved in writing by all the Shareholders entitled to vote at a general meeting of the Company in one or more instruments each signed by one or more of the Shareholders aforesaid, and the effective date of the resolution so adopted shall be the date on which the instrument or the last of such instruments, if more than one, is executed; Ordinary Shares means, collectively, the Class A Ordinary Shares and the Class B Ordinary Shares; Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

4 paid up means paid up as to the par value and any premium payable in respect of the issue of any Shares and includes credited as paid up; Permitted Entity with respect to any Key Executive: (a) any person in respect of which such Key Executive has, directly or indirectly: (i) control with respect to the voting of all the Class B Ordinary Shares held by or to be transferred to such person; (ii) the ability to direct or cause the direction of the management and policies of such person or any other person having the authority referred to in the preceding clause (a)(i) (whether by contract, as executor, trustee, trust protector or otherwise); or (iii) the operational or practical control of such person, including through the right to appoint, designate, remove or replace the person having the authority referred to in the preceding clauses (a)(i) or (ii); (b) any trust the beneficiaries of which consist primarily of a Key Executive, his or her Family Members, and/or any persons Controlled directly or indirectly Controlled by such a trust; and (c) any person Controlled by a trust described in the immediately preceding clause (b); Permitted Transferee with respect to the Class B Ordinary Shareholders, any or all of the following: (a) any Key Executive; (b) any Key Executive’s Permitted Entities; (c) the transferee or other recipient in any transfer of any Class B Ordinary Shares by any Class B Ordinary Shareholder: (i) to (A) his or her Family Members; (B) any other relative or individual approved by the Board of Directors; or (C) any trust or estate planning entity (including partnerships, limited companies, and limited liability companies), that is primarily for the benefit of, or the ownership interests of which are Controlled by, such Class B Ordinary Shareholder, his or her Family Members, and/or other trusts or estate planning entities described in this paragraph (c), or any entity Controlled by such Key Executive or a trust or estate planning entity; or (ii) occurring by operation of law, including in connection with divorce proceedings; (d) any charitable organization, foundation, or similar entity; (e) the Company or any of its subsidiaries; (f) in connection with a transfer as a result of, or in connection with, the death or Incapacity of a Key Executive: any Key Executive’s Family Members, another Class B Ordinary Shareholder, or a designee approved by majority of all Directors , provided that in case of any transfer of Class B Ordinary Shares pursuant to clauses (b) through (e) above to a person who at any later time ceases to be a Permitted Transferee under the relevant clause, the Company shall be entitled to refuse registration of any subsequent transfer of such Class B Ordinary Shares except back to the transferor of such Class B Ordinary Shares pursuant to clauses (b) through (e) (or to a Key Executive or his or her Permitted Transferees) and in the absence of such transfer back to the transferor (or to a Key Executive or his or her Permitted Transferees), the applicable Class B Ordinary Shares shall convert in accordance with Article 21(d)(iv) applied mutatis mutandis; person means, any natural person, firm, company, joint venture, partnership, corporation, association or other entity (whether Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

5 or not having separate legal personality) or any of them as the context so requires; present means in respect of any person, such person's presence at a general meeting of the Company (or any meeting of the holders of any class of Shares), which may be satisfied by means of such person or, if a corporation or other non-natural person, its duly authorized representative (or, in the case of any Shareholder, a proxy which has been validly appointed by such Shareholder in accordance with these Articles), being: (a) physically present at the meeting; or (b) in the case of any meeting at which Communication Facilities are permitted in accordance with these Articles, including any Virtual Meeting, connected by means of the use of such Communication Facilities; Register of Members means the register of Shareholders to be kept pursuant to these Articles and the Companies Act; Registered Office means the registered office of the Company for the time being; Seal means the common seal of the Company including any duplicate seal; Secretary means any person appointed by the Directors to perform any of the duties of the secretary of the Company, including a joint, assistant or deputy secretary; Securities Act means the Securities Act of 1933 of the United States of America, as amended, or any similar federal statute and the rules and regulations of the Securities and Exchange Commission of the United States of America thereunder, all as the same shall be in effect at the time; Share means any share in the capital of the Company of any class including a fraction of a share; Share Premium Account means the share premium account established in accordance with these Articles and the Companies Act; Shareholder means any person registered in the Register of Members as the holder of Shares of the Company; signed includes an electronic signature and a signature or representation of a signature affixed by mechanical means; Special Resolution means a special resolution: (a) passed by a majority of at least two-thirds of such Shareholders as, being entitled to do so, vote in person or, where proxies are allowed, by proxy, at a general meeting of the Company of which notice specifying the intention to propose the resolution as a special resolution has been duly given and where a poll is taken regard shall be had in computing a majority to the number of votes to which each Shareholder is entitled; or (b) approved in writing by all the Shareholders entitled to vote at a general meeting of the Company in one or more instruments each signed by one or more of the Shareholders aforesaid, and the effective date of the resolution so adopted shall be the date on which the Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

6 instrument or the last of such instruments, if more than one, is executed; Treasury Shares means Shares that were previously issued but were purchased, redeemed, surrendered or otherwise acquired by the Company and not cancelled; and Virtual Meeting means any general meeting of the Company (or any meeting of the holders of any class of shares) at which the Shareholders (and any other permitted participants of such meeting, including without limitation the chairperson of the meeting and any Directors) are permitted to attend and participate solely by means of Communication Facilities. 3. In these Articles, unless there be something in the subject or context inconsistent with such construction: (a) words importing the singular number shall include the plural number and vice versa; (b) words importing a gender shall include other genders; (c) words importing persons only shall include companies, partnerships, trusts or associations or bodies of persons, whether corporate or not; (d) the word "may" shall be construed as permissive and the word "shall" shall be construed as imperative; (e) the word "year" shall mean calendar year, the word "quarter" shall mean calendar quarter and the word "month" shall mean calendar month; (f) a reference to a "dollar" or "$" is a reference to the legal currency of the United States of America; (g) a reference to any enactment includes a reference to any modification or re-enactment thereof for the time being in force; (h) a reference to any meeting (whether of the Directors, a committee appointed by the Board of Directors or the Shareholders or any class of Shareholders) includes any adjournment of that meeting; (i) Sections 8 and 19 of the Electronic Transactions Act shall not apply; (j) a reference to "written" or "in writing" includes a reference to all modes of representing or reproducing words in visible form, including in the form of an Electronic Record; and (k) the term "holder" in relation to a Share means a person whose name is entered in the Register of Members as the holder of such Share. 4. Subject to the two preceding Articles, any words defined in the Companies Act shall, if not inconsistent with the subject or context, bear the same meaning in these Articles. 5. The table of contents to, and the headings in, these Articles are for convenience of reference only and are to be ignored in construing these Articles. COMMENCEMENT OF BUSINESS 6. The business of the Company may be conducted as the Board of Directors shall see fit. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

7 SITUATION OF REGISTERED OFFICE 7. The Registered Office shall be at such address in the Cayman Islands as the Directors shall from time to time determine. The Company, in addition to the Registered Office, may establish and maintain such other offices and places of business and agencies in such places as the Directors may from time to time determine. SHARES 8. The Directors may impose such restrictions as they think necessary on the offer and sale of any Shares. 9. Subject to these Articles (including Article 21(c)(iv)) and to any direction that may be given by the Shareholders in a general meeting, and without prejudice to any rights previously conferred on the holders of existing Shares, all Shares for the time being unissued shall be under the control of the Directors who may issue, allot and dispose of or grant options over the same and issue warrants or similar instruments with respect thereto to such persons, on such terms, and with or without preferred, deferred or other rights and restrictions, whether in regard to dividend, voting, return of capital or otherwise, and otherwise in such manner as they may think fit. For such purposes, the Directors may reserve an appropriate number of Shares for the time being unissued. No holder of Ordinary Shares shall have pre-emptive rights. 10. Subject to the Companies Act, and without prejudice to any rights previously conferred on the holders of existing Shares, any share or fraction of a share in the Company's share capital may be issued either at a premium or at par, and with such preferred, deferred, other special rights, or restrictions, whether in regard to dividend, voting, return of share capital or otherwise, as the Board of Directors may from time to time by resolution determine, and any share may be issued by the Directors on the terms that it is, or at the option of the Directors is liable, to be redeemed or purchased by the Company whether out of capital in whole or in part or otherwise. No Share may be issued at a discount except in accordance with the Companies Act. Except as set forth otherwise in Article 21(c)(iv), the Directors may provide, out of the unissued shares (other than unissued Ordinary Shares), for series of preference shares. Before any preference shares of any such series are issued, the Directors shall fix, by resolution or resolutions of the Board of Directors, the following provisions of the preference shares thereof, if applicable: (a) the designation of such series, the number of preference shares to constitute such series and the subscription price thereof if different from the par value thereof; (b) whether the preference shares of such series shall have voting rights, in addition to any voting rights provided by law, and, if so, the terms of such voting rights, which may be general or limited; (c) the dividends, if any, payable on such series, whether any such dividends shall be cumulative, and, if so, from what dates, the conditions and dates upon which such dividends shall be payable, and the preference or relation which such dividends shall bear to the dividends payable on any Shares of any other class or any other series of preference shares; (d) whether the preference shares of such series shall be subject to redemption by the Company, and, if so, the times, prices and other conditions of such redemption; (e) the amount or amounts payable upon preference shares of such series upon, and the rights of the holders of such series in, a voluntary or involuntary liquidation, dissolution or winding up, or upon any distribution of the assets, of the Company; (f) whether the preference shares of such series shall be subject to the operation of a retirement or sinking fund and, if so, the extent to and manner in which any such retirement or sinking fund shall be applied to the purchase or redemption of the Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

8 preference shares of such series for retirement or other corporate purposes and the terms and provisions relative to the operation thereof; (g) whether the preference shares of such series shall be convertible into, or exchangeable for, Shares of any other class or any other series of preference shares or any other securities and, if so, the price or prices or the rate or rates of conversion or exchange and the method, if any, of adjusting the same, and any other terms and conditions of conversion or exchange; (h) the limitations and restrictions, if any, to be effective while any preference shares of such series are outstanding upon the payment of dividends or the making of other distributions on, and upon the purchase, redemption or other acquisition by the Company of, the existing Shares or Shares of any other class or any other series of preference shares; (i) the conditions or restrictions, if any, upon the creation of indebtedness of the Company or upon the issue of any additional Shares, including additional preference shares of such series or of any other class of Shares or any other series of preference shares; and (j) any other powers, preferences and relative, participating, optional and other special rights, and any qualifications, limitations and restrictions thereof. Without limiting the foregoing and subject to Article 21(c)(iv) and Article 86, the voting powers of any series of preference shares may include the right, in the circumstances specified in the resolution or resolutions of the Board of Directors providing for the issuance of such preference shares, to elect one or more Directors who shall serve for such term and have such voting powers as shall be stated in the resolution or resolutions of the Board of Directors providing for the issuance of such preference shares. The term of office and voting powers of any Director elected in the manner provided in the immediately preceding sentence of this Article 10 may be greater than or less than those of any other Director or class of Directors. The powers, preferences and relative, participating, optional and other special rights of each series of preference shares, and the qualifications, limitations or restrictions thereof, if any, may differ from those of any and all other series at any time outstanding. All Shares of any one series of preference shares shall be identical in all respects with all other Shares of such series, except that Shares of any one series issued at different times may differ as to the dates from which dividends thereon shall be cumulative. 11. The Directors may in their absolute discretion refuse to accept any application for Shares and may accept any application in whole or in part. 12. The Company may on any issue of Shares deduct any sales charge or subscription fee from the amount subscribed for the Shares. 13. No person shall be recognised by the Company as holding any Share upon any trust (other than any trust recognized as a Permitted Entity or Permitted Transferee), and the Company shall not be bound by or recognise (even when having notice thereof) any equitable, contingent, future or partial interest in any Share, or (except as otherwise provided by these Articles or as required by law) any other right in respect of any Share except an absolute right thereto in the registered holder, provided that, notwithstanding the foregoing, the Company shall be entitled to recognise any such interests as shall be determined by the Directors. 14. The Directors shall keep or cause to be kept a Register of Members as required by the Companies Act at such place or places as the Directors may from time to time determine. In the absence of any such determination, the Register of Members shall be kept at the Registered Office. Title to Shares may be evidenced and transferred in accordance with the laws applicable to and the rules and regulations of the Designated Stock Exchange. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

9 15. The Directors in each year shall prepare or cause to be prepared an annual return and declaration setting forth the particulars required by the Companies Act in respect of exempted companies and deliver a copy thereof to the Registrar of Companies in the Cayman Islands. 16. The Company shall not issue Shares to bearer. 17. The Directors may issue fractions of a Share and, if so issued, a fraction of a Share shall be subject to and carry the corresponding fraction of liabilities (whether with respect to nominal or par value, premium, calls or otherwise howsoever), limitations, preferences, privileges, qualifications, restrictions, rights (including, without prejudice to the foregoing generality, voting and participation rights) and other attributes of a Share. If more than one fraction of a Share is issued to or acquired by the same Shareholder, such fractions shall be accumulated. 18. The premium arising on all issues of Shares shall be held in the Share Premium Account established in accordance with these Articles. 19. The Company may, insofar as permitted by law, pay a commission to any person in consideration of his subscribing or agreeing to subscribe whether absolutely or conditionally for any Shares. Such commissions may be satisfied by the payment of cash or the lodgement of fully or partly paid-up Shares or partly in one way and partly in the other. The Company may also on any issue of Shares pay such brokerage as may be lawful. 20. Payment for Shares shall be made at such time and place and to such person on behalf of the Company as the Directors may from time to time determine. Payment for any Shares shall be made in such currency as the Directors may determine from time to time, provided that the Directors shall have the discretion to accept payment in any other currency or in kind or a combination of cash and in kind. 21. Rights and Restrictions Attaching to Ordinary Shares: Except as otherwise provided in these Articles (including Articles 21(c)(iv), 21(d) and 86), the Class A Ordinary Shares and Class B Ordinary Shares have the same rights and powers, and rank equally (including as to dividends and distributions, and upon the occurrence of any liquidation or winding up of the Company), share ratably and are identical in all respects and as to all matters, unless different treatment of the Shares of each such class is approved by the affirmative vote of the holders of a majority of the Class A Ordinary Shares and the holders of a majority of the Class B Ordinary Shares, each voting exclusively and as a separate class. (a) Income: Holders of Ordinary Shares shall be entitled to such dividends as the Directors may in their absolute discretion lawfully declare from time to time. (b) Capital: Holders of Ordinary Shares shall be entitled to a return of capital on liquidation, dissolution or winding-up of the Company in accordance with Article 171 et seq. (c) Attendance at General Meetings; Class Voting: (i) Holders of Ordinary Shares have the right to receive notice of, attend, speak and vote at general meetings of the Company. (ii) Except as otherwise provided in these Articles (including Article 21(c)(iv)), holders of Class A Ordinary Shares and Class B Ordinary Shares shall at all times vote together as one class on all matters submitted to a vote for Shareholders’ consent. (iii) On all matters subject to a vote of the Shareholders, Ordinary Shares shall be entitled to voting rights as set forth in Article 86. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

10 (iv) Subject to applicable law, in addition to any rights provided by applicable law or otherwise set forth in these Articles, the Company shall not, without the approval by vote or written consent of the holders of a majority of the voting power of the Class B Ordinary Shares, voting exclusively and as a separate class, directly or indirectly, or whether by amendment or through merger, recapitalization, consolidation or otherwise: (1) increase the number of authorized Class B Ordinary Shares; (2) issue any Class B Ordinary Shares or securities convertible into or exchangeable for Class B Ordinary Shares, other than (i) to any Key Executive or his or her Affiliates, or (ii) on a pro rata basis to all holders of Class B Ordinary Shares permitted to hold such shares under these Articles; (3) create, authorize, issue, or reclassify into, any preference shares in the capital of the Company or any Shares in the capital of the Company that carry more than one (1) vote per share; (4) reclassify any Class B Ordinary Shares into any other class of Shares or consolidate or combine any Class B Ordinary Shares without proportionately increasing the number of votes per Class B Ordinary Share; or (5) amend, restate, waive, adopt any provision inconsistent with or otherwise vary or alter any provision of the Memorandum or these Articles relating to the voting, conversion or other rights, powers, preferences, privileges or restrictions of the Class B Ordinary Shares. (d) Optional and Automatic Conversion of Class B Ordinary Shares: (i) Each Class B Ordinary Share is convertible into one (1) Class A Ordinary Share (as adjusted for share splits, share combinations and similar transactions occurring after the Acquisition Effective Time) at any time at the option of the holder thereof. In no event shall any Class A Ordinary Share be convertible into any Class B Ordinary Shares. (ii) Any number of Class B Ordinary Shares held by a holder thereof will be automatically and immediately converted into an equal number of Class A Ordinary Shares upon the occurrence of any of the following: (1) Any direct or indirect sale, transfer, assignment, or disposition of such number of Class B Ordinary Shares by the holder thereof or the direct or indirect transfer or assignment of the voting power attached to such number of Class B Ordinary Shares through voting proxy or otherwise to any person that is not an Permitted Transferee of such holder; for the avoidance of doubt, the creation of any pledge, charge, encumbrance, or other third party right of whatever description on any of Class B Ordinary Shares to secure contractual or legal obligations shall not be deemed as a sale, transfer, assignment, or disposition under this Article Error! Reference source not found. unless and until any such pledge, charge, encumbrance, or other third party right is enforced and results in a third party that is not an Permitted Transferee of such holder holding directly or indirectly legal or beneficial ownership or voting power through voting proxy or otherwise to the related Class B Ordinary Shares, in which case all the related Class B Ordinary Shares Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

11 shall be automatically converted into the same number of Class A Ordinary Shares; (2) The direct or indirect sale, transfer, assignment, or disposition of a majority of the issued and outstanding voting securities of, or the direct or indirect transfer or assignment of the voting power attached to such voting securities through voting proxy or otherwise, or the direct or indirect sale, transfer, assignment, or disposition of all or substantially all of the assets of, a holder of Class B Ordinary Shares that is an entity to any person that is not an Permitted Transferee of the such holder; for the avoidance of doubt, the creation of any pledge, charge, encumbrance, or other third party right of whatever description on the issued and outstanding voting securities or the assets of a holder of Class B Ordinary Shares to secure contractual or legal obligations shall not be deemed as a sale, transfer, assignment, or disposition under this Article Error! Reference source not found. unless and until any such pledge, charge, encumbrance or other third party right is enforced and results in a third party that is not an Permitted Transferee of such holder holding directly or indirectly legal or beneficial ownership or voting power through voting proxy or otherwise to the related issued and outstanding voting securities or the assets; or (3) Notwithstanding the foregoing, if a person becomes a holder of Class B Ordinary Shares by will or intestacy, then the Class B Ordinary Shares transferred to such holder by will or intestacy shall be automatically converted into the same number of Class A Ordinary Shares. (iii) Notwithstanding Article 21(d)(ii), all Class B Ordinary Shares issued and outstanding will be automatically and immediately converted into an equal number of Class A Ordinary Shares upon the occurrence of any of the following: (1) on Danny Yeung’s death or Incapacity; (2) on the date on which Danny Yeung is terminated for cause (as defined in the employment agreement with Danny Yeung (and in the event of a dispute regarding whether there was cause, cause will be deemed not to exist unless and until an affirmative ruling regarding such cause has been made by a court or arbitral panel of competent jurisdiction, and such ruling has become final and non-appealable)); or (3) on the first date that both of the following conditions are satisfied: (I) Danny Yeung and his Affiliates and Permitted Transferees together own less than thirty three per cent (33%) of the number of Class B Ordinary Shares (which for these purposes shall be deemed to include all Class B Ordinary Shares issuable upon exercise of all outstanding restricted share units to acquire Class B Ordinary Shares that are held by Danny Yeung immediately following the Acquisition Effective Time) that Danny Yeung and his Affiliates and Permitted Transferees owned immediately following the Acquisition Effective Time, as adjusted for share splits, share combinations and similar transactions occurring after the Acquisition Effective Time; and (II) Danny Yeung ceases to be a Director or officer of the Company. (iv) No Class B Ordinary Shares shall be issued by the Company after conversion of all Class B Ordinary Shares into Class A Ordinary Shares. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

12 (e) Procedure of Conversion. Any conversion of Class B Ordinary Shares into Class A Ordinary Shares pursuant to these Articles shall be effected by means of either: (i) the re-designation and re-classification of each relevant Class B Ordinary Share as a Class A Ordinary Share, such conversion to become effective forthwith upon entries being made in the Register of Members to record the re-designation and re-classification of the relevant Class B Ordinary Shares as Class A Ordinary Shares; or (ii) the compulsory redemption without notice of Class B Ordinary Shares of any Class B Ordinary Shareholder and, on behalf of such Shareholder, automatic application of such redemption proceeds in paying for such new Class A Ordinary Shares into which the Class B Ordinary Shares have been converted or exchanged at a price per Class B Ordinary Share necessary to give effect to a conversion or exchange calculated on the basis that the Class A Ordinary Shares to be issued as part of the conversion or exchange will be issued at par. The Class A Ordinary Shares to be issued on an exchange or conversion shall be registered in the name of such Shareholder or in such name as the Shareholder may direct in the Register of Members. (f) Reservation of Class A Ordinary Shares Issuable upon Conversion of Class B Ordinary Shares. The Company shall at all times reserve and keep available out of its authorized but unissued Class A Ordinary Shares, solely for the purpose of effecting the conversion of the Class B Ordinary Shares, such number of its Class A Ordinary Shares as shall from time to time be sufficient to effect the conversion of all outstanding Class B Ordinary Shares; and if at any time the number of authorized but unissued Class A Ordinary Shares shall not be sufficient to effect the conversion of all then-outstanding Class B Ordinary Shares, the Company will take such corporate action as may, in the opinion of its counsel, be necessary to increase its authorized but unissued Class A Ordinary Shares to such numbers of shares as shall be sufficient for such purpose. REDEMPTION, PURCHASE AND SURRENDER OF SHARES 22. Subject to the Companies Act, the Company may: (a) issue Shares on terms that they are to be redeemed or are liable to be redeemed at the option of the Company and/or the Shareholder on such terms and in such manner as the Company may, before the issue of such Shares, determine by either resolution of the Board of Directors or by Special Resolution; (b) purchase its own Shares (including any redeemable Shares) on such terms and in such manner agreed with the relevant Shareholder as have been approved by the Directors or by the Shareholders by Ordinary Resolution, or are otherwise authorized by these Articles; and (c) make a payment in respect of the redemption or purchase of Shares in any manner authorised by the Companies Act, including out of its capital, profits or the proceeds of a fresh issue of Shares. 23. Unless the Directors determine otherwise, any Share in respect of which notice of redemption has been given shall not be entitled to participate in the profits of the Company in respect of the period after the date specified as the date of redemption in the notice of redemption. 24. The redemption, purchase or surrender of any Share shall not be deemed to give rise to the redemption, purchase or surrender of any other Share. 25. The Directors may when making payments in respect of a redemption or purchase of Shares, if authorised by the terms of issue of the Shares being redeemed or purchased or with the agreement of the holder of such Shares, make such payment either in cash or in specie. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

13 26. Subject to the Companies Act, the Company may accept the surrender for no consideration of any fully paid Share (including any redeemable Share) on such terms and in such manner as the Directors may determine. TREASURY SHARES 27. Shares that the Company purchases, redeems or acquires (by way of surrender or otherwise) may, at the option of the Company, be cancelled immediately or held as Treasury Shares in accordance with the Companies Act. In the event that the Directors do not specify that the relevant Shares are to be held as Treasury Shares, such Shares shall be cancelled. 28. No dividend may be declared or paid, and no other distribution (whether in cash or otherwise) of the Company's assets (including any distribution of assets to Shareholders on a winding up) may be declared or paid in respect of a Treasury Share. 29. The Company shall be entered in the Register of Members as the holder of the Treasury Shares, provided that: (a) the Company shall not be treated as a Shareholder for any purpose and shall not exercise any right in respect of the Treasury Shares, and any purported exercise of such a right shall be void; and (b) a Treasury Share shall not be voted, directly or indirectly, at any meeting of the Company and shall not be counted in determining the total number of issued shares at any given time, whether for the purposes of these Articles or the Companies Act, save that an allotment of Shares as fully paid bonus shares in respect of Treasury Shares is permitted and Shares allotted as fully paid bonus shares in respect of Treasury Shares shall be treated as Treasury Shares. 30. Treasury Shares may be disposed of by the Company on any terms and conditions determined by the Directors. MODIFICATION OF RIGHTS 31. Subject to Article 21(c)(iv), if at any time the share capital of the Company is divided into different classes of Shares, the rights attached to any class (unless otherwise provided by the terms of issue of the Shares of that class) may, whether or not the Company is being wound up, be varied or abrogated without the consent of the holders of the issued Shares of that class where such variation or abrogation is considered by the Directors not to have a material adverse effect upon such rights; otherwise, any such variation or abrogation shall be made only with the consent in writing of the holders of not less than two-thirds of the issued Shares of that class, or with the approval of a resolution passed by a majority of not less than two-thirds of the votes cast at a separate meeting of the holders of the Shares of that class. For the avoidance of doubt, the Directors reserve the right, notwithstanding that any such variation or abrogation may not have a material adverse effect, to obtain consent from the holders of Shares of the relevant class. 32. The provisions of these Articles relating to general meetings shall apply, mutatis mutandis, to every such meeting of the holders of one class of Shares except the following: (a) separate meetings of the holders of a class of Shares may be called only by: (i) the Chairperson; (ii) a majority of the entire Board of Directors (unless otherwise specifically provided by the terms of issue of the Shares of such class); or Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

14 (iii) with respect to meetings of the holders of Class B Ordinary Shares, Danny Yeung; (b) except as set forth in clause (a) above or provided in Article 70 below, nothing in this Article 32 or in Article 31 shall be deemed to give any Shareholder or Shareholders the right to call a class or series meeting; and (c) the necessary quorum shall be one or more persons holding or representing by proxy at least one-third of the issued Shares of the class (but if at any adjourned meeting of such holders a quorum as aforementioned is not present, those Shareholders who are present in person or by proxy shall form a quorum). 33. For the purposes of Articles 31 and 32, the Directors may treat all classes of Shares, or any two classes of Shares, as forming a single class if they consider that each class would be affected in the same way by the proposal or proposals under consideration. In any other case, the Directors shall treat all classes of Shares, or any two classes of Shares, as separate classes. 34. The rights conferred upon the holders of the Shares of any class shall not, unless otherwise expressly provided by the terms of issue of the Shares of that class, be deemed to be varied by the creation or issue of further Shares ranking in priority thereto or pari passu therewith. SHARE CERTIFICATES 35. The Shares will be issued in fully registered, book-entry form. Certificates will not be issued unless the Directors determine otherwise. Share certificates (if any) shall specify the Share or Shares held by that Shareholder and the amount paid up thereon; provided, that in respect of a Share or Shares held jointly by several persons the Company shall not be bound to issue more than one certificate, and delivery of a certificate for a Share to one of several joint holders shall be sufficient delivery to all. All certificates for Shares shall be delivered personally or sent through the post addressed to the Shareholder entitled thereto at the Shareholder’s registered address as appearing in the Register of Members. All share certificates shall bear legends required under the applicable laws, including the Securities Act. Any two or more certificates representing Shares of any one class held by any Shareholder may at the Shareholder’s request be cancelled and a single new certificate for such Shares issued in lieu on payment (if the Directors shall so require) of $1.00 or such smaller sum as the Directors shall determine. 36. If a share certificate shall be damaged or defaced or alleged to have been lost, stolen or destroyed, a new certificate representing the same Shares may be issued to the relevant Shareholder upon request subject to delivery up of the old certificate or (if alleged to have been lost, stolen or destroyed) compliance with such conditions as to evidence and indemnity and the payment of out-of-pocket expenses of the Company in connection with the request as the Directors may think fit. In the event that Shares are held jointly by several persons, any request may be made by any one of the joint holders and if so made shall be binding on all of the joint holders. TRANSFER AND TRANSMISSION OF SHARES 37. Any Shareholder may transfer all or any of its Shares by an instrument of transfer in the usual or common form in use in the Cayman Islands, in a form prescribed by the Designated Stock Exchange or in any other form approved by the Board of Directors and may be under hand or, if the transferor or transferee is a clearing house or its nominee(s), by hand or by machine imprinted signature or by such other manner of execution as the Board of Directors may approve from time to time. 38. The Directors shall not refuse to register any transfer of a Share which is permitted under these Articles save that the Directors may decline to register any transfer of any Share in the event that any of the following is known by the Directors not to be both applicable and true with respect to such transfer: Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

15 (a) the instrument of transfer is lodged with the Company, or the designated transfer agent or share registrar, accompanied by the certificate for the shares to which it relates (if any) and such other evidence as the Board of Directors may reasonably require to show the right of the transferor to make the transfer; (b) the instrument of transfer is in respect of only one class of Shares; (c) the instrument of transfer is properly stamped, if required; (d) the transferred Shares are fully paid up and free of any lien in favor of the Company (it being understood and agreed that all other liens, including pursuant to a bona fide loan or indebtedness transaction, shall be permitted); and (e) a fee of such maximum sum as the Designated Stock Exchange may determine to be payable, or such lesser sum as the Board of Directors may from time to time require, is paid to the Company in respect thereof. 39. If the Directors refuse to register a transfer they shall, within two months after the date on which the instrument of transfer was lodged, send to each of the transferor and the transferee notice of such refusal stating the facts which are considered to justify the refusal to register the transfer. 40. The registration of transfers may, on 14 calendar days’ notice being given by advertisement in such one or more newspapers or by electronic means, be suspended and the Register of Members closed at such times and for such periods as the Board of Directors may from time to time determine; provided, however, that the registration of transfers shall not be suspended nor the Register of Members closed for more than 30 calendar days in any year. 41. An instrument of transfer must be executed by or on behalf of the transferor (and if in respect of a nil or partly paid up Share or the Directors so require, signed by the transferee). Such instrument of transfer must be accompanied by such evidence as the Directors may reasonably require to show the right of the transferor to make the transfer and the transferor is deemed to remain the holder until the transferee’s name is entered in the Register of Members. The instrument of transfer must be completed and signed in the exact name or names in which such Shares are registered, indicating any special capacity in which it is being signed with relevant details supplied to the Company. 42. All instruments of transfer which are registered shall be retained by the Company, but any instrument of transfer which the Directors may decline to register shall (except in any case of fraud) be returned to the person depositing the same. 43. In case of the death of a Shareholder, the survivors or survivor (where the deceased was a joint holder) and the executors or administrators of the deceased where the deceased was the sole or only surviving holder, shall be the only persons recognised by the Company as having title to the deceased's interest in the Shares, but nothing in this Article shall release the estate of the deceased holder whether sole or joint from any liability in respect of any Share solely or jointly held by the deceased. 44. Any guardian of an infant Shareholder and any curator or other legal representative of a Shareholder under legal disability and any person entitled to a Share in consequence of the death or bankruptcy of a Shareholder shall, upon producing such evidence of title as the Directors may require, have the right either to be registered as the holder of the Share or to make such transfer thereof as the deceased or bankrupt Shareholder could have made, but the Directors shall in either case have the same right to refuse or suspend registration as they would have had in the case of a transfer of the Shares by the infant or by the deceased or bankrupt Shareholder before the death or bankruptcy or by the Shareholder under legal disability before such disability. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

16 45. A person so becoming entitled to a Share in consequence of the death or bankruptcy of a Shareholder shall have the right to receive and may give a discharge for all dividends and other money payable or other advantages due on or in respect of the Share, but such person shall not be entitled to receive notice of or to attend or vote at meetings of the Company, or save as aforesaid, to any of the rights or privileges of a Shareholder unless and until such person shall be registered as a Shareholder in respect of the Share, provided always that the Directors may at any time give notice requiring any such person to elect either to be registered or to transfer the Share and if the notice is not complied with within ninety (90) calendar days the Directors may thereafter withhold all dividends or other monies payable or other advantages due in respect of the Share until the requirements of the notice have been complied with. 46. The transferor shall be deemed to remain a holder of the Share until the name of the transferee is entered in the Register of Members in respect of the relevant Share. LIEN 47. The Company shall have a first and paramount lien on all Shares (whether fully paid-up or not) registered in the name of a Shareholder (whether solely or jointly with others) for all debts, liabilities or engagements to or with the Company (whether presently payable or not) by such Shareholder or the Shareholder's estate, either alone or jointly with any other person, whether a Shareholder or not, but the Directors may at any time declare any Share to be wholly or in part exempt from the provisions of this Article. The registration of a transfer of any such Share shall operate as a waiver of the Company's lien (if any) thereon. The Company's lien (if any) on a Share shall also extend to all dividends or any amount payable in respect of that Share. 48. The Company may sell, in such manner as the Directors think fit, any Shares on which the Company has a lien, if a sum in respect of which the lien exists is presently payable, and is not paid within fourteen (14) calendar days after notice has been given to the holder of the Shares, or to the person entitled to it in consequence of the death or bankruptcy of the holder, demanding payment and stating that if the notice is not complied with the Shares may be sold. 49. To give effect to any such sale the Directors may authorise any person to execute an instrument of transfer of the Shares sold to, or in accordance with the directions of, the purchaser. The purchaser or the purchaser's nominee shall be registered as the holder of the Shares comprised in any such transfer, and the purchaser shall not be bound to see to the application of the purchase money, nor shall the purchaser's title to the Shares be affected by any irregularity or invalidity in the sale or the exercise of the Company's power of sale under these Articles. 50. The net proceeds of such sale, after payment of costs, shall be applied in payment of such part of the amount in respect of which the lien exists as is presently payable and any residue shall (subject to a like lien for sums not presently payable as existed upon the Shares before the sale) be paid to the person entitled to the Shares at the date of the sale. CALL ON SHARES 51. Subject to the terms of the allotment the Directors may from time to time make calls upon the Shareholders in respect of any monies unpaid on their Shares (whether in respect of par value or premium), and each Shareholder shall (subject to receiving at least fourteen (14) calendar days' notice specifying the time or times of payment) pay to the Company at the time or times so specified the amount called on the Shares. A call may be revoked or postponed as the Directors may determine. A call may be required to be paid by instalments. A person upon whom a call is made shall remain liable for calls made upon them notwithstanding the subsequent transfer of the Shares in respect of which the call was made. 52. A call shall be deemed to have been made at the time when the resolution of the Directors authorising such call was passed. 53. The joint holders of a Share shall be jointly and severally liable to pay all calls in respect thereof. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

17 54. If a sum called in respect of a Share is not paid before or on the day appointed for payment thereof, the person from whom the sum is due shall pay interest upon the sum at the rate of eight percent per annum from the day appointed for the payment thereof to the time of the actual payment, but the Directors shall be at liberty to waive payment of that interest wholly or in part. 55. An amount payable in respect of a Share on allotment or at any fixed date, whether on account of the par value of the Share or premium or otherwise, shall be deemed to be a call and if it is not paid all the provisions of these Articles shall apply as if that amount had become due and payable by virtue of a call. 56. The Directors may make arrangements on the issue of Shares for a difference between the Shareholders as to the amount and times of payment of calls, or the interest to be paid. 57. The Directors may, if they think fit, receive an amount from any Shareholder willing to advance all or any part of the monies uncalled and unpaid upon any Shares held by such Shareholder, and upon all or any of the monies so advanced may (until the same would, but for such advance, become presently payable) pay interest at such rate (not exceeding without the sanction of an Ordinary Resolution, eight percent per annum) as may be agreed upon between the Directors and the Shareholder paying such amount in advance. 58. No such amount paid in advance of calls shall entitle the Shareholder paying such amount to any portion of a dividend declared in respect of any period prior to the date upon which such amount would, but for such payment, become payable. FORFEITURE OF SHARES 59. If a call remains unpaid after it has become due and payable the Directors may give to the person from whom it is due not less than fourteen (14) calendar days' notice requiring payment of the amount unpaid together with any interest which may have accrued. The notice shall specify where payment is to be made and shall state that if the notice is not complied with the Shares in respect of which the call was made will be liable to be forfeited. 60. If the notice is not complied with any Share in respect of which it was given may, before the payment required by the notice has been made, be forfeited by a resolution of the Directors. Such forfeiture shall include all dividends or other monies declared payable in respect of the forfeited Share and not paid before the forfeiture. 61. A forfeited Share may be sold, re-allotted or otherwise disposed of on such terms and in such manner as the Directors think fit and at any time before a sale, re-allotment or disposition the forfeiture may be cancelled on such terms as the Directors think fit. Where for the purposes of its disposal a forfeited Share is to be transferred to any person the Directors may authorise some person to execute an instrument of transfer of the Share in favour of that person. 62. A person any of whose Shares have been forfeited shall cease to be a Shareholder in respect of them and shall surrender to the Company for cancellation the certificate for the Shares forfeited and shall remain liable to pay to the Company all monies which at the date of forfeiture were payable by such person to the Company in respect of those Shares together with interest, but such person's liability shall cease if and when the Company shall have received payment in full of all monies due and payable by such person in respect of those Shares. 63. A certificate in writing under the hand of one Director or officer of the Company that a Share has been forfeited on a specified date shall be conclusive evidence of the fact as against all persons claiming to be entitled to the Share. The certificate shall (subject to the execution of any instrument of transfer) constitute a good title to the Share and the person to whom the Share is disposed of shall not be bound to see to the application of the purchase money, if any, nor shall such person's title to the Share be affected by any irregularity or invalidity in the proceedings in reference to the forfeiture, sale or disposal of the Share. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

18 64. The provisions of these Articles as to forfeiture shall apply in the case of non-payment of any sum which, by the terms of issue of a Share, becomes payable at a fixed time, whether on account of the par value of the Share or by way of premium as if it had been payable by virtue of a call duly made and notified. ALTERATION OF SHARE CAPITAL 65. Subject to the rights of Class B Ordinary Shares, including under Article 21(c)(iv), the Company may from time to time by Ordinary Resolution: (a) increase its share capital by such sum to be divided into Shares of such amounts as the resolution shall prescribe; (b) consolidate and divide all or any of its share capital into Shares of a larger amount than its existing Shares; (c) sub-divide its existing Shares or any of them into Shares of a smaller amount; provided, that in the subdivision the proportion between the amount paid and the amount, if any, unpaid on each reduced Share shall be the same as it was in case of the Share from which the reduced Share is derived; or (d) cancel any Shares which, at the date of the passing of the resolution, have not been taken or agreed to be taken by any person and diminish the amount of its share capital by the amount of the Shares so cancelled. 66. All new Shares created hereunder shall be subject to the same provisions with reference to the payment of calls, liens, transfer, transmission, forfeiture, and otherwise as the Shares in the original share capital. 67. Subject to the Companies Act and the rights of Class B Ordinary Shares, including under Article 21(c)(iv), the Company may by Special Resolution from time to time reduce its share capital and any capital redemption reserve in any way, and in particular, without prejudice to the generality of the foregoing power, may: (a) cancel any paid-up share capital which is lost, or which is not represented by available assets; or (b) pay off any paid-up share capital which is in excess of the requirements of the Company, and may, if and so far as is necessary, alter the Memorandum by reducing the amounts of its share capital and of its Shares accordingly. GENERAL MEETINGS 68. All general meetings of the Company other than annual general meetings shall be called extraordinary general meetings. The Company shall hold an annual general meeting and shall specify the meeting as such in the notices calling it. The annual general meeting shall be held at such time and place as the Directors shall determine. At these annual general meetings, the report of the Directors (if any) shall be presented. 69. The Directors may proceed to convene a general meeting whenever they think fit, including, without limitation, for the purposes of considering a liquidation of the Company, and they shall convene a general meeting on the requisition of the Shareholders in accordance with these Articles. 70. A Shareholders requisition is a requisition in writing of: Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

19 (a) Shareholders holding at the date of deposit of the requisition not less than one third of the votes that may be cast by all of the issued share capital of the Company as at that date carries the right of voting at general meetings of the Company; or (b) the holders of Class B Ordinary Shares entitled to cast a majority of the votes that all Class B Ordinary Shares are entitled to cast. 71. The requisition must state the objects of the meeting and must be signed by the requisitionists and deposited at the principal place of business of the Company (with a copy forwarded to the Registered Office), and may consist of several documents in like form each signed by one or more requisitionists. 72. If the Directors do not within 21 calendar days from the date of the deposit of the requisition duly proceed to convene a general meeting to be held within a further 21 calendar days, the requisitionists, or any of them representing more than one half of the total voting rights of all of them, may themselves convene a general meeting, but any meeting so convened shall not be held after the expiration of three months after the expiration of the second said 21 calendar days. 73. A general meeting convened as aforesaid by requisitionists shall be convened in the same manner as nearly as possible as that in which general meetings are convened by the Directors. A general meeting may be convened in the Cayman Islands or at such other location, as the Directors think fit. NOTICE OF GENERAL MEETINGS 74. At least seven (7) calendar days’ notice in writing shall be given for any general meeting. Every notice shall be exclusive of the day on which it is given or deemed to be given and shall specify the place, the day and the hour of the meeting and the general nature of the business to be conducted at the meeting and shall be given in the manner hereinafter mentioned or in such other manner if any as may be prescribed by the Company by Ordinary Resolution, provided, that a general meeting of the Company shall, whether or not the notice specified in this Article 74 has been given and whether or not the provisions of these Articles regarding general meetings have been complied with, be deemed to have been duly convened if it is so agreed: (a) in the case of an annual general meeting by all the Shareholders (or their proxies) entitled to attend and vote thereat; and (b) in the case of an extraordinary general meeting, by Shareholders (or their proxies) having a right to attend and vote at the meeting, together holding Shares entitling the holders thereof to not less than two-thirds of the votes entitled to be cast at such extraordinary general meeting. 75. The accidental omission to give notice of a general meeting to or the non-receipt of a notice of a general meeting by any person entitled to receive such notice shall not invalidate the proceedings at that general meeting. PROCEEDINGS AT GENERAL MEETINGS 76. No business shall be transacted at any general meeting unless a quorum is present at the time when the meeting proceeds to business. One or more Shareholders holding not less than one- third of the total issued share capital of the Company in issue present in person or by proxy and entitled to vote shall be a quorum for all purposes; provided, that, from and after the Acquisition Effective Time where there are Class B Ordinary Shares in issue, the presence in person or by proxy of holders of a majority of the issued Class B Ordinary Shares shall be required in any event. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

20 77. Save as otherwise provided for in these Articles, if within half an hour from the time appointed for the meeting a quorum is not present, the meeting, if convened on the requisition of or by Shareholders, shall be dissolved. In any other case it shall stand adjourned to the same day in the next week, at the same time and place or to such other day and at such other time and place as the Chairperson may determine and if at such adjourned meeting a quorum is not present within half an hour from the time appointed for holding the meeting, the meeting shall be dissolved. 78. If the Directors wish to make this facility available for a specific general meeting or all general meetings of the Company, attendance and participation in any general meeting of the Company may be by means of Communication Facilities. Without limiting the generality of the foregoing, the Directors may determine that any general meeting may be held as a Virtual Meeting. The notice of any general meeting at which Communication Facilities will be utilized (including any Virtual Meeting) must disclose the Communication Facilities that will be used, including the procedures to be followed by any Shareholder or other participant of the meeting who wishes to utilize such Communication Facilities for the purposes of attending and participating in such meeting, including attending and casting any vote thereat. 79. The Chairperson, if any, of the Board of Directors shall preside as chairperson at every general meeting. If there is no such Chairperson, or if at any general meeting the appointed chairperson is not present within fifteen (15) minutes after the time appointed for holding the meeting or is unwilling to act as chairperson of the meeting, any Director or person nominated by the Directors shall preside as chairperson of that meeting, failing which the Shareholders present shall choose any person present to be chairperson of that meeting. 80. The chairperson of any general meeting (including any Virtual Meeting) shall be entitled to attend and participate at any such general meeting by means of Communication Facilities, and to act as the chairperson of such general meeting, in which event the following provisions shall apply: (a) The chairperson of the meeting shall be deemed to be present at the meeting; and (b) If the Communication Facilities are interrupted or fail for any reason to enable the chairperson of the meeting to hear and be heard by all other persons participating in the meeting, then the other Directors present at the meeting shall choose another Director present to act as chairperson of the meeting for the remainder of the meeting; provided that if no other Director is present at the meeting, or if all the Directors present decline to take the chair, then the meeting shall be automatically adjourned to the same day in the next week and at such time and place as shall be decided by the Board of Directors. 81. The chairperson of the general meeting may with the consent of any general meeting at which a quorum is present (and shall if so directed by the meeting) adjourn the meeting from time to time and from place to place (provided, that no general meeting called by a holder of Class B Ordinary Shares may be adjourned unless a quorum does not exist), but no business shall be transacted at any adjourned meeting except business which might lawfully have been transacted at the meeting from which the adjournment took place. When a meeting is adjourned for ten (10) calendar days or more, not less than seven (7) calendar days' notice in writing specifying the place, the day and the hour of the adjourned meeting shall be given as in the case of the original meeting but it shall not be necessary to specify in such notice the nature of the business to be transacted at the adjourned meeting. Save as aforesaid, it shall not be necessary to give any notice of an adjournment or of the business to be transacted at an adjourned meeting. 82. The Directors may cancel or postpone any duly convened general meeting at any time prior to such meeting, except for general meetings requisitioned by the Shareholders in accordance with these Articles, for any reason or for no reason, upon notice in writing to Shareholders. A postponement may be for a stated period of any length or indefinitely as the Directors may determine. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

21 83. At any general meeting a resolution put to the vote of the meeting shall be decided by way of a poll and not on a show of hands. 84. A poll shall be taken in such manner and at such place as the chairperson of the meeting may direct (including the use of a ballot or voting papers, or tickets) and the result of a poll shall be deemed to be the resolution of the meeting. 85. All questions submitted to a meeting shall be decided by an Ordinary Resolution except where a greater majority is required by these Articles or by the Companies Act. VOTES OF SHAREHOLDERS 86. Subject to any rights and restrictions for the time being attached to any class or classes of Shares, including in Articles 21(c)(iv) and 21(d), each Class A Ordinary Share shall be entitled to one (1) vote on all matters subject to a vote of the Shareholders, and each Class B Ordinary Share shall be entitled to twenty (20) votes on all matters subject to a vote of the Shareholders. 87. In the case of joint holders of a Share, the vote of the senior holder who tenders a vote, whether in person or by proxy, shall be accepted to the exclusion of the votes of the other joint holders, and for this purpose seniority shall be determined by the order in which the names stand in the Register of Members in respect of the Shares. 88. A Shareholder who has appointed special or general attorneys or a Shareholder who is subject to a disability may vote, by such Shareholder's attorney, committee, receiver, curator bonis or other person in the nature of a committee, receiver, or curator bonis appointed by a court and such attorney, committee, receiver, curator bonis or other person may vote by proxy; provided that such evidence as the Directors may require of the authority of the person claiming to vote shall, unless otherwise waived by the Directors, have been deposited at the Registered Office not less than forty eight (48) hours before the time for holding the meeting or adjourned meeting at which such person claims to vote. No Shareholder shall be entitled to vote at any general meeting unless all calls or other sums presently payable by him in respect of Shares in the Company have been paid. 89. No objection shall be raised to the qualification of any voter except at the meeting or adjourned meeting at which the vote objected to is given or tendered, and every vote not disallowed at such meeting shall be valid for all purposes. Any such objection made in due time shall be referred to the chairperson of the meeting, whose decision shall be final and conclusive. 90. On a poll votes may be given either personally or by proxy and a Shareholder entitled to more than one vote need not, if the Shareholder votes, use all their votes or cast all the votes the Shareholder uses in the same way. 91. The instrument appointing a proxy shall be in writing under the hand of the appointor or of the appointor's attorney duly authorised in writing, or if the appointor is a corporation, either under its common seal or under the hand of an officer or attorney so authorised. 92. Any person (whether a Shareholder or not) may be appointed to act as a proxy. A Shareholder may appoint more than one proxy to attend on the same occasion. Where a Shareholder appoints more than one proxy the instrument of proxy shall specify the number of Shares in respect of which each proxy is entitled to exercise the related votes. 93. The instrument appointing a proxy and the power of attorney or other authority (if any) under which it is signed, or a certified copy of such power or authority, must be deposited at the Registered Office, or at such other place as is specified for that purpose in the notice of meeting or in the instrument of proxy issued by the Company: (a) not less than 48 hours before the time for holding the general meeting or adjourned meeting at which the person named in the instrument proposes to vote; or Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

22 (b) not less than 24 hours before the time appointed for the taking of the poll, provided, that the Directors may in the notice convening the general meeting, or in an instrument of proxy sent out by the Company, direct that the instrument appointing a proxy may be deposited (no later than the time for holding the meeting or adjourned meeting) at the Registered Office or at such other place as is specified for that purpose in the notice convening the meeting, or in any instrument of proxy sent out by the Company. The chairperson of the meeting may in any event at his discretion direct that an instrument of proxy shall be deemed to have been duly deposited. An instrument of proxy that is not deposited in the manner permitted, or which has not been declared to have been duly deposited by the chairperson, shall be invalid. 94. An instrument of proxy shall: (a) be in any common form or in such other form as the Directors may approve; (b) be deemed to confer authority to vote on any amendment of a resolution put to the general meeting for which it is given as the proxy thinks fit; and (c) subject to its terms, be valid for any adjournment of the general meeting for which it is given. 95. The Directors may at the expense of the Company send to the Shareholders instruments of proxy (with or without prepaid postage for their return) for use at any general meeting, either in blank or nominating in the alternative any one or more of the Directors or any other persons. If for the purpose of any general meeting invitations to appoint as proxy a person or one of a number of persons specified in the invitations are issued at the expense of the Company, such invitations shall be issued to all (and not to some only) of the Shareholders entitled to be sent a notice of the meeting and to vote thereat by proxy. 96. A vote given in accordance with the terms of an instrument of proxy shall be valid notwithstanding the death or insanity of the principal or the revocation of the instrument of proxy, or of the authority under which the instrument of proxy was executed, or the transfer of the share in respect of which the proxy is given provided that no intimation in writing of such death, insanity, revocation or transfer shall have been received by the Company at the Registered Office before commencement of the meeting or adjourned meeting at which the instrument of proxy is used. 97. Anything which under these Articles a Shareholder may do by proxy that Shareholder may also do by a duly appointed attorney. The provisions of these Articles relating to proxies and instruments appointing proxies apply, mutatis mutandis, to any such attorney and the instrument appointing that attorney. CORPORATIONS ACTING BY REPRESENTATIVES AT MEETINGS 98. Any Shareholder which is a corporation or other non-natural person may, in accordance with its constitutional documents, or in the absence of such provision by a resolution of its directors or other governing body, authorise such person as it thinks fit to act as its representative at any meeting or meetings of the Company. The person so authorised shall be entitled to exercise the same powers on behalf of such corporation or other non-natural person as the corporation or other non-natural person could exercise if it were a Shareholder who was an individual and such corporation or other non-natural person shall for the purposes of these Articles be deemed to be present in person at any such meeting if a person so authorised is present. CLEARING HOUSES 99. If a recognised clearing house (or its nominee(s)) or depositary (or its nominee(s)) is a Shareholder of the Company it may authorise such person(s) as it thinks fit to act as its representative(s) at any general meeting of the Company or of any class of Shareholders of the Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

23 Company; provided, that, if more than one person is so authorised, the authorisation shall specify the number and class of shares in respect of which each such person is so authorised. Each person so authorised pursuant to this Article shall be deemed to have been duly authorised without further evidence of the facts and shall be entitled to exercise the same powers on behalf of the recognised clearing house (or its nominee(s)) or depositary (or its nominee(s)) which he represents as that recognised clearing house (or its nominee(s)) or depositary (or its nominee(s)) could exercise if it were an individual Shareholder holding the number and class of shares specified in such authorisation. WRITTEN RESOLUTIONS OF SHAREHOLDERS 100. A resolution (including a Special Resolution) in writing signed by all the Shareholders for the time being entitled to receive notice of, attend and vote at a general meeting (or, being entities, signed by their duly authorised representatives) shall be as valid and effective as a resolution passed at a general meeting duly convened and held and may consist of several documents in the like form each signed by one or more of the Shareholders. DIRECTORS 101. (a) Unless otherwise determined by the Company by Ordinary Resolution, the number of Directors shall not be less than two (2) Directors and the exact number of Directors shall be determined from time to time by the Board of Directors. (b) The Chairperson shall be Danny Yeung, as long as Danny Yeung is a Director. In the event that Danny Yeung is not a Director, the Board of Directors shall elect and appoint a Chairperson by the affirmative vote of a simple majority of the Directors then in office, and the period for which the Chairperson will hold office will also be determined by the affirmative vote of a simple majority of the Directors then in office. The Chairperson shall preside as chairperson at every meeting of the Board of Directors. To the extent the Chairperson is not present at a meeting of the Board of Directors within fifteen minutes after the time appointed for holding the same, the attending Directors may choose one of their member to be the chairperson of that meeting. (c) An appointment of a Director may be on terms that the Director shall automatically retire from office (unless he has sooner vacated office) at the next or a subsequent annual general meeting or upon any specified event or after any specified period in a written agreement between the Company and the Director, if any; but no such term shall be implied in the absence of express provision. Each Director whose term of office expires shall be eligible for re-election at a general meeting of the Company or re- appointment by the Board of Directors. 102. A Director shall not be required to hold any Shares in the Company by way of qualification. A Director who is not a Shareholder of the Company shall nevertheless be entitled to receive notice of and to attend and speak at general meetings of the Company and all classes of Shares of the Company. 103. The Company may, by Ordinary Resolution, appoint any person to be a Director and may in like manner remove any Director and may appoint another person in the Director's stead. Without prejudice to the power of the Company by Ordinary Resolution to appoint a person to be a Director, the Board of Directors, so long as a quorum of Directors remains in office, shall have the power at any time and from time to time to appoint any person to be a Director so as to fill a casual vacancy or as an addition to the existing Board of Directors or otherwise. 104. Each Director shall be entitled to such remuneration as approved by the Board of Directors or by Ordinary Resolution and this may be in addition to such remuneration as may be payable under any other Article. Such remuneration shall be deemed to accrue from day to day. The Directors and the Secretary may also be paid all travelling, hotel and other expenses properly incurred by them in attending and returning from meetings of the Directors or any committee Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

24 of the Directors or general meetings or in connection with the business of the Company. The Directors may, in addition to such remuneration as aforesaid, grant special remuneration to any Director who, being called upon, shall perform any special or extra services to or at the request of the Company. 105. Each Director shall have the power to nominate in writing another Director or any other person to act as alternate Director in the Director's place at any meeting of the Directors at which the Director is unable to be present and at the Director's discretion to remove such alternate Director. On such appointment being made the alternate Director shall (except as regards the power to appoint an alternate Director or as provided otherwise in the form of appointment) be subject in all respects to the terms and conditions existing with reference to the other Directors and each alternate Director, whilst acting in the place of an absent Director, shall exercise and discharge all the functions, powers and duties of the Director being represented. Any Director who is appointed as alternate Director shall be entitled at a meeting of the Directors to cast a vote on behalf of their appointor in addition to the vote to which such Director is entitled in his or her own capacity as a Director, and shall also be considered as two Directors for the purpose of making a quorum of Directors. Any person appointed as an alternate Director shall automatically vacate such office as an alternate Director if and when the Director by whom the alternate Director has been appointed vacates his or her office of Director. The remuneration of an alternate Director shall be payable out of the remuneration of the Director appointing such alternate Director and shall be agreed between them. 106. Every instrument appointing an alternate Director shall be in any usual or common form or such other form as the Directors may approve. 107. The appointment and removal of an alternate Director shall take effect when lodged at the Registered Office or delivered at a meeting of the Directors. 108. Any Director may appoint any individual, whether or not a Director, to be the proxy of that Director to attend and vote on his behalf, in accordance with instructions given by that Director, or in the absence of such instructions at the discretion of the proxy, at a meeting or meetings of the Directors which that Director is unable to attend personally. The instrument appointing the proxy shall be in writing under the hand of the appointing Director and shall be in any usual or common form or such other form as the Directors may approve, and must be lodged with the chairperson of the meeting at which such proxy is to be used, or first used, prior to the commencement of the meeting. 109. The office of a Director shall be vacated in any of the following events namely: (a) if the Director resigns their office by notice in writing signed by such Director and left at the Registered Office; (b) if the Director becomes bankrupt or makes any arrangement or composition with such Director's creditors generally; (c) if the Director dies or is found to be or becomes of unsound mind; (d) if the Director ceases to be a Director by virtue of, or becomes prohibited from being a Director by reason of, an order made under any provisions of any law or enactment; (e) if the Director is removed from office by notice addressed to such Director at their last known address and signed by all of the co-Directors (not being less than two in number); or (f) if the Director is removed from office by Ordinary Resolution. 110. The Board of Directors may, from time to time, and except as required by applicable law or the Designated Stock Exchange Rules, adopt, institute, amend, modify or revoke the corporate Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

25 governance policies or initiatives, which shall be intended to set forth the policies of the Company and the Board of Directors on various corporate governance related matters as the Board of Directors shall determine by resolution of Directors from time to time. TRANSACTIONS WITH DIRECTORS 111. A Director or alternate Director may hold any other office or place of profit under the Company (other than the office of Auditor) in conjunction with his office of Director on such terms as to tenure of office and otherwise as the Directors may determine. A Director or alternate Director may act by himself or by, through or on behalf of his firm in a professional capacity for the Company and he or his firm shall be entitled to remuneration for professional services as if he were not a Director or alternate Director; provided, that nothing herein contained shall authorise a Director or his firm to act as Auditor to the Company. 112. No Director or intending Director shall be disqualified by their office from contracting with the Company either as vendor, purchaser or otherwise, nor shall any such contract or any contract or arrangement entered into by or on behalf of the Company in which any Director is in any way interested be liable to be avoided, nor shall any Director so contracting or being so interested be liable to account to the Company for any profit realised by any such contract or arrangement by reason of such Director holding that office or of the fiduciary relationship thereby established, but the nature of the Director's interest must be declared by such Director at the meeting of the Directors at which the question of entering into the contract or arrangement is first taken into consideration, or if the Director was not at the date of that meeting interested in the proposed contract or arrangement, then at the next meeting of the Directors held after such Director becomes so interested, and in a case where the Director becomes interested in a contract or arrangement after it is made, then at the first meeting of the Directors held after such Director becomes so interested. 113. In the absence of some other material interest than is indicated below, provided a Director who is in any way, whether directly or indirectly, interested in a contract or proposed contract with the Company declares (whether by specific or general notice) the nature of their interest at a meeting of the Directors that Director may vote in respect of any contract or proposed contract or arrangement notwithstanding that such Director may be interested therein and if such Director does so his vote shall be counted and such Director may be counted in the quorum at any meeting of the Directors at which any such contract or proposed contract or arrangement shall come before the meeting for consideration. A general notice that a Director or alternate Director is a shareholder, director, officer or employee of any specified firm or company and is to be regarded as interested in any transaction with such firm or company shall be sufficient disclosure for the purposes of voting on a resolution in respect of a contract or transaction in which he has an interest and he may be counted in the quorum, and after such general notice it shall not be necessary to give special notice relating to any particular transaction. 114. Where proposals are under consideration concerning the appointment (including fixing or varying the terms of appointment) of two or more Directors to offices or employments with the Company or any company in which the Company is interested, such proposals may be divided and considered in relation to each Director separately and in such cases each of the Directors concerned shall be entitled to vote (and be counted in the quorum) in respect of each resolution except that concerning the Director's own appointment. 115. Any Director may continue to be or become a director, managing director, manager or other officer or shareholder of any company promoted by the Company or in which the Company may be interested, and no such Director shall be accountable for any remuneration or other benefits received by the Director as a director, managing director, manager or other officer or shareholder of any such other company. The Directors may exercise the voting power conferred by the shares in any other company held or owned by the Company or exercisable by them as directors of such other company, in such manner in all respects as they think fit (including the exercise thereof in favour of any resolution appointing themselves or any of them directors, Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

26 managing directors or other officers of such company, or voting or providing for the payment of remuneration to the directors, managing directors or other officers of such company). POWERS AND DUTIES OF DIRECTORS 116. The business of the Company shall be managed by the Directors, who may exercise all such powers of the Company as are not by the Companies Act or by these Articles required to be exercised by the Company in general meeting, subject nevertheless to any regulations of these Articles, to the Companies Act, and to such regulations being not inconsistent with the aforesaid regulations or provisions as may be prescribed by the Company in general meeting, but no regulations made by the Company in general meeting shall invalidate any prior act of the Directors which would have been valid if such regulations had not been made. The general powers given by this Article shall not be limited or restricted by any special authority or power given to the Directors by any other Article. 117. Subject to these Articles, the Directors may from time to time appoint any individual, whether or not a Director of the Company, to hold such office in the Company as the Directors may think necessary for the administration of the Company, including without prejudice to the foregoing generality, the office of the Chief Executive Officer, President, Chief Operating Officer, Chief Technology Officer, Chief Financial Officer, one or more Vice Presidents, Managers or Controllers, and for such term and at such remuneration (whether by way of salary or commission or participation in profits or partly in one way and partly in another), and with such powers and duties as the Directors may think fit. Unless otherwise specified in the terms of the officer's appointment an officer may be removed by resolution of the Directors or by Ordinary Resolution of the Shareholders. The Directors may also appoint one or more of their body (but not an alternate Director) to the office of managing director upon like terms, but any such appointment shall ipso facto determine if any managing director ceases from any cause to be a Director, or if the Shareholders by Ordinary Resolution resolves that his tenure of office be terminated. 118. The Directors may from time to time and at any time by power of attorney or otherwise appoint any person, whether nominated directly or indirectly by the Directors, to be the attorney or attorneys or authorised signatory of the Company for such purposes and with such powers authorities and discretions (not exceeding those vested in or exercisable by the Directors under these Articles) and for such period and subject to such conditions as they may think fit, and any such appointment may contain such provisions for the protection and convenience of persons dealing with any such attorneys or authorised signatory as the Directors may think fit, and may also authorise any such attorney or authorised signatory to sub-delegate all or any of the powers, authorities and discretions vested in such attorney or authorised signatory. The Directors may also appoint any person to be the agent of the Company for such purposes and with such powers, authorities and discretions (not exceeding those vested in or exercisable by the Directors under these Articles) and for such period and on such conditions as they determine, including authority for the agent to delegate all or any of their powers. 119. The Directors may from time to time provide for the management of the affairs of the Company in such manner as they shall think fit and the provisions contained in the following paragraphs shall be without prejudice to the general powers conferred by this paragraph. 120. All cheques, promissory notes, drafts, bills of exchange and other negotiable or transferable instruments drawn by the Company, and all receipts for monies paid to the Company shall be signed, drawn, accepted, endorsed or otherwise executed, as the case may be, in such manner as the Directors shall from time to time by resolution determine. 121. The Directors from time to time and at any time may establish any committees, local boards or agencies for managing any of the affairs of the Company and may appoint any persons to be members of such committees, local boards or agencies, and may appoint any managers or agents of the Company and may fix the remuneration of any of the aforesaid. PROCEEDINGS OF DIRECTORS Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

27 122. The Directors may meet together (whether within or outside the Cayman Islands) for the dispatch of business, adjourn and otherwise regulate their meetings, as they think fit. Questions and matters arising at any meeting shall be determined by a majority of votes. Each Director present in person or represented by his proxy or alternate shall be entitled to one (1) vote in deciding matters deliberated at any meeting of the Directors. Before the Acquisition Effective Time, in the case of an equality of votes, the Chairperson shall not have a second or casting vote and the resolution shall fail. From and after the Acquisition Effective Time, in the case of an equality of votes, the Chairperson shall have a second or casting vote. A Director may, and the Secretary on the requisition of a Director shall, at any time summon a meeting of the Directors. 123. At least three (3) Business Days’ notice in writing shall be given to all Directors and their respective alternates (if any) for a Board of Directors meeting which notice shall specify a date, time and agenda for such meeting; provided, that such notice period may be reduced or waived with the consent of all the Directors or their respective alternates (if any) either at, before or after the meeting is held; provided, further, that such notice period may, in the event of an emergency as determined by a majority of all Directors, be shortened to such notice period as the Chairperson may determine to be appropriate. The applicable notice period under this Article 123 for the applicable meeting of the Board of Directors shall be referred to as the Notice Period. 124. An agenda identifying in reasonable detail the issues to be considered by the Directors at any such meeting and copies (in printed or electronic form) of any relevant papers to be discussed at the meeting together with all relevant information shall be provided to and received by all Directors and their alternates (if any) within the Notice Period. The agenda for each meeting shall include any matter submitted to the Company by any Director within the Notice Period. 125. Unless approved by all Directors (whether or not present or represented at such meeting), matters not set out in the agenda need not be considered at a Board of Directors meeting. 126. A Director or Directors may participate in any meeting of the Board of Directors, or of any committee appointed by the Board of Directors of which such Director or Directors are members, by means of Communication Facilities and such participation shall be deemed to constitute presence in person at the meeting. 127. The quorum necessary for the transaction of the business of the Directors may be fixed by the Directors and unless so fixed shall be a majority of the Directors then in office, including the Chairperson; provided, however, a quorum shall nevertheless exist at a meeting at which a quorum would exist but for the fact that the Chairperson is voluntarily absent from the meeting and notifies the Board of Directors his decision to be absent from that meeting, before or at the meeting; provided, further, that a Director and his appointed alternate Director shall be considered only one person for this purpose. A Director represented by proxy or by an alternate Director at any meeting shall be deemed to be present for the purposes of determining whether or not a quorum is present. If a quorum is not present at a Board of Directors meeting within thirty (30) minutes following the time appointed for such Board of Directors meeting, the relevant meeting shall be adjourned for a period of at least three (3) Business Days and the presence of any two (2) Directors shall constitute a quorum at such adjourned meeting. A meeting of the Directors at which a quorum is present when the meeting proceeds to business shall be competent to exercise all powers and discretions for the time being exercisable by the Directors. 128. The continuing Directors or sole continuing Director may act notwithstanding any vacancies in their body, but if and so long as their number is reduced below the minimum number fixed by or pursuant to these Articles, then the continuing Directors or Director may act only to summon a general meeting of the Company, but for no other purpose. 129. Without prejudice to the powers conferred by these Articles, the Directors from time to time and at any time may establish and delegate any of their powers to committees consisting of such Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

28 member or members of their body as they think fit, and may authorise the members for the time being of any such committee, or any of them, to fill up any vacancies therein and to act notwithstanding vacancies.. Any committee so formed shall, in the exercise of the powers so delegated, conform to any regulations that may be imposed on them by the Directors. The Directors may adopt formal written charters for committees. 130. The meetings and proceedings of any such committee consisting of two or more Directors shall be governed by the provisions of these Articles regulating the meetings and proceedings of the Directors so far as the same are applicable and are not superseded by any regulations made by the Directors under the preceding Article. 131. A committee appointed by the Directors may elect a chairperson of its meetings. If no such chairperson is elected, or if at any meeting the chairperson is not present within fifteen minutes after the time appointed for holding the same, the members present may choose one of their number to be chairperson of the meeting. A committee appointed by the Directors may meet and adjourn as it thinks proper. Questions arising at any meeting shall be determined by a majority of votes of the committee members present and in case of an equality of votes the chairperson of the committee shall have a second or casting vote. 132. All acts done by any meeting of the Directors or of a committee of Directors, or by any individual acting as a Director, shall notwithstanding that it be afterwards discovered that there was some defect in the appointment of any such Director or individual acting as aforesaid, or that they or any of them were disqualified, or had vacated office, or were not entitled to vote, be as valid as if every such individual had been duly appointed and was qualified and had continued to be a Director and had been entitled to vote. 133. The Directors shall cause minutes to be made of: (a) all appointments of officers made by the Directors; (b) the names of the Directors present at each meeting of the Directors and of any committee of Directors; and (c) all resolutions and proceedings of all meetings of the Company and of the Directors and of any committee of Directors. Any such minutes, if purporting to be signed by the chairperson of the meeting at which the proceedings took place, or by the chairperson of the next succeeding meeting, shall, until the contrary be proved, be conclusive evidence of the proceedings. WRITTEN RESOLUTIONS OF DIRECTORS 134. A resolution in writing signed by all the Directors for the time being or all the members of a committee of Directors entitled to receive notice of a meeting of the Board of Directors or committee of Directors, as the case may be (an alternate Director, subject as provided otherwise in the terms of appointment of the alternate Director, being entitled to sign such a resolution on behalf of his appointer), shall be as valid and effective as a resolution passed at a duly convened meeting of the Directors or committee of Directors, as the case may be. When signed a resolution may consist of several documents in the like form each signed by one or more of the Directors (or their alternates). PRESUMPTION OF ASSENT 135. A Director who is present at a meeting of the Board of Directors or of a committee of Directors at which action on any Company matter is taken shall be presumed to have assented to the action taken unless the Director's dissent shall be entered in the minutes of the meeting or unless the Director shall file their written dissent from such action with the person acting as the chairperson or secretary of the meeting before the adjournment thereof or shall forward such Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

29 dissent by registered mail to such person immediately after the adjournment of the meeting. Such right to dissent shall not apply to a Director who voted in favour of such action. BORROWING POWERS 136. The Directors may exercise all the powers of the Company to borrow money and hypothecate, mortgage, charge or pledge its undertaking, property, and assets or any part thereof, and to issue debentures, debenture stock or other securities, whether outright or as collateral security for any debt liability or obligation of the Company or of any third party. SECRETARY 137. The Directors may appoint any person to be a Secretary who shall hold office for such term, at such remuneration and upon such conditions and with such powers as they think fit. Any Secretary so appointed by the Directors may be removed by the Directors or by the Company by Ordinary Resolution. Anything required or authorised to be done by or to the Secretary may, if the office is vacant or there is for any other reason no Secretary capable of acting, be done by or to any assistant or deputy Secretary or if there is no assistant or deputy Secretary capable of acting, by or to any officer of the Company authorised generally or specially in that behalf by the Directors, provided that any provisions of these Articles requiring or authorising a thing to be done by or to a Director and the Secretary shall not be satisfied by its being done by or to the same person acting both as Director and as, or in the place of, the Secretary. 138. No person shall be appointed or hold office as Secretary who is: (a) the sole Director; (b) a corporation the sole director of which is the sole Director; or (c) the sole director of a corporation which is the sole Director. THE SEAL 139. The Company may, if the Directors so determine, have a Seal. The Directors shall provide for the safe custody of the Seal and the Seal shall never be used except by the authority of a resolution of the Directors or of a committee of the Directors authorised by the Directors in that behalf. The Directors may keep for use outside the Cayman Islands a duplicate Seal. The Directors may from time to time as they see fit (subject to the provisions of these Articles relating to share certificates) determine the persons and the number of such persons in whose presence the Seal or the facsimile thereof shall be used, and until otherwise so determined the Seal or the duplicate thereof shall be affixed in the presence of any one Director or the Secretary, or of some other person duly authorised by the Directors. DIVIDENDS, DISTRIBUTIONS AND RESERVES 140. Subject to the Companies Act, these Articles, and any rights and restrictions for the time being attached to any class or classes of Shares, the Directors may, in their absolute discretion, declare dividends and distributions on Shares in issue and authorise payment of the dividends or distributions out of the funds of the Company lawfully available therefor. No dividend or distribution shall be paid except out of the realised or unrealised profits of the Company, or out of the Share Premium Account, or as otherwise permitted by the Companies Act. Subject to any rights and restrictions for the time being attached to any class or classes of Shares and these Articles, the Company by Ordinary Resolution may declare dividends out of the funds of the Company lawfully available therefor, but no dividend shall exceed the amount recommended by the Directors. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

30 141. The Directors may, before recommending or declaring any dividend, set aside out of the funds legally available for distribution such sums as they think proper as a reserve or reserves which shall, at the discretion of the Directors, be applicable for meeting contingencies, or for equalising dividends or for any other purpose to which those funds may be properly applied and pending such application may, at the like discretion, either be employed in the business of the Company or be invested in such investments (other than Shares of the Company) as the Directors may from time to time think fit. 142. Except as otherwise provided by the rights attached to Shares, or as otherwise determined by the Directors, all dividends shall be declared and paid according to the amounts paid or credited as fully paid on the Shares, but if and so long as nothing is paid up on any of the Shares in the Company dividends may be declared and paid according to the par value of the Shares. No amount paid on a Share in advance of calls shall, while carrying interest, be treated for the purposes of this Article as paid on the Share. If any Share is issued on terms providing that it shall rank for dividend or distribution as from a particular date, that Share shall rank for dividend or distribution accordingly. 143. The Directors may deduct and withhold from any dividend or distribution otherwise payable to any Shareholder all sums of money (if any) then payable by the Shareholder to the Company on account of calls or otherwise or any monies which the Company is obliged by law to pay to any taxing or other authority. 144. The Directors may declare that any dividend or distribution be paid wholly or partly by the distribution of specific assets and in particular of shares, debentures or securities of any other company or in any one or more of such ways and, where any difficulty arises in regard to such distribution, the Directors may settle the same as they think expedient and in particular may issue fractional Shares and fix the value for distribution of such specific assets or any part thereof and may determine that cash payments shall be made to any Shareholder upon the basis of the value so fixed in order to adjust the rights of all Shareholders and may vest any such specific assets in trustees as may seem expedient to the Directors. No dividend shall be made in specie on any Class A Ordinary Shares unless a dividend in specie in equal proportion is made on the Class B Ordinary Shares. 145. Any dividend, distribution, interest or other monies payable in cash in respect of Shares may be paid in any manner determined by the Directors. If paid by cheque it will be sent by mail addressed to the holder at his address in the Register of Members, or addressed to such person and at such address as the holder or joint holders may in writing direct. Every such cheque shall (unless the Directors in their sole discretion otherwise determine) be made payable to the order of the person to whom it is sent or to the order of such other person as the Shareholder entitled, or such joint holders as the case may be, may direct. Any one of two or more joint holders may give effectual receipts for any dividends, bonuses, or other monies payable in respect of the Share held by them as joint holders. 146. Any dividend or distribution which cannot be paid to a Shareholder and/or which remains unclaimed after six (6) months from the date of declaration of such dividend or distribution may, in the discretion of the Directors, be paid into a separate account in the Company's name, provided that the Company shall not be constituted as a trustee in respect of that account and the dividend or distribution shall remain as a debt due to the Shareholder. Any dividend or distribution which remains unclaimed after a period of six years from the date on which such dividend or other distribution becomes payable shall be forfeited and shall revert to the Company. 147. No dividend or distribution shall bear interest against the Company. SHARE PREMIUM ACCOUNT Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

31 148. The Directors shall establish an account on the books and records of the Company to be called the Share Premium Account and shall carry to the credit of such account from time to time a sum equal to the amount or value of the premium paid on the issue of any Share. ACCOUNTS 149. The Directors shall cause proper books of account (including, where applicable, material underlying documentation including contracts and invoices) to be kept with respect to all sums of money received and expended by the Company and the matters in respect of which the receipt or expenditure takes place, all sales and purchases of goods by the Company and the assets and liabilities of the Company. Such books of account must be retained for a minimum period of five years from the date on which they are prepared. Proper books shall not be deemed to be kept if there are not kept such books of account as are necessary to give a true and fair view of the state of the Company's affairs and to explain its transactions. 150. The books of account shall be kept at the Registered Office or at such other place as the Directors think fit, and shall always be open to inspection by the Directors. 151. The Board of Directors shall from time to time determine whether and to what extent and at what time and places and under what conditions or articles the accounts and books of the Company or any of them shall be open to the inspection of Shareholders not being Directors, and no Shareholder (not being a Director) shall have any right of inspection of any account or book or document of the Company except as conferred by law or authorised by the Board of Directors or by Ordinary Resolution of the Shareholders. 152. The Directors may cause to be prepared and to be laid before the Company in general meeting profit and loss accounts, balance sheets, group accounts (if any) and such other reports and accounts as may be required by law and the listing rules of the Designated Stock Exchange. 153. Subject to the requirements of applicable law and the listing rules of the Designated Stock Exchange, the accounts relating to the Company’s affairs shall be audited with such financial year end as set forth in Article 180 and in such manner as may be determined from time to time by the Company by Ordinary Resolution or failing any such determination by the Directors or failing any determination as aforesaid shall not be audited. AUDIT 154. The Directors may appoint an auditor of the Company who shall hold office until removed from office by a resolution of the Directors and may fix his or their remuneration. 155. Every Auditor of the Company shall have a right of access at all times to the books and accounts and vouchers of the Company and shall be entitled to require from the Directors and officers of the Company such information and explanation as may be necessary for the performance of the duties of auditors. 156. Auditors shall, if so required by the Directors, make a report on the accounts of the Company during their tenure of office at the next general meeting following their appointment, and at any time during their term of office, upon request of the Directors at any general meeting of the Company. 157. The accounts relating to the Company's affairs shall be audited in such manner as may be determined from time to time by Ordinary Resolution of the Shareholders or failing any such Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

32 determination, by the Board of Directors, or failing any determination as aforesaid, shall not be audited. CAPITALISATION OF PROFITS 158. Subject to the Companies Act, these Articles and any rights or restrictions for the time being attached to any class or classes of Shares, the Board of Directors may: (a) resolve to capitalise any amount standing to the credit of reserves (including a share premium account, capital redemption reserve and profit and loss account) or otherwise available for distribution; (b) appropriate the sum resolved to be capitalised to the Shareholders in the proportions in which such sum would have been divisible amongst such Shareholders had the same been a distribution of profits by way of dividend or other distribution, and apply that sum on their behalf in or towards: (i) paying up the amounts (if any) for the time being unpaid on Shares held by them respectively; or (ii) paying up in full unissued Shares or debentures of a nominal amount equal to that sum, and allot the Shares or debentures, credited as fully paid, to the Shareholders (or as they may direct) in those proportions, or partly in one way and partly in the other, but the share premium account, the capital redemption reserve and profits which are not available for distribution may, for the purposes of this Article, only be applied in paying up unissued Shares to be allotted to Shareholders credited as fully paid; (c) make any arrangements it thinks fit to resolve a difficulty arising in the distribution of a capitalised reserve and in particular, without limitation, where Shares or debentures become distributable in fractions the Board of Directors may deal with the fractions as it thinks fit (including provisions whereby the benefit of fractional entitlements accrue to the Company rather than to the Shareholders concerned); (d) authorise a person to enter (on behalf of all the Shareholders concerned) an agreement with the Company providing for either: (i) the allotment to the Shareholders respectively, credited as fully paid, of Shares or debentures to which they may be entitled on the capitalisation, or (ii) the payment by the Company on behalf of the Shareholders (by the application of their respective operations of the reserves resolved to be capitalised) of the amounts or part of the amounts remaining unpaid on their existing Shares, an agreement made under the authority being effective and binding on all those Shareholders; and (e) generally do all acts and things required to give effect to the resolution. NOTICES AND INFORMATION 159. Any notice or document may be served by the Company on any Shareholder: (a) personally; Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

33 (b) by registered post or courier to that Shareholder's address as appearing in the Register of Members; or (c) by cable, telex, facsimile, e-mail or any other electronic means should the Directors deem it appropriate. 160. In the case of joint holders of a Share, all notices shall be given to that one of the joint holders whose name stands first in the Register of Members in respect of the joint holding, and notice so given shall be sufficient notice to all the joint holders. 161. Notices posted to addresses outside the Cayman Islands shall be forwarded by prepaid airmail or a recognized courier service. 162. Any Shareholder present, either personally or by proxy, at any meeting of the Company shall for all purposes be deemed to have received due notice of such meeting and, where requisite, of the purposes for which such meeting was convened. 163. Any summons, notice, order or other document required to be sent to or served upon the Company, or upon any Director or officer of the Company may be sent or served by leaving the same or sending it through the post in a prepaid letter envelope or wrapper, addressed to the Company or to such Director or officer at the Registered Office. 164. Any notice or other document, if served by: (a) registered post, shall be deemed to have been served five calendar days after the time when the letter containing the same is posted; (b) facsimile, shall be deemed to have been served upon production by the transmitting facsimile machine of a report confirming transmission of the facsimile in full to the facsimile number of the recipient ; (c) recognised courier service, shall be deemed to have been served 48 hours after the time when the letter containing the same is delivered to the courier service; or (d) electronic means, shall be deemed to have been served immediately (i) upon the time of the transmission to the electronic mail address supplied by the Shareholder to the Company or (ii) upon the time of its placement on the Company's website. In proving service by post or courier service it shall be sufficient to prove that the letter containing the notice or documents was properly addressed and duly posted or delivered to the courier service. 165. Any notice or document delivered or sent by post to or left at the registered address of any Shareholder in accordance with these Articles shall notwithstanding that such Shareholder be then dead, bankrupt or dissolved, and whether or not the Company has notice of such death, bankruptcy or dissolution, be deemed to have been duly served in respect of any Share registered in the name of such Shareholder as sole or joint holder, unless the Shareholder's name shall at the time of the service of the notice or document, have been removed from the Register of Members as the holder of the Share, and such service shall for all purposes be deemed a sufficient service of such notice or document on all persons interested (whether jointly with or as claiming through or under such Shareholder) in the Share. 166. Notice of every general meeting shall be given to: (a) all Shareholders holding Shares with the right to receive notice and who have supplied to the Company an address for the giving of notices to them; Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

34 (b) every person entitled to a Share in consequence of the death or bankruptcy of a Shareholder, who but for his death or bankruptcy would be entitled to receive notice of the meeting; and (c) each Director and alternate Director. No other person shall be entitled to receive notices of general meetings. 167. No Shareholder shall be entitled to require discovery of any information in respect of any detail of the Company’s trading or any information which is or may be in the nature of a trade secret or secret process which may relate to the conduct of the business of the Company and which in the opinion of the Board of Directors would not be in the interests of the Shareholders of the Company to communicate to the public. 168. The Board of Directors shall be entitled to release or disclose any information in its possession, custody or control regarding the Company or its affairs to any of its members including, without limitation, information contained in the Register of Members and transfer books of the Company. WINDING UP AND FINAL DISTRIBUTION OF ASSETS 169. The Directors may present a winding up petition on behalf of the Company without the sanction of a resolution of the Shareholders passed at a general meeting. 170. If the Company shall be wound up the liquidator shall apply the assets of the Company in satisfaction of creditors' claims in such manner and order as such liquidator thinks fit. 171. If the Company shall be wound up, and the assets available for distribution amongst the Shareholders shall be insufficient to repay the whole of the share capital, such assets shall be distributed so that, as nearly as may be, the losses shall be borne by the Shareholders in proportion to the par value of the Shares held by them. If in a winding up the assets available for distribution amongst the Shareholders shall be more than sufficient to repay the whole of the share capital at the commencement of the winding up, the surplus shall be distributed amongst the Shareholders in proportion to the par value of the Shares held by them at the commencement of the winding up subject to a deduction from those Shares in respect of which there are monies due of all monies payable to the Company for unpaid calls or otherwise. This Article is without prejudice to the rights of the holders of Shares issued upon special terms and conditions. 172. If the Company shall be wound up (whether the liquidation is voluntary, under supervision or by the court) the liquidator may, subject to the rights attaching to any Shares and with the authority of a Special Resolution, divide among the Shareholders in specie the whole or any part of the assets of the Company, and whether or not the assets shall consist of property of a single kind, and may for such purposes set such value as the liquidator deems fair upon any one or more class or classes of property, and may determine how such division shall be carried out as between the Shareholders. The liquidator may, with the like authority, vest any part of the assets in trustees upon such trusts for the benefit of Shareholders as the liquidator, with the like authority, shall think fit, and the liquidation of the Company may be closed and the Company dissolved, but so that no Shareholder shall be compelled to accept any assets in respect of which there is liability. INDEMNITY 173. To the maximum extent permitted by applicable law, every Director (including for the purposes of this Article any alternate Director appointed pursuant to the provisions of these Articles) or officer of the Company together with every former Director and former officer of the Company and the personal representatives of the same (but not including the Company's auditors) (each an Indemnified Person) shall be indemnified out of the assets of the Company against any liability, action, proceeding, claim, demand, costs, damages or expenses, including legal expenses, whatsoever which they or any of them may incur as a result of any act or failure to Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

35 act in carrying out their functions other than such liability (if any) that they may incur by their own dishonesty, actual fraud or wilful default. No such Indemnified Person shall be liable to the Company for any loss or damage in carrying out their functions unless that liability arises through the dishonesty, actual fraud or wilful default of such Indemnified Person. No Indemnified Person shall be found to be dishonest or have committed actual fraud or wilful default under this Article unless or until a court of competent jurisdiction shall have made a finding to that effect, and such finding shall have become final and non-appealable. 174. The Directors on behalf of the Company, shall have the power to purchase and maintain insurance for the benefit of any person who is or was a Director or officer of the Company indemnifying them against any liability which may lawfully be insured against by the Company. DISCLOSURE 175. Any Director, officer or authorised agent of the Company shall, if lawfully required to do so under the laws of any jurisdiction to which the Company is subject or in compliance with the rules of any stock exchange upon which the Company’s shares are listed, be entitled to release or disclose to any regulatory or judicial authority, or to any stock exchange upon which the Company’s shares are listed, any information in their possession regarding the affairs of the Company including, without limitation, any information contained in the Register of Members. CLOSING REGISTER OF MEMBERS OR FIXING RECORD DATE 176. For the purpose of determining those Shareholders that are entitled to receive notice of, attend or vote at any meeting of Shareholders or any adjournment thereof, or those Shareholders that are entitled to receive payment of any dividend, or in order to make a determination as to who is a Shareholder for any other purpose, the Directors may provide that the Register of Members shall be closed for transfers for a stated period but not to exceed in any case 30 calendar days. If the Register of Members shall be so closed for the purpose of determining those Shareholders that are entitled to receive notice of, attend or vote at a meeting of Shareholders such register shall be so closed for at least 10 calendar days immediately preceding such meeting and the record date for such determination shall be the date of the closure of the Register of Members. 177. In lieu of or apart from closing the Register of Members, the Directors may fix in advance a date as the record date for any such determination of those Shareholders that are entitled to receive notice of, attend or vote at a meeting of the Shareholders and for the purpose of determining those Shareholders that are entitled to receive payment of any dividend, the Directors may, at or within 90 calendar days prior to the date of declaration of such dividend fix a subsequent date as the record date of such determination. 178. If the Register of Members is not so closed and no record date is fixed for the determination of those Shareholders entitled to receive notice of, attend or vote at a meeting of Shareholders or those Shareholders that are entitled to receive payment of a dividend, the date on which notice of the meeting is sent or the date on which the resolution of the Directors declaring such dividend is adopted, as the case may be, shall be the record date for such determination of Shareholders. When a determination of those Shareholders that are entitled to receive notice of, attend or vote at a meeting of Shareholders has been made as provided in this section, such determination shall apply to any adjournment thereof. REGISTRATION BY WAY OF CONTINUATION 179. The Company may by Special Resolution resolve to be registered by way of continuation in a jurisdiction outside the Cayman Islands or such other jurisdiction in which it is for the time being incorporated, registered or existing. The Directors may cause an application to be made to the Registrar of Companies to deregister the Company in the Cayman Islands or such other jurisdiction in which it is for the time being incorporated, registered or existing and may cause all such further steps as they consider appropriate to be taken to effect the transfer by way of continuation of the Company. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682

36 FINANCIAL YEAR 180. The Directors shall determine the financial year of the Company and may change the same from time to time. Unless they determine otherwise, the financial year of the Company shall end on December 31st of each year. AMENDMENTS TO MEMORANDUM AND ARTICLES OF ASSOCIATION 181. Subject to Article 21(c)(iv)(5), the Company may from time to time alter or add to these Articles or alter or add to the Memorandum with respect to any objects, powers or other matters specified therein or change the name of the Company by passing a Special Resolution in the manner prescribed by the Companies Act. MERGERS AND CONSOLIDATION 182. The Company shall have the power to merge or consolidate with one or more other constituent companies (as defined in the Companies Act) upon such terms as the Directors may determine and (to the extent required by the Companies Act) with the approval of a Special Resolution. Filed: 31-May-2022 16:13 EST Auth Code: J29211648896www.verify.gov.ky File#: 378682
Exhibit 2.11
DESCRIPTION OF THE REGISTRANT’S SECURITIES
REGISTERED UNDER SECTION 12 OF THE SECURITIES EXCHANGE ACT OF 1934
As of December 31, 2025, Prenetics Global Limited (the “Company”, “we”, “us” and “our”) had the following series of securities registered pursuant to Section 12(b) of the Exchange Act:
| Title of each class | Trading symbol(s) | Name of each exchange on which registered | ||||||||||||
Class A Ordinary Shares, par value $0.0015 per share | PRE | The Nasdaq Stock Market LLC | ||||||||||||
| Warrants | PRENW | The Nasdaq Stock Market LLC | ||||||||||||
Description of Ordinary Shares
For a summary of certain material provisions of our Amended and Restated Memorandum and Articles of Association, as amended by special resolution on August 1, 2025 (the “Memorandum and Articles of Association”), as well as the Companies Act (As Revised) of the Cayman Islands (the “Companies Act”) insofar as they relate to the material terms of our ordinary shares, refer to "Item 10. Additional Information—B. Memorandum and Articles of Association" in our annual report on Form 20-F for the fiscal year ended December 31, 2025, filed with the SEC on April 30, 2026 ("2025 Form 20-F"). Notwithstanding this, because it is a summary, it may not contain all the information that you may otherwise deem important. For more complete information, you should read the entire Memorandum and Articles of Association, which has been filed with the SEC as exhibit 1.1 to our 2025 Form 20-F.
Debt Securities
Not applicable.
Warrants and Rights
The number of Warrants that were outstanding as of December 31, 2025 is provided on the cover of our 2025 Form 20-F. Our Warrants are issued in registered (book-entry) form under the warrant agreement, dated May 13, 2021, by and between Artisan Acquisition Corp. (“Artisan”) and Continental Stock Transfer & Trust Company (“Continental”), as warrant agent, as amended by the Assignment, Assumption and Amendment Agreement, dated September 15, 2021, by and among Artisan, the Company and Continental (the warrant agreement, as amended, the “Warrant Agreement”).
In connection with and upon the consummation of the Business Combination, each Artisan Warrant outstanding immediately prior to the Business Combination was assumed by the Company and converted into a Warrant entitling the holder thereof to purchase such number of Class A Ordinary Shares equal to the Class A Exchange Ratio upon exercise. Each Warrant continues to have and be subject to substantially the same terms and conditions as were applicable to such Artisan Warrant immediately prior to the consummation of the Business Combination (including any repurchase rights and cashless exercise provisions).
The following summary of certain provisions relating to our Warrants does not purport to be complete and is subject to, and is qualified in its entirety by reference to the Warrant Agreement.
General
1
Each Warrant entitles its holder to purchase 1.29 Class A Ordinary Share at an exercise price (the “Exercise Price”) of $133.65 per 1.29 shares (or an effective price of $103.60 per share), subject to adjustment pursuant to the terms of the Assignment, Assumption and Amendment Agreement and the warrant agreement, dated May 13, 2021, by and between Artisan and Continental. Our Warrants became exercisable on June 17, 2022. The Warrants will expire at the earliest to occur of (i) 5:00 p.m., New York City time on May 18, 2027 and (ii) 5:00 p.m., New York City time on the redemption date, if any, that we may fix in accordance with the Warrant Agreement, except that in the case of any private warrants held by the Sponsor or any of its Permitted Transferees, they will expire at 5:00 p.m., New York City time on May 18, 2027 (the expiration dates of the warrants, the “Expiration Date”). We may in our sole discretion extend the duration of the warrants so long as we provide at least twenty (20) days’ prior written notice to all registered holders. Any such extension must be identical among all of the warrants. Any warrant not exercised prior to its expiration will become void.
Exercise of Warrants
A Warrant may be exercised by delivering to the warrant agent (i) the Warrant, (ii) an election to purchase form, and (iii) the payment in full of the Exercise Price and any and all applicable taxes due in connection with the exercise.
As soon as practicable after the exercise of any Warrant we will issue a book-entry position or certificate, as applicable, for the Class A Ordinary Shares. All Class A Ordinary Shares issued upon the proper exercise of a Warrant in conformity with the Warrant Agreement will be validly issued, fully paid and non-assessable.
A Warrant holder may notify us in writing of the holder’s election to be subject to a provision of the Warrant Agreement preventing the holder from exercising a Warrant, to the extent that, after giving effect to such exercise, the holder (together with its affiliates), to the warrant agent’s actual knowledge, would beneficially own in excess of 9.8% (or such other amount as a holder may specify) (the “Maximum Percentage”) of our outstanding Class A Ordinary Shares immediately after giving effect to such exercise. By written notice to us, a Warrant holder may increase or decrease the Maximum Percentage to any other percentage specified in such notice; provided, however, that any such increase will not be effective until the sixty-first (61st) day after such notice is delivered to us.
Notwithstanding the above, we are not obligated to deliver any Class A Ordinary Shares pursuant to the exercise of a Warrant and do not have the obligation to settle such Warrant exercise unless a registration statement under the Securities Act with respect to the Class A Ordinary Shares underlying the Warrants is then effective and a prospectus relating thereto is current, subject to our satisfaction of obligations with respect to registration under the Warrant Agreement, or a valid exemption from registration is available. No Warrant will be exercisable and we will not be obligated to issue a Class A Ordinary Share upon exercise of a Warrant unless the Class A Ordinary Share issuable upon such Warrant exercise has been registered, qualified or deemed to be exempt under the securities laws of the state of residence of the registered holder of the Warrants.
Adjustments
We may, in our sole discretion, lower the Exercise Price at any time prior to the Expiration Date for a period of not less than fifteen business days, unless otherwise required by applicable laws, stock exchange rules or the U.S. Securities and Exchange Commission (the “SEC”), provided that we provide at least five days’ prior written notice to the holders and any such reduction shall be identical among all of the Warrants.
The number of Class A Ordinary Shares issuable upon the exercise of the Warrants is subject to customary adjustments in certain circumstances, such as a capitalization or share dividend of ordinary shares, or by a sub-division of ordinary shares or other similar event, as described in the Warrant Agreement. In the event the number of Class A Ordinary Shares purchasable upon the exercise of the
2
Warrants is adjusted, the Exercise Price will be adjusted (to the nearest cent) by multiplying the Exercise Price immediately prior to such adjustment, by a fraction (x) the numerator of which shall be the number of ordinary shares purchasable upon the exercise of the Warrants immediately prior to such adjustment, and (y) the denominator of which shall be the number of ordinary shares so purchasable immediately thereafter.
If, by reason of any adjustment made pursuant to the events described above, the holder of any Warrant would be entitled, upon the exercise of such Warrant, to receive a fractional interest in a share, we will, upon such exercise, round down to the nearest whole number the number of ordinary shares to be issued to such holder.
Warrant holders also have replacement rights in the case of certain reclassification, reorganization, merger, consolidation or sale transactions involving our company or substantially all of our assets (each a “Replacement Event”). Upon the occurrence of any Replacement Event, Warrant holders will have the right to purchase and receive (in lieu of our Class A Ordinary Shares) the kind and amount of stock or other securities or property (including cash) receivable upon such Replacement Event that the holder would have received if the Warrants were exercised immediately prior to such event.
Upon any adjustment of the Exercise Price or the number of shares issuable upon exercise of a Warrant, we will provide written notice of such adjustment to the warrant agent stating the Exercise Price resulting from such adjustment and the increase or decrease, if any, in the number of Class A Ordinary Shares purchasable at such price upon the exercise of a Warrant. We will also provide written notice of any adjustment described above to each Warrant holder at the last address set forth in the warrant register, of the record date or the effective date of the event.
Cashless Exercise
We agreed to use commercially reasonable efforts to file with the SEC as soon as practicable a registration statement for the registration, under the Securities Act, of the Class A Ordinary Shares issuable upon exercise of the Warrants. We are obligated to use commercially reasonable efforts to cause the registration statement to become effective and to maintain its effectiveness, and a current prospectus relating thereto, until the expiration or redemption of the Warrants. Warrant holders have the right, during any period that we may fail to, as agreed, maintain an effective registration statement covering the Class A Ordinary Shares issuable upon exercise of the Warrants, to exercise such Warrants on a “cashless basis.” In a cashless exercise, holders may exchange their Warrants for a number of Class A Ordinary Shares equal to the lesser of (A) the quotient obtained by dividing (x) the product of the number of Class A Ordinary Shares underlying the Warrants, multiplied by the excess of the Fair Market Value (as defined hereinafter) less the Exercise Price by (y) the Fair Market Value and (B) 0.361 per Warrant. “Fair Market Value” in this paragraph means the volume weighted average price of the Class A Ordinary Shares as reported during the ten trading days ending on the trading day prior to the date that notice of exercise is received by the warrant agent.
If, by reason of any exercise of Warrants on a “cashless basis”, the holder of any Warrant would be entitled, upon the exercise of such Warrant, to receive a fractional interest in a Class A Ordinary Share, we will round down to the nearest whole number, the number of Class A Ordinary Shares to be issued to such holder.
Redemption
We have the right to redeem all the Warrants (but not less than all the Warrants), at any time while they are exercisable and prior to their expiration, at the office of the warrant agent, upon notice to the registered holders of the Warrants, at the price of $0.01 per Warrant (the “Redemption Price”), if (i) the last reported sale price of our Class A Ordinary Shares has been at least $18.00 per share (subject to certain adjustments), for any twenty (20) trading days within the thirty (30) trading-day period ending on the third trading day prior to the date on which notice of the redemption is given and (ii) there is an effective
3
registration statement covering issuance of the Class A Ordinary Shares issuable upon exercise of the Warrants, and a current prospectus relating thereto, available throughout the 30 days prior to the redemption date.
In the event we choose to redeem all outstanding Warrants, we are required to (i) fix a date for the redemption and (ii) provide notice to the registered holders of the Warrants at least 30 days prior to the redemption date. We will mail any such notice of redemption by first class mail, postage prepaid, not less than 30 days prior to the redemption date to registered Warrant holders. The notice will be sent to each registered holder’s last address as it appears on the registration books. Any notice so mailed will be conclusively presumed to have been duly given, whether or not the registered holder actually receives such notice.
If there is no effective registration statement and current prospectus available, we may nonetheless redeem the Warrants on a “cashless basis,” provided that (i) the last reported sale price of our Class A Ordinary Shares has been at least $10.00 per share (subject to certain adjustments) the last reported sales price of the Class A Ordinary Shares for any twenty (20) trading days within the thirty (30) trading-day period ending on the third trading day prior to the date on which notice of the redemption is given; and (ii) the holders are able to exercise their Warrants on a cashless basis prior to redemption and receive that number of Class A Ordinary Shares determined based on the date of redemption and the volume weighted average price of the Class A Ordinary Shares during the ten trading days immediately following the date on which the notice of redemption is sent to holders of the Warrants.
On and after the redemption date, the record holder of the Warrants will have no further rights except to receive, upon surrender of the Warrants, the Redemption Price.
The redemption rights above do not apply to the private warrants unless and until they are transferred to persons other than the Sponsor and its Permitted Transferees.
Other Securities
Not applicable.
Description of American Depositary Shares
Not applicable.
4
Exhibit 4.27
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. Certain schedules have been excluded from this exhibit because they are both (i) not material and (ii) not otherwise disclosed in the exhibit or our annual report on Form 20-F for the fiscal year ended December 31, 2025. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
DELTA ELECTRONICS, INC. as the Buyer THE PERSONS LISTED IN PART A OF SCHEDULE 1 as the Sellers [***] as the Holders' Representative AND ACT GENOMICS HOLDINGS COMPANY LIMITED as the Company | ||||||||||||||
AGREEMENT FOR SALE AND PURCHASE OF THE ISSUED SHARES IN ACT GENOMICS HOLDINGS COMPANY LIMITED | ||||||||||||||
Contents | |||||
Clause | Page | ||||
- 1 - | ||||||||
- 2 - | ||||||||
THIS AGREEMENT is made on June 20, 2025
AMONG:
(1)DELTA ELECTRONICS, INC., a company incorporated under the laws of Taiwan, having its registered office at No.186 Ruey Kuang Road, Neihu District, Taipei 114501, Taiwan (the "Buyer");
(2)THE PERSONS whose names and addresses are set out in column (A) in Part A of Schedule 1 (the "Sellers" and each a "Seller");
(3)[***], an individual with [***] ID number [***] (the "Holders' Representative"); and
(4)ACT GENOMICS HOLDINGS COMPANY LIMITED, an exempted company incorporated under the laws of the Cayman Islands (registered number: 336039), having its registered office at PO Box 309, Ugland House, Grand Cayman, KY1-1104, Cayman Islands (the "Company").
THE PARTIES AGREE as follows:
1.Interpretation¶
1.1.In this Agreement:
"Accounting Policies" means the accounting policies and procedures set out in Schedule 5;
"Accounts" means the consolidated audited accounts of the Group, including the profit and loss statement, balance sheet, cashflow statement, the auditors' reports on those accounts, the directors' reports and the notes to those accounts, for the financial years ended on 31 December 2021, 31 December 2022 and the Last Accounting Date, prepared in accordance with the IFRS; in each case, provided by the Company to the Buyer prior to the date of this Agreement;
"Accounts Relief" means a Relief which has been shown or treated as an asset in the Accounts or Management Accounts, or which has been taken into account in computing a provision for Tax which appears in the Accounts or Management Accounts (or which has resulted in no such provision for Tax being made);
"ACT Singapore" means ACT Genomics (Singapore) Pte. Ltd., a limited liability company incorporated in Singapore with its registered office at 1 Phillip Street, #09-00, Royal One Phillip, Singapore 048692;
"ACT Taiwan" means ACT Genomics Co., Ltd. 行動基因生技股份有限公司, a company limited by shares incorporated under the laws of the R.O.C. with its registered address at 3F., No. 345, Xinhu 2nd. Rd., Neihu Dist., Taipei City 11494, Taiwan (R.O.C.);
"ACT Thailand" means ACT Genomics (Thailand) Limited., a company limited by shares incorporated under the laws of Thailand. with its registered address at No.4,
- 3 - | ||||||||
4/5 Unit 1503, Zen Tower, 15/F, Ratchadamri Rd, Pathum Wan, Bangkok 10330, Thailand;
"Action" means any civil charge, dispute, criminal or administrative action, claim, lawsuit, counter-suit, investigation, injunction, hearing, order, audit, settlement, arbitration, mediation, civil or criminal legal proceeding, or administrative enforcement proceeding, by or before any Government Authority, authorised arbitrator or mediation tribunal;
"Actual Cash" means the amount of Cash of the Group as at the Effective Time, as set out in the Completion Statement;
"Actual Indebtedness" means the amount of Indebtedness of the Group as at the Effective Time, as set out in the Completion Statement;
"Actual Net Debt" means the amount equal to the difference between the Actual Indebtedness and the Actual Cash (expressed as a positive number, if the Actual Indebtedness is greater than the Actual Cash; or expressed as a negative number, if the Actual Indebtedness is less than the Actual Cash), in each case, as set out in the Completion Statement;
"Actual Transaction Expenses" means the aggregate amount of all Transaction Expenses as at the Effective Time, as set out in the Completion Statement;
"Actual Working Capital Adjustment" means the amount equal to the difference between the Actual Working Capital Amount and the Target Working Capital Amount (expressed as a negative number, if the Actual Working Capital Amount is less than the Target Working Capital Amount; or expressed as a positive number, if the Actual Working Capital Amount is greater than the Target Working Capital Amount), in each case, as set out in the Completion Statement;
"Actual Working Capital Amount" means the amount of Working Capital of the Group as at the Effective Time, as set out in the Completion Statement;
"Adjustment Amount" has the meaning given in clause 4.5;
"Adjustment Escrow Account" means an account designated by the Escrow Agent, into which the Adjustment Escrow Amount shall be deposited pursuant to clause 6.2.2;
"Adjustment Escrow Amount" means an amount equal to US$[***];
"Adjustment Ratio" means a ratio (expressed as a percentage) equal to the Initial Consideration divided by the Headline Price;
"Affiliate" means:
- 4 - | ||||||||
(a) in respect of a person who is a natural person, his or her Immediate Family Members or any other person directly or indirectly Controlled by such natural person or his or her Immediate Family Members; and
(b) in respect of a person who is not a natural person, any other person which, directly or indirectly, Controls, is Controlled by or is under the common Control of the first mentioned person;
"Aged A/R" means any A/R over 180 days past due;
"Amended Articles" means the Memorandum and Articles of Association in substantial form as set out in Appendix II to this Agreement, to be adopted by the Company prior to, and to be effective as at, Completion;
"Applicable Laws" means, with respect to a person, any laws, regulations, rules, measures, guidelines, treaties, judgments, determination, orders or notices of any Government Authority or stock exchange that is applicable to such person;
"Audited Completion Accounts" has the meaning given in paragraph 1 of Schedule 3;
"A/P" means accounts payable of the Group at the relevant time;
"A/R" means accounts receivable of the Group at the relevant time;
"Business" means provision of cancer genomics diagnostics and research services specializing in precision oncology;
"Business Day" means any day (other than a Saturday or Sunday or public holiday) on which banks in Hong Kong, the Cayman Islands, Taiwan and the United States are open for the transaction of normal business, and on which no tropical cyclone warning No. 8 or above and no "black" rainstorm warning signal is hoisted in Hong Kong at any time between 8:00 a.m. and 6:00 p.m. (Hong Kong time);
"Business IP" means all Intellectual Property legally or beneficially owned by any Group Company, including all Registered Owned IP;
"Business IT" means all Information Technology which is owned or used by the Group Companies in conducting the Business of the Group;
"Buyer Notice" has the meaning given in paragraph 4 of Schedule 3;
"Buyer Warranty" means a statement contained in Schedule 8, and collectively the "Buyer Warranties";
"Buyer's Bank Account" means the bank account of the Buyer, details of which shall be delivered by the Buyer to the Holders' Representative at least three (3) Business Days before the Completion Date;
- 5 - | ||||||||
"Cash" means with respect to the Group on a consolidated basis, the aggregate of all unrestricted cash or cash equivalents held by each Group Company, including any cash balances credited to the account of each such Group Company with banks or other financial institutions as set out in that Group Company's nominal ledger (including any cash in transit from the Group Company to its banks and, for the avoidance of doubt, shall not be reduced by any overdrafts included as Indebtedness), in each case at the relevant time;
"Claim Notice" has the meaning given in clause 9.2.1;
"Company ESOP(s)" means the employee share option plan(s) of the Company duly adopted on (i) 24 August 2018 (as amended on 3 October 2028 and 21 November 2019), (ii) 20 September 2018 (as amended on 1 April 2020), (iii) 1 April 2020 (as amended on 16 November 2022) and (iv) 28 December 2023, as amended from time to time;
"Competing Business" has the meaning given in clause 12.2.1;
"Completion" means completion of the sale and purchase of all of the Sale Shares in accordance with this Agreement;
"Completion Date" has the meaning given in clause 6.1;
"Completion Statement" means the Draft Completion Statement as accepted, agreed or determined in accordance with Schedule 3;
"Condition" means a condition set out in clauses 5.1 or clause 5.2, as applicable;
"Confidential Information" means all information which relates to the business and affairs of any Group Company or any party and the Transaction Information, but does not include information:
(a)to the extent that it is generally known to the public not as a result of any breach of duty of confidentiality;
(b)(other than in respect of the Transaction Information) that was lawfully in the possession of the receiving party prior to its disclosure by the disclosing party; or
(c)(other than in respect of the Transaction Information) that is or becomes available to the receiving party other than as a result of a disclosure by a person which the receiving party knows is in breach of a duty of confidentiality owed to the disclosing party;
"Control" means the ownership, directly or indirectly, of more than 50 per cent. (50%) of the voting shares, registered capital or other equity interest of the relevant person or the possession, directly or indirectly, of the power to direct the shareholders' general meeting, to appoint or elect a majority of the directors, or otherwise to direct the management of the relevant person;
- 6 - | ||||||||
"Convertible Bond" means the convertible bond in the principal amount of US$[***] issued by the Company to Prenetics on 30 December 2022;
"CP Defaulting Party" has the meaning given in clause 5.6;
"CP Non-defaulting Party" has the meaning given in clause 5.65.6;
"Cure Period" has the meaning given in clause 5.6;
"Deed of Release" means the deed of release in relation to the outstanding Convertible Bond to be executed by Prenetics on or before the Completion Date, substantially in the form as set out in Appendix I to this Agreement;
"Defaulting Party" has the meaning given in clause 6.6;
"Designated Bank Account" means the bank account set out opposite the name of each Seller or each Option Holder (as applicable) in column (E) in Part A or Part B of Schedule 1 (as applicable), or otherwise provided to the Buyer no later than three (3) Business Days before the Completion Date;
"Determination Date" means the date on which the Completion Statement is accepted, agreed or otherwise determined in accordance with the provisions of Schedule 3;
"Disclosed" has the meaning given in clause 7.4;
"Disclosure Letter" means the letter from the Warrantors to the Buyer in relation to the Warrantor Warranties having the same date as this Agreement;
"Dispute" has the meaning given in clause 22.1;
"Draft Completion Accounts" has the meaning given in paragraph 1 of Schedule 3;
"Draft Completion Statement" has the meaning given in paragraph 3 of Schedule 3;
"Effective Time" means 12:01 a.m. HKT on the Completion Date;
"Encumbrance" means a mortgage, charge, pledge, lien, option, restriction, right of first refusal, right of pre-emption, third-party right or interest, assignment, deed of trust, other encumbrance or security interest of any kind, or another type of preferential arrangement (including a title transfer or retention arrangement) having similar effect;
"Escrow Agent" means CITIBANK, N.A., HONG KONG BRANCH;
"Escrow and Paying Agent Agreement" means the escrow and paying agent agreement by and among the Buyer, Prenetics, the Holders' Representative, the Escrow Agent and the Paying Agent, substantially in the form set out in Appendix III to this Agreement (subject to any changes reasonably requested by the Escrow Agent and the Paying Agent);
- 7 - | ||||||||
"Estimated Cash" means the Sellers' good faith estimate of the Cash of the Group as at the Effective Time, as set out in the Estimated Statement;
"Estimated Indebtedness" means the Sellers' good faith estimate of the Indebtedness of the Group as at the Effective Time, as set out in the Estimated Statement;
"Estimated Net Debt" means the amount equal to the difference between the Estimated Indebtedness and the Estimated Cash (expressed as a positive number, if the Estimated Indebtedness is greater than the Estimated Cash; or expressed as a negative number, if the Estimated Indebtedness is less than the Estimated Cash), in each case, as set out in the Estimated Statement;
"Estimated Statement" means a statement setting out the Estimated Cash, Estimated Indebtedness, Estimated Transaction Expenses and Estimated Working Capital Amount;
"Estimated Transaction Expenses" means the Sellers' good faith estimate of the Transaction Expenses as at the Effective Time, as set out in the Estimated Statement;
"Estimated Working Capital Adjustment" means the amount equal to the difference between the Estimated Working Capital Amount and the Target Working Capital Amount (expressed as a negative number, if the Estimated Working Capital Amount is less than the Target Working Capital Amount; or expressed as a positive number, if the Estimated Working Capital Amount is greater than the Target Working Capital Amount), in each case, as set out in the Estimated Statement;
"Estimated Working Capital Amount" means the Sellers' good faith estimate of the Working Capital as at the Effective Time, as set out in the Estimated Statement;
"Existing Shareholder Ordinary Shares" means the 'existing shareholder ordinary shares' of par value US$0.0001 each in the capital of the Company;
"Expiration Date" means the date that is eighteen (18) months from the Completion Date;
"Financial Investor Seller" means any of the Unrepresented Holders other than Prenetics.
"Fundamental Warranty Claim" means any claim made by the Buyer for breach of any Fundamental Warranty;
"Fundamental Warranties" means collectively, the Holder Warranties and paragraphs 2.1, 2.2, 2.3, 2.4 and 2.5 of Part B of Schedule 7, and "Fundamental Warranty" means any one of them;
"Government Authority" means any national, provincial, municipal or local government, administrative or regulatory body or department, court, tribunal, arbitrator or any entity that exercises the function of a regulator;
- 8 - | ||||||||
"Government Official" means:
(a)an employee, officer or representative of, or any person otherwise acting in an official capacity for or on behalf of, a Government Authority;
(b)a person holding any legislative, administrative or judicial position of any kind, regardless of whether elected or appointed;
(c)an officer of, or an individual who holds a position in, a political party;
(d)a candidate for political office;
(e)an individual who holds any other official, ceremonial or other appointed or inherited position with a government or any of its agencies; or
(f)an individual who exercises a public function for or on behalf of a country or territory or for any public agency or public enterprise of that country or territory;
"Group Company" means the Company or any of its Subsidiaries, and "Group" and "member of the Group" shall be construed accordingly;
"Headline Price" means the sum of the Sale Shares Headline Price and Option Shares Headline Price, and the maximum Headline Price shall not exceed US$[***] multiplied by the Sale Ratio;
"HKIAC" has the meaning given in clause 22.1;
"Holder Individual Claim" means a Holder Warranty Claim or a Relevant Claim for non-performance of a Holder's own obligations under this Agreement;
"Holder Warranty" means a statement contained in Part A of Schedule 7, and collectively the "Holder Warranties";
"Holder Warranty Claim" means any claim made by the Buyer for breach of any Holder Warranty;
"Holders" means all of the Sellers and Option Holders, each a "Holder";
"Holders' Expenses" has the meaning given in clause 16.3;
"Holders' Representative" has the meaning given in the preamble;
"Holders' Representative Expenses" has the meaning given in clause 11.2;
"Hong Kong" means the Hong Kong Special Administrative Region of the People's Republic of China;
- 9 - | ||||||||
"IFRS" means the International Financial Reporting Standards and applicable accounting requirements set by the International Accounting Standards Board or any successor thereto, as in effect from time to time;
"Immediate Family Members" means:
(a)spouse, parents, parents-in-law, grandparents, children, grandchildren, siblings and spouses of children, grandchildren and siblings (in each case, whether adoptive or biological); and
(b)estates, trusts, partnerships and other persons which directly or indirectly through one or more intermediaries are Controlled by the foregoing;
"Indebtedness" means, as of any particular time with respect to the Group on a consolidated basis, the aggregate amount (expressed as a positive number) of any Liability, without duplication, (a) for money borrowed from third parties evidenced by loans, notes, letter of credit, bonds, financial support, debentures or similar instruments and bank overdrafts or negative balance cash accounts, (b) for all or any part of the deferred purchase price for property or services, including all seller notes and "earn-out" payments, all conditional sale obligations and all obligations, (c) for any finance or capital leases, (d) for any factoring arrangements, currency or interests swap, and/or other interest or currency protection, hedging or financial futures transaction or arrangement, (e) for accounts payable or accruals not reflected in the calculation of Working Capital, (f) for declared but unpaid dividends or distributions, (g) for guaranties with respect to any of the foregoing, (h) for all principal, interest, penalties, fees and expenses in relation to any of the foregoing, (i) for all cut but uncashed checks issued by any Group Company, but excluding, for the avoidance of doubt, (i) any short-term Liabilities comprising Working Capital at the relevant time; (ii) any Transaction Expenses; (iii) any aged A/P which has been written off pursuant to this Agreement, (iv) the Intercompany Debt; and (v) the Convertible Bond and any interest payable in relation to the Convertible Bond;
"Indemnification Percentage" means, with respect to any Indemnifying Party, a ratio (expressed as a percentage) equal to (i) the portion of the Purchase Price actually received by such Indemnifying Party has the right to receive, divided by (ii) the aggregate Purchase Price actually received by all Indemnifying Parties; provided that, Indemnification Percentage of an Indemnifying Party shall be deemed to be 100% with respect to any Holder Individual Claim;
"Indemnifying Party" or "Indemnifying Parties" has the meaning given in clause 9.1;
"Indemnity Escrow Account" means an account designated by the Escrow Agent, into which the Indemnity Escrow Amount shall be deposited pursuant to clause 6.2.2;
"Indemnity Escrow Amount" means an amount in U.S. Dollars equal to 10% of the aggregate Initial Consideration;
- 10 - | ||||||||
"Indemnity Escrow Fund" means the Indemnity Escrow Amount deposited with the Escrow Agent, as such sum may be increased or decreased as provided in this Agreement and the Escrow and Paying Agent Agreement, including any remaining interest or other amounts earned thereon;
"Indemnity Notice" has the meaning given in clause 9.3.1;
"Information Technology" means material computer systems, communication systems, software, hardware, databases and related services;
"Initial Consideration" has the meaning given in clause 3.1;
"Initial Consideration Per Option Share" means, with respect to each Option Share, an amount equal to the Initial Lower Per Share Price, multiplied by the Adjustment Ratio;
"Initial Consideration Per Sale Share" means:
(a) with respect to each Sale Share subject to Initial Higher Per Share Price, an amount equal to the Initial Higher Per Share Price, multiplied by the Adjustment Ratio; or
(b) with respect to each Sale Share subject to Initial Lower Per Share Price, an amount equal to the Initial Lower Per Share Price, multiplied by the Adjustment Ratio;
"Initial Higher Per Share Price" means US$[***];
"Initial Lower Per Share Price" means US$[***];
"Instrument of Transfer" means the instrument of transfer in the form set out in Appendix IV to this Agreement.
"Intellectual Property" means all industrial and intellectual property rights, whether registered or not, including pending applications for registration of such rights and the right to apply for registration or extension of such rights including patents, petty patents, utility models, design patents, designs, copyright (including moral rights and neighbouring rights), database rights, rights in integrated circuits and other sui generis rights, trade marks, trading names, company names, service marks, logos, the get-up of products and packaging, geographical indications and appellations and other signs used in trade, internet domain names, social media user names, rights in know-how (including information comprised in formulae, algorithms, techniques, specifications, computation models, software and manuals), trade secrets, right to use and protection of the confidentiality of Confidential Information and any rights of the same or similar effect or nature as any of the foregoing anywhere in the world;
"Intercompany Debts" means any and all amounts owed by any Group Company to another Group Company or its parent company, whether currently due or to become due, and whether the debts are present or future, actual or contingent;
- 11 - | ||||||||
"Investor Ordinary Shares" means the 'investor ordinary shares' of par value US$0.0001 each in the capital of the Company;
"Last Accounting Date" means 31 December 2023;
"Liabilities" means any and all debts, liabilities and obligations of any kind or nature, whether accrued or fixed, absolute or contingent, matured or unmatured, or determined or determinable;
"Longstop Date" means the date which is six (6) months following the date of this Agreement, or such later date as the Buyer and the Sellers may agree in writing;
"Loss" means all costs, claims, damages, Liabilities, losses and expenses (including Tax), including reasonable legal costs actually incurred as a result of asserting, defending or settling a claim alleging a Liability, excluding any punitive damages and consequential loss to the extent such loss is not a reasonably foreseeable consequence of the fact, matter or circumstance giving rise to the relevant loss, and "Losses" shall be construed accordingly;
"LTC Agreement" means the License and Supply Agreement executed between [***] and ACT Genomics Co., Ltd on January 6, 2023.
"Management Accounts" means the unaudited consolidated profit and loss account of the Group in respect of the period starting on the day after the Last Accounting Date and ending on the Management Accounts Date and the unaudited consolidated balance sheet of the Group as at the Management Accounts Date to which the relevant Management Accounts have been drawn up; in each case, provided by the Company to the Buyer prior to the date of this Agreement;
"Management Accounts Date" means 31 December 2024;
"Material Adverse Effect" means, with respect to any person, any change, fact, circumstance, event, condition, development, effect or occurrence that, individually or in the aggregate with all other changes, facts, circumstances, events, conditions, developments, effects or occurrences has had or would reasonably be expected to (i) have a material adverse effect on the condition (financing, trading or otherwise), assets, Liabilities, revenue, profits, prospects, operations, results or business of the Group taken as a whole; or (ii) prevent the Sellers from consummating, or materially impair the ability of the Sellers to consummate, the transactions contemplated under this Agreement, but excluding any results, occurrences, facts, changes, conditions, circumstances, events or effects to the extent arising from:
(a)any change or fluctuation in financial markets, interest rates, exchange rates, commodity prices or other general economic, business or political or geopolitical conditions;
(b)any natural disaster, including any hurricane, flood, tornado, earthquake or other natural disaster;
- 12 - | ||||||||
(c)any changes in the Applicable Law or applicable accounting standards and principles, including the IFRS, or the enforcement or interpretation in respect of any of the foregoing;
(d)any act of civil unrest, war, sabotage, cyberattack, terrorism or military action, or any outbreak or escalation of hostilities involving the United States or another sovereign political power, the declaration by the United States, the Taiwan Government Authority or another sovereign political power of a national emergency or war or the occurrence of any other calamity or crisis, including the war in Ukraine, tension across the Taiwan Strait, or the escalation or worsening of any of the foregoing;
(e)changes in conditions generally affecting the industry in which the Group operates;
(f)any action or omission required pursuant to the express terms and conditions of this Agreement or any other Transaction Documents or otherwise required at the express written direction of the Buyer; or
(g)the negotiation, execution and delivery of this Agreement or other Transaction Documents, or the public announcement or pendency of the transactions contemplated hereby,
except in the case of items (a) to (e) above, to the extent that such change or event does not materially and disproportionately affect the Group when compared with other businesses operating in the same industry;
"Material Contract" has the meaning given in paragraph 9.2.1 of Part B of Schedule 7;
"Notice" has the meaning given in clause 20.1;
"NT$" or "NTD" means New Taiwanese dollars, the lawful currency of Taiwan;
"Option Holders" means the holders of the Options, whose names and addresses as of the date of this Agreement are set out in column (A) in Part B of Schedule 1;
"Option Shares" means the Other Ordinary Shares issuable to each Option Holder pursuant to an exercise of the Options pursuant to this Agreement;
"Option Shares Headline Price" means the total number of Option Shares, multiplied by an amount equal to the Initial Lower Per Share Price less the applicable exercise price of such Options.
"Option Shares Initial Consideration" means, in respect of an Option Holder, the amount equal to the Initial Consideration Per Option Share multiplied by the total number of Option Shares of such Option Holder;
- 13 - | ||||||||
"Options" means the options granted by the Company pursuant to the Company ESOP;
"Ordinary Course of Business" means the ordinary and usual course of the operations of the Business of the relevant Group Company and/or the Group, conducted on an arm's length basis in compliance in all material respects with Applicable Laws and in accordance with the relevant Group Company's and/or the Group's practices or policies consistently applied over the twenty-four (24) months preceding the date of this Agreement;
"Other Ordinary Shares" means the 'other ordinary shares' of par value US$0.0001 each in the capital of the Company;
"Paying Agent" has the meaning given in clause 3.2.
"Personal Information" means any information about an identifiable individual (including any Group Company's employees, customers, suppliers, directors and officers), regardless of whether the information is Confidential Information;
"Prenetics" means Prenetics Global Limited, a company incorporated under the laws of the Cayman Islands, trading on NASDAQ with ticker PRE, having its principal executive office at Unit 703-706, K11 Atelier King's Road, 728 Kings Road, Hong Kong;
"Pro-Rata Share" means, with respect to each Holder, a ratio (expressed as a percentage) equal to (a) the amount of Initial Consideration actually received by such Holder, divided by (b) the aggregate Initial Consideration actually received by all Holders;
"Properties" means the properties of the Group as Disclosed in the Disclosure Letter;
"Protected Information" means Confidential Information and Personal Information;
"Purchase Price" has the meaning given in clause 4.5;
“Redesignation of Class” means the redesignation of the three classes of shares into one single class of shares for the Company, where all outstanding issued Investor Ordinary Shares, Existing Shareholder Ordinary Shares and Other Ordinary Shares to be redesignated into a single class of Ordinary Shares.
"Registered Owned IP" has the meaning given in paragraph 8.1.1 of Part B of Schedule 7;
"Reimbursement Cap" is US$[***];
"Relevant Claim" means any Warranty Claim, or any claim made by the Buyer for non-performance of any Holder's or Warrantor's obligations under this Agreement;
- 14 - | ||||||||
"Relief" means any loss, relief, allowance, exemption, set-off, deduction, right to repayment or credit or other relief of a similar nature granted by or available in relation to Tax pursuant to any legislation or otherwise;
"Reporting Accountants" means PWC Taiwan, unless another "Big Four" accounting firm in Taiwan is agreed between the Buyer and the Holders' Representative;
"Representatives" has the meaning given in clause 8.4;
"Represented Holders" means all of the Holders other than the Unrepresented Holder;
"Rules" has the meaning given in clause 22.1;
"Sale Ratio" means the ratio (expressed as a percentage) equal to (a) the total number of Sale Shares divided by (b) the total number of the Shares in the share capital of the Company on a fully-diluted basis as of the Completion;
"Sale Shares" means the Shares to be sold by the Sellers and Option Holders in accordance with this Agreement; and, with respect to each Seller, means those shares set out against his/her/its name in column (B) in Part A of Schedule 1 as of the date of this Agreement, and with respect to each Option Holder, means those Option Shares set out again his/her name in column (B) of Part B of Schedule 1 as of the date of this Agreement; which, in each case, may be further updated by the Company no later than eight (8) Business Days before the Completion to reflect any change in the number of Sale Shares, including, without limitation, any change resulting from any issuance of Option Shares by the Company or any forfeiture of Options pursuant to the terms of the Company ESOP;
"Sale Shares Headline Price" means the sum of:
(a)the total number of Sale Shares to be sold by all Sellers subject to the Initial Higher Per Share Price (as indicated in column (C) in Part A of Schedule 1), multiplied by the Initial Higher Per Share Price; and
(b)the total number of Sale Shares to be sold by all Sellers subject to the Initial Lower Per Share Price (as indicated in column (C) in Part A of Schedule 1), multiplied by the Initial Lower Per Share Price;
"Sale Shares Initial Consideration" means, in respect of a Seller, the amount equal to the applicable Initial Consideration Per Sale Share multiplied by the total number of Sale Shares of such Seller;
"Shareholder" means a holder of Shares in the Company;
"Shareholders' Agreement" means the shareholders' agreement in relation to the Company dated 30 December 2022, as supplemented and amended;
- 15 - | ||||||||
"Shares" means the Investor Ordinary Shares, the Existing Shareholder Ordinary Shares and the Other Ordinary Shares;
“Special Tax Claim” means any claim made by the Buyer in respect of the Taxes and Liabilities from any of the matters specified in Part B of Schedule 12;
"Special Tax Expiration Date" means the date that is two (2) years from the Completion Date;
"Subsidiaries" means the subsidiaries of the Company listed in Part B of Schedule 2 as of the date of this Agreement;
"SG JV" means CERBACT Asia Holdings Pte. Ltd. a limited liability company incorporated in Singapore with its registered office at 380 Jalan Besar #06-06 ARC 380 Singapore 209000;
"SG JVA" means the joint venture shareholders' agreement in relation to SG JV dated 11 August 2021 between ACT Singapore, CERBACT Research NV and SG JV;
"Target Working Capital Amount" means US$[***];
"Tax" and "Taxation" mean any form of taxation, levy, duty, charge, contribution, withholding or impost of whatever nature (including any related fine, penalty, surcharge or interest) imposed, collected or assessed by, or payable to, a Tax Authority;
"Tax Indemnity Escrow Account" means an account designated by the Escrow Agent, into which the Tax Indemnity Escrow Amount shall be deposited pursuant to clause 6.2.2;
"Tax Indemnity Escrow Amount" means US$[***];
"Tax Indemnity Escrow Fund" means the Tax Indemnity Escrow Amount deposited with the Escrow Agent, as such sum may be increased or decreased as provided in this Agreement and the Escrow and Paying Agent Agreement, including any remaining interest or other amounts earned thereon;
"Tax Authority" mean any government, state or municipality or any local, state, federal or other authority, body or official anywhere in the world exercising a fiscal, revenue, customs or excise function;
"Tax Warranty" means a statement contained in paragraph 14 of Part B of Schedule 7;
"Tax Warranty Claim" means any claim made by the Buyer for breach of any Tax Warranty;
"Third Parties Ordinance" means the Contracts (Rights of Third Parties) Ordinance (Cap. 623, Laws of Hong Kong);
- 16 - | ||||||||
"TIC" means the Ministry of Economic Affairs Investment Commission of Taiwan;
"Transaction Documents" means:
(a)this Agreement;
(b)the Disclosure Letter;
(c)the Deed of Release;
(d)the Amended Articles; and
(e)any other document(s) and instrument(s) designated as Transaction Document(s) as agreed between the Buyer, Prenetics and the Holders' Representative;
"Transaction Expenses" means, without duplication, in each case to the extent unpaid as of Completion, the out-of-pocket fees and expenses (if any) incurred and payable by the Group prior to or at Completion in connection with this Agreement and the Transaction Documents and the transactions contemplated hereby (including any such fees and expenses payable upon consummation of the Completion). For the avoidance of doubt, the Transaction Expenses shall not include (i) the fees and expenses of the Holders' Representative, if any; (ii) any Holders' Expenses; (iii) any amount already included in the calculation of Indebtedness or Working Capital;
"Transaction Information" means any information relating to the terms of the transactions contemplated under and the provisions or subject matter of this Agreement or any other Transaction Documents;
"Tribunal" has the meaning given in clause 22.2.1;
"Unrepresented Holder" means each of the Holders whose names are set out in Part C of Schedule 1;
"US$" or "USD" means United States dollars, the lawful currency of the United States of America;
"Warrantor" means each of [***], [***], [***] and [***], and collectively the "Warrantors";
"Warrantor Warranty" means a statement contained in Part B of Schedule 7, and collectively the "Warrantor Warranties";
"Warrantor Warranty Claim" means any claim made by the Buyer for breach of any Warrantor Warranty;
"Warranty" means a Warrantor Warranty or a Holder Warranty, as applicable, and collectively the "Warranties";
- 17 - | ||||||||
"Warranty Claim" means any claim made by the Buyer for breach of a Warranty under this Agreement, including Holder Warranty Claim and Warrantor Warranty Claim; and
"Working Capital" means the aggregate value, at the relevant time, of:
(a)(i) trade receivables, and (ii) inventories; minus
(b)(i) trade payables,
in each case, of the Group on a consolidated basis.
1.2.In this Agreement, a reference to:
1.2.1a "subsidiary" or "holding company" is to be construed in accordance with sections 13 to 15 of the Companies Ordinance (Cap. 622, Laws of Hong Kong);
1.2.2Liability under, pursuant to or arising out of (or any analogous expression) any agreement, contract, deed or other instrument includes a reference to contingent Liability under, pursuant to or arising out of (or any analogous expression) that agreement, contract, deed or other instrument;
1.2.3a party being liable to another party, or to Liability, includes any Liability in equity, contract or tort (including negligence) or under the Misrepresentation Ordinance (Cap. 284, Laws of Hong Kong);
1.2.4a document in the "agreed form" is a reference to a document in a form approved and confirmed in writing by or on behalf of each party;
1.2.5a statutory provision includes a reference to the statutory provision as modified or re-enacted or both from time to time before the date of this Agreement and any subordinate legislation made under the statutory provision (as so modified or re-enacted) before the date of this Agreement;
1.2.6a "person" includes a reference to any individual, firm, company, corporation or other body corporate, government, state or agency of a state or any joint venture, association or partnership, works council or employee representative body (whether or not having separate legal personality);
1.2.7a person includes a reference to that person's legal personal representatives, successors and permitted assigns;
1.2.8a "party" includes a reference to that party's successors and permitted assigns;
1.2.9a clause, paragraph, Schedule or Appendix, unless the context otherwise requires, is a reference to a clause or paragraph of, Schedule or Appendix to, this Agreement;
- 18 - | ||||||||
1.2.10any Hong Kong legal term for any action, remedy, method of judicial proceeding, legal document, legal status, court, official or any legal concept or thing shall in respect of any jurisdiction other than Hong Kong be deemed to include what most nearly approximates in that jurisdiction to the Hong Kong legal term and to any Hong Kong ordinance or regulation shall be construed so as to include equivalent or analogous laws of any other jurisdiction;
1.2.11time of the day is to Hong Kong/Taiwan time;
1.2.12the singular includes the plural and vice versa; and
1.2.13one gender includes all genders.
1.3.The ejusdem generis principle of construction shall not apply to this Agreement. Accordingly, general words shall not be given a restrictive meaning by reason of their being preceded or followed by words indicating a particular class of acts, matters or things or by examples falling within the general words. Any phrase introduced by the terms "other", "including", "include" and "in particular" or any similar expression shall be construed as illustrative and shall not limit the sense of the words preceding those terms.
1.4.The headings in this Agreement do not affect its interpretation.
1.5.A reference in Schedule 7 to "to each Warrantor's knowledge" includes the actual knowledge, information and belief of each of the Warrantors acquired on or after the date on which he becomes an employee of any Group Company and such knowledge, information and belief that such persons would reasonably be expected to have after making due and diligent inquiries of relevant persons, documents, and records within the Group Company.
1.6.Notwithstanding anything contained herein to the contrary, all Liabilities and obligations of the Sellers, Option Holders and/or the Warrantors shall be several and not joint.
2.Sale and Purchase¶
2.1.Sale Shares. Subject to the terms and conditions of this Agreement, each Seller agrees to sell to the Buyer or a wholly-owned subsidiary of and designated by Buyer, and the Buyer agrees to buy or cause a wholly-owned subsidiary of Buyer to buy, the Sale Shares set out opposite such Seller's name in column (B) in Part A of Schedule 1 (being all Shares legally and beneficially owned and held by such Seller) and each right attaching to such Sale Shares at Completion, free from any Encumbrance, in each case, in consideration of the payment by the Buyer (or its designee) of such amount set out opposite each such Seller’s name in column (D) in Part A of Schedule 1, amounting to the Sale Shares Headline Price in aggregate prior to all adjustments contemplated herein.
2.2.Consent; Waiver of Rights. Each Seller hereby consents to the sale and purchase of the Sale Shares, and waives all rights of pre-emption, first refusal, tag-along, co-sale
- 19 - | ||||||||
and other restrictions on transfer over, the Sale Shares conferred on them (if any), whether conferred by law, the Company's constitutional documents, the Shareholders' Agreement, or any other agreement or arrangement to which such Seller is a party, so as to permit the sale and purchase of the Sale Shares.
2.3.Option Shares; Company ESOP. Subject to and conditional upon the Completion:
2.3.1without any further action on the part of the Company or any Option Holder, on the Completion Date, all Options shall automatically vest, and any Options not exercised upon the Completion Date shall be deemed to be forfeited according to the terms of the Company ESOP. The Options exercised by the relevant Option Holder will be exercised into such number of Option Shares as set out opposite such Option Holder's name in column (B) in Part B of Schedule 1, in each case, in accordance with the terms of the Company ESOP; and the Buyer shall pay on behalf of such relevant Option Holders, or pay to the Paying Agent and cause the Paying Agent to pay, on the Completion Date, the applicable exercise price of such Options Shares to a bank account in the name of the Company for receiving such exercise price, as designated by Company at least three (3) Business Days prior to the Completion Date;
2.3.2the Warrantors shall cause all of the Option Holders to execute and deliver to the Buyer a deed of accession to this Agreement to sell to the Buyer or a wholly-owned subsidiary of and designated by Buyer, and the Buyer hereby agrees to purchase or cause a wholly-owned subsidiary of Buyer to purchase from the Option Holders, all of the Option Shares to be issued to the Option Holders pursuant to clause 2.3.1, in each case in consideration of the payment by the Buyer (or its designee) of such amount set out opposite such Option Holder's name in column (D) in Part B of Schedule 1, amounting to the Option Shares Headline Price in aggregate prior to all adjustments contemplated herein; and
2.3.3The Company ESOP shall be terminated at the Completion.
3.Initial Consideration¶
3.1.Initial Consideration. The parties agree that the aggregate consideration for the sale of the Sale Shares payable by the Buyer (or its designee) at Completion (the "Initial Consideration") shall be an amount equal to the Headline Price, as adjusted as follows:
3.1.1increased by the Estimated Working Capital Adjustment if it is a positive number or reduced by the Estimated Working Capital Adjustment if it is a negative number, in each case multiplied by the Sale Ratio;
3.1.2increased by Estimated Net Debt if it is a negative number or reduced by Estimated Net Debt if it is a positive number, in each case multiplied by the Sale Ratio; and
3.1.3reduced by Estimated Transaction Expenses.
- 20 - | ||||||||
For the purposes of determining the Initial Consideration, the Holders' Representative shall deliver to the Buyer the Estimated Statement no later than ten (10) Business Days prior to the Completion Date.
3.2.Payment. Prior to Completion, the Buyer shall designate CITIBANK, N.A., HONG KONG BRANCH to act as paying agent in connection with payment of the Purchase Price ("Paying Agent") pursuant to the Paying Agent Agreement providing for, among other things, the matters set forth in this clause 3.2. On the Completion Date, the Buyer shall pay or cause to be paid (i) the Initial Consideration, less the Adjustment Escrow Amount, less the Indemnity Escrow Amount, less the Tax Indemnity Escrow Amount, and less 50% of the total fees and costs of the Paying Agent and Escrow Agent under the Escrow and Paying Agent Agreement, by wire transfer of immediately available funds in U.S. Dollars to the Paying Agent, to such account as may be designated in writing by the Paying Agent prior to the Completion Date, and (ii) 100% of the total fees and costs of the Paying Agent and Escrow Agent under the Escrow and Paying Agent Agreement respectively to the Paying Agent and Escrow Agent, which payments shall discharge in full all of the Buyer’s obligation of payment of the Initial Consideration as at Completion under this Agreement. The Paying Agent shall further pay such funds in the following manner:
3.2.1in respect of each Seller other than Prenetics, the amount equal to (a) its/his/her Initial Consideration Per Sale Share multiplied by the number of Sale Shares of such Seller minus (b) its/his/her Pro-Rata Share of the Holders' Expenses (the amount of which shall be delivered by Prenetics to the Buyer in writing no later than eight (8) Business Days before the Completion Date) minus (c) its/his/her Pro-Rata Share of the Adjustment Escrow Amount, the Indemnity Escrow Amount, and the Tax Indemnity Escrow Amount, to such Seller's Designated Bank Account;
3.2.2in respect of each Option Holder, the amount equal to (a) its/his/her Initial Consideration Per Option Share multiplied by the number of Option Shares of such Option Holder minus (b) its/his/her Pro-Rata Share of the Holders' Expenses (the amount of which shall be delivered by Prenetics to the Buyer in writing no later than eight (8) Business Days before the Completion Date) minus (c) its/his/her Pro-Rata Share of the Adjustment Escrow Amount, the Indemnity Escrow Amount, and the Tax Indemnity Escrow Amount, to such Option Holder's Designated Bank Account; and
3.2.3in respect of Prenetics, the amount equal to (a) its Initial Consideration Per Sale Share multiplied by the number of Sale Shares of Prenetics, minus (b) its Pro-Rata Share of the Adjustment Escrow Amount, the Indemnity Escrow Amount, and the Tax Indemnity Escrow Amount, plus (c) the aggregate amount deducted from the Sale Shares Initial Consideration payable to each other Seller pursuant to clause 3.2.1 and from the Option Shares Initial Consideration payable to each Option Holder pursuant to clause 3.2.2, to Prenetics' Designated Bank Account.
- 21 - | ||||||||
Each Holder hereby acknowledges and agrees that the Buyer and the Paying Agent each shall have no obligation to verify, and shall have full right to rely solely upon, Prenetics’ calculation of the Holders’ Expenses and each Holder’s Pro-Rata Share thereof as delivered to the Buyer in writing in accordance with this clause 3.2. Any inaccuracy, discrepancy or other dispute with respect to Holders’ Expenses and/or each Holder’s Pro Rata Share thereof shall be solely on account of Prenetics, without any liability to the Buyer.
4.Post-Completion Adjustment¶
4.1.The Initial Consideration paid by the Buyer shall be subject to further adjustments after the Completion pursuant to clauses 4.2, 4.3 and 4.4 (as applicable), as determined in accordance with Schedule 3.
4.2.If the Actual Working Capital Amount exceeds or is less than the Target Working Capital Amount:
4.2.1where the Actual Working Capital Adjustment exceeds the Estimated Working Capital Adjustment, the Buyer shall pay to the Holders an amount equal to such excess; or
4.2.2where the Actual Working Capital Adjustment is less than the Estimated Working Capital Adjustment, the Holders shall, severally and not jointly, pay the Buyer an amount equal to such shortfall,
in either case, in accordance with the provisions of clauses 4.5 and 4.6.
4.3.If the Actual Net Debt:
4.3.1exceeds the Estimated Net Debt, the Holders shall, severally and not jointly, pay the Buyer an amount equal to such excess; or
4.3.2is less than the Estimated Net Debt, the Buyer shall pay to the Holders an amount equal to such shortfall,
in either case, in accordance with the provisions of clauses 4.5 and 4.6.
4.4.If the Actual Transaction Expenses:
4.4.1exceed the Estimated Transaction Expenses, the Holders shall, severally and not jointly, pay the Buyer the amount of difference,
4.4.2is less than the Estimated Transaction Expenses, the Buyer shall pay to the Holders an amount equal to such shortfall,
in either case, in accordance with the provisions of clauses 4.5 and 4.6.
4.5.The amounts (if any) payable pursuant to clauses 4.2, 4.3 or 4.4 shall be set off against each other so that only any balance payable (such balance being the "Adjustment Amount", and the amount equal to the Initial Consideration as adjusted
- 22 - | ||||||||
by the Adjustment Amount being the "Purchase Price") shall be paid on any date on which a payment is to be made pursuant to clause 4.6.
4.6.If the Buyer shall make a payment of Adjustment Amount as determined in accordance with clause 4.5 to the Holders pursuant to this clause 4, the Buyer shall, within ten (10) Business Days of the Determination Date, pay the Adjustment Amount to the Paying Agent, for the Paying Agent to pay to each Holder their respective Pro-Rata Share of the Adjustment Amount by wire transfer of immediately available funds in U.S. Dollars for same day value, to such Holder's Designated Bank Account. If the Holders shall make a payment of Adjustment Amount as determined in accordance with clause 4.5 to the Buyer, then (i) the Buyer, Prenetics and Holders' Representative shall, within five (5) Business Days of the Determination Date, deliver a joint written instruction to the Escrow Agent to pay to the Buyer an amount equal to the Adjustment Amount from the Adjustment Escrow Account to the Buyer's Bank Account; and (ii) solely to the extent the Adjustment Amount exceeds the Adjustment Escrow Amount, then (A) the Holder's Representative and Buyer shall, within five (5) Business Days of the Determination Date, deliver a joint written notice to each Holder; and each Holder shall, within ten (10) Business Days from the date of receipt of such written notice, pay such Holder’s respective Pro-Rata Share of such excess amount to the Buyer by wire transfer of immediately available funds in U.S. Dollars for same day value to the Buyer's Bank Account, or (B) at the Buyer's sole option and discretion, the Buyer may elect to satisfy such shortfall from the Indemnity Escrow Funds, in which case, upon the Buyer’s written request, the Buyer, Prenetics and Holders' Representative shall promptly deliver a joint written instruction to the Escrow Agent to pay to the Buyer an amount equal to such shortfall from the Indemnity Escrow Account to the Buyer's Bank Account.
4.7.Within three (3) Business Days following the determination of the Adjustment Amount in accordance with clause 4.5, or the completion of payments in accordance with in clause 4.6, as applicable, the Buyer, Prenetics and Holders' Representative shall deliver a joint written instruction to the Escrow Agent to distribute the balance of the Adjustment Escrow Account, if any, to the Holders in accordance with their respective Pro-Rata Share pursuant to the terms of the Escrow and Paying Agent Agreement.
5.Conditions¶
5.1.Conditions of Buyer's Obligations at Completion. Completion by the Buyer is conditional on the following Conditions being satisfied, or waived by the Buyer in accordance with clause 5.5 before the Long Stop Date:
5.1.1each of the Holder Warranties and Fundamental Warranties remaining true, accurate and not misleading in all respects as of the date of this Agreement and as at Completion, except to the extent any such Holder Warranty and Fundamental Warranty expressly relates to a specific date (in which case it shall be true, accurate and not misleading in all respects as of such specific date);
- 23 - | ||||||||
5.1.2each of the Warrantor Warranties (other than Fundamental Warranties) shall be true, accurate and not misleading (i) in all respects for those that are qualified by any materiality or similar qualifiers and (ii) in all material respects for those not so qualified, in each case as of the date of this Agreement and as at Completion, except to the extent any such Warrantor Warranty expressly relates to a specific date (in which case it shall be true, accurate and not misleading (i) in all respects if qualified by any materiality or similar qualifiers and (ii) in all material respects if not so qualified, as of such specific date);
5.1.3there having been no material breach by any Holder of its obligations under this Agreement between the date of this Agreement and Completion;
5.1.4sufficient number of Shareholders having entered into this Agreement with the Buyer (whether by way of executing this Agreement on the date of this Agreement or subsequently by way of a duly executed deed of accession to this Agreement), such that the aggregate number of Sale Shares shall represent: (a) more than 90% of the share capital of the Company on a fully-diluted basis, (b) more than 90% of all issued and outstanding Investor Ordinary Shares of the Company, (c) more than 90% of all issued and outstanding Existing Shareholder Ordinary Shares of the Company, and (d) more than 90% of all issued and outstanding Other Ordinary Shares of the Company, in each case, immediately prior to Completion;
5.1.5the execution, delivery and performance of this Agreement and the consummation of the transactions contemplated under this Agreement having been duly approved and adopted by a duly convened board meeting and Shareholders' meeting (including separate meetings of each class of Shareholders) of the Company (or by way of written resolutions of the board of directors and/or the Shareholders in accordance with the Memorandum and Articles of Association of the Company), and the approvals remain in full force and effect as at Completion and not been revoked prior to Completion;
5.1.6any and all consents, approvals and waivers in connection with the execution, delivery and performance of this Agreement and the consummation of the transactions contemplated under this Agreement by the Sellers and the Warrantors set out in Part A of Schedule 11 having been obtained and remaining in full force and effect as at Completion, and no such consent, approval or clearance having been revoked or modified prior to Completion;
5.1.7there being no litigation, regulatory action or other legal, regulatory, or other administrative proceedings that would reasonably be expected to prohibit, enjoin, challenge, interfere or delay the consummation of any of the transactions contemplated under this Agreement;
5.1.8no Material Adverse Effect having occurred between the date of this Agreement and Completion;
- 24 - | ||||||||
5.1.9the Company having duly approved and adopted the Amended Articles and approved the Redesignation of Class, effective on and from the Completion Date;
5.1.10the Shareholders' Agreement having been duly amended in accordance with its terms, to provide for automatic termination of the Shareholders' Agreement upon the closing of any transaction resulting in the transfer of more than 90% of the issued and outstanding Shares in the Company for consideration (the "Amended Shareholders' Agreement");
5.1.11ACT Thailand's two minority shareholders other than [***] (i.e. [***] and [***]) having executed appropriate transfer documentation to transfer all of their shares and equity interests in ACT Thailand to Buyer's designated person(s);
5.1.12each of the following accounting and finance related actions having been duly taken and completed to Buyer’s reasonable satisfaction prior to Completion:
5.1.12.1.all Intercompany Debts as of the Completion Date (other than (i) the intercompany loan between [***] and [***] in the principal amount of approximately US$[***] and (ii) intercompany A/R and intercompany A/P that, in each case, arise within 30 days prior to the Completion Date) shall have been fully settled;
5.1.12.2.all Aged A/R shall have been appropriately provisioned and shall be excluded from the calculation of Actual Working Capital;
5.1.12.3.the Company shall have fully paid any underpayment of Royalty Fee for 2023 and 2024 under the LTC Agreement;
5.1.12.4.any and all inventory that expire as of the Completion Date (but excluding those expressly accepted by Prenetics on behalf of CircleDNA) shall have been written-off (and, for the avoidance of doubt, shall be excluded from line item “Inventory” in Schedule 4, Part A for purposes of calculation of Actual Working Capital);
5.1.12.5.the Convertible Bond shall have been fully released and waived (without interest, penalty or other claims) by Prenetics in 2024 and duly reflected in the audited financial report of the Group for the financial year ending on 31 December 2024; and
5.1.12.6.each of the items of Taxes specified in Part A of Schedule 12 shall have been duly filed and paid by the relevant Group Company, and tax receipts evidencing full payment of Taxes thereof shall have been provided to the Buyer;
5.1.13each of the following non-compliances having been appropriately rectified to the Buyer's reasonable satisfaction prior to Completion:
- 25 - | ||||||||
5.1.13.1.[***];
5.1.13.2.[***]; and
5.1.13.3.[***];
5.1.14the following third-party consents and/or waivers with respect to the transactions contemplated under this Agreement having been obtained:
5.1.14.1.[***]; and
5.1.14.2.[***];
5.1.15the Company having entered into the following agreements:
5.1.15.1.a CircleDNA Sequencing Service Agreement with Prenetics for CircleDNA for a period of three years after the Completion; and
5.1.15.2.a Circle DNA Data Sharing and Use Agreement with Prenetics for accessing sequencing data for research purpose;
5.1.16[***]; and
5.1.17the audited financial report of the Group for the financial year ended on 31 December 2024 shall have been submitted to the Buyer.
5.2.Conditions of Sellers' Obligations at Completion. Completion by the Sellers is conditional on the following Conditions being satisfied, or jointly waived by the Holders' Representative (on behalf of the Represented Holders) and each Unrepresented Holder in accordance with clause 5.5 before the Long Stop Date:
5.2.1each of the Buyer Warranties shall be true, accurate and not misleading (i) in all respects for those that are qualified by any materiality or similar qualifiers and (ii) in all material respects for those not so qualified, in each case as of the date of this Agreement and as at Completion, except to the extent any such Buyer Warranty expressly relates to a specific date (in which case it shall be true, accurate and not misleading (i) in all respects for if qualified by any materiality or similar qualifiers and (ii) in all material respects if not so qualified, as of such specific date);
5.2.2any and all consents, approvals and waivers in connection with the execution, delivery and performance of this Agreement and the consummation of the transactions contemplated under this Agreement by the Buyer having been obtained and remaining in full force and effect as at Completion, and no such consent, approval or clearance having been revoked or modified prior to Completion; and
5.2.3there being no litigation, regulatory action or other legal, regulatory or other administrative proceedings that would reasonably be expected to prohibit,
- 26 - | ||||||||
enjoin, challenge, interfere or delay the consummation of any of the transactions contemplated under this Agreement.
5.3.Reasonable Endeavours to Satisfy Conditions. Each Seller (in the capacity as a Shareholder) and each Warrantor shall use its reasonable endeavours to achieve satisfaction of each of the Conditions set out in clause 5.1 as soon as possible after the date of this Agreement and in any event not later than the Longstop Date. The Buyer shall use its reasonable endeavours to achieve satisfaction of each of the Conditions set out in clause 5.2 as soon as possible after the date of this Agreement and in any event not later than the Longstop Date.
5.4.Notification. If, at any time, any of the parties becomes aware of a fact or circumstance that might prevent a Condition being satisfied by the Longstop Date, it shall immediately inform the Buyer (in the case of any Seller or any Warrantor becoming aware of such fact or circumstance) or the Warrantors and the Holders' Representative (in the case of the Buyer becoming aware of such fact or circumstance).
5.5.Waiver of Conditions. At any time on or before the Completion Date, the Buyer may waive any Condition set out in clauses 5.1 by Notice to the Holders' Representative on any terms it decides, and (i) the Holders' Representative (on behalf of the Represented Holders may waive any Condition set out in clause 5.2 in respect of the Represented Holders, by Notice to the Buyer on any terms the Holders' Representative decides, or (ii) each Unrepresented Holder may waive any Condition set out in clause 5.2 in respect of itself, by Notice to the Buyer on any terms such Unrepresented Holder decides.
5.6.Cure Period. If the Sellers or the Warrantors fail to satisfy, or procure the satisfaction of, any Condition set out in clauses 5.1 or the Buyer fails to satisfy, or procure the satisfaction of, any Condition set out in clause 5.2 (each a "CP Defaulting Party"), in each case by 5.00 p.m. on the Longstop Date, and the Buyer (if any Seller or any Warrantor is a CP Defaulting Party) or the Holders' Representative (on behalf of the Represented Holders) and the Unrepresented Holders (if the Buyer is a CP Defaulting Party) do not waive such Condition, the CP Defaulting Party shall be entitled to remedy such failure and achieve the satisfaction of the relevant Condition within twenty (20) Business Days after the Longstop Date (the "Cure Period"). If, following expiry of the Cure Period, the relevant Condition remains unsatisfied and the Buyer (for any Condition set out in clause 5.1) or the Holders' Representative (on behalf of the Represented Holders) and the Unrepresented Holders (for any Condition set out in clause 5.2) have not waived such Condition before the expiry of the Cure Period, the CP Non-defaulting Party (as defined below) shall have the option (but shall not be required) to terminate this Agreement by giving written notice to the CP Defaulting Party. In the event of such termination, the Company shall indemnify the Buyer (if any Seller or any Warrantor is the CP Defaulting Party) or the Buyer shall indemnify the Company (if the Buyer is the CP Defaulting Party) (the Buyer or the Company (as applicable) who is being indemnified shall be a "CP Non-defaulting Party") for a breach of the obligations of the Buyer, on the one hand, or the Sellers or Warrantors, on the other hand, under clause 5.3, and keep the CP Non-defaulting Party
- 27 - | ||||||||
indemnified, on demand against all external advisor fees and any other fees directly incurred by the CP Non-defaulting Party relating to the transactions contemplated by the Transaction Documents (including the negotiation, preparation, execution and performance by it of this Agreement and of each other Transaction Document), up to the Reimbursement Cap.
5.7.If this Agreement is terminated pursuant to clause 5.6, each party's further rights and obligations cease immediately on termination, but termination does not affect a party's accrued rights and obligations as at the date of termination or its rights and obligations arising as a result of termination, provided that the remedies set forth in clause 5.6 shall be the CP Non-defaulting Party's sole and exclusive remedy for any claim made by CP Non-defaulting Party against the CP Defaulting Party for the termination of this Agreement pursuant to clause 5.6 (unless the termination of this Agreement is as a result of any fraud or wilful misconduct of the CP Defaulting Party).
6.Completion¶
6.1.Completion Date. Subject to the satisfaction or waiver of all Conditions, Completion shall take place via electronic exchange on 1 September, 2025 or such other date as the Buyer and the Sellers may agree in writing (the "Completion Date"). Completion shall be deemed to have occurred and effective as of 12:00am Hong Kong time on the Completion Date.
6.2.Completion Requirements. At Completion, the relevant Sellers, the Warrantors and the Buyer shall each do all those things respectively required of them in Schedule 6 and:
6.2.1the Buyer shall pay the Initial Consideration, less the Adjustment Escrow Amount, less the Indemnity Escrow Amount, less the Tax Indemnity Escrow Amount, and less 50% of the total fees and costs of the Paying Agent and Escrow Agent under the Escrow and Paying Agent Agreement, to the Sellers and Option Holders in accordance with clauses 3.1 and 3.2; and
6.2.2the Buyer shall deposit the Adjustment Escrow Amount into the Adjustment Escrow Account, the Indemnity Escrow Amount into the Indemnity Escrow Account, and the Tax Indemnity Escrow Amount into the Tax Indemnity Escrow Account, in each case, established pursuant to the terms and conditions of the Escrow and Paying Agent Agreement; and
6.2.3the Buyer shall pay the full amount of the fees and costs of Paying Agent and Escrow Agent under the Escrow and Paying Agent Agreement respectively to the Paying Agent and the Escrow Agent.
6.3.No Obligation to Complete by Buyer. The Buyer is not obliged to proceed to Completion unless:
6.3.1the Sellers and Warrantors comply with all their obligations under this clause 6 and Schedule 6; and
- 28 - | ||||||||
6.3.2the sale and purchase of all the Sale Shares is completed simultaneously and, all Sale Shares represent: (a) more than 90% of the share capital of the Company on a fully-diluted basis, (b) more than 90% of all issued and outstanding Investor Ordinary Shares of the Company, (c) more than 90% of all issued and outstanding Existing Shareholder Ordinary Shares of the Company, and (d) more than 90% of all issued and outstanding Other Ordinary Shares of the Company, in each case, immediately prior to Completion.
6.4.No Obligation to Complete by Sellers. The Sellers are not obligated to proceed to Completion unless the Buyer complies with its obligations under this clause 6 and Schedule 6.
6.5.Delivery. All documents and items delivered and payments made in connection with Completion shall be held by the recipient to the order of the person delivering or paying them (as the case may be) until such time as Completion takes place.
6.6.Default. If Completion does not take place on the Completion Date because any Seller, Warrantor or the Buyer (each a "Defaulting Party") fails to comply with any of its obligations under this clause 6 and Schedule 6 (whether or not such failure by such Seller, Warrantor or Buyer would amount to a repudiatory breach at common law), the Buyer (if any Seller or any Warrantor is a Defaulting Party) or the Holders' Representative (on behalf of the Represented Holders) and each Unrepresented Holder (if the Buyer is a Defaulting Party) (in each case, the "Non-defaulting Party") may by Notice to each other parties:
6.6.1proceed to Completion to the extent reasonably practicable;
6.6.2postpone Completion to such date as the Non-defaulting Party may specify (being a date not later than the date on which the Cure Period expires); or
6.6.3terminate this Agreement.
6.7.Postponed Completion Date. If the Non-defaulting Party chooses to postpone Completion to another date in accordance with clause 6.6.2, the provisions of this Agreement apply as if that other date is the relevant Completion Date.
6.8.Termination. If the Non-defaulting Party terminates this Agreement pursuant to clause 6.6.3:
6.8.1the Defaulting Party shall indemnify the Buyer (if any Seller, any Warrantor or the Company is the Defaulting Party) or the Sellers, the Warrantors and the Company (if the Buyer is the Defaulting Party), and keep such party indemnified, on demand against all external advisor fees and any other fees directly incurred by the Non-defaulting Party relating to the transactions contemplated by the Transaction Documents (including the negotiation, preparation, execution and performance by it of this Agreement and of each other Transaction Document), up to the Reimbursement Cap; and
- 29 - | ||||||||
6.8.2each party's further rights and obligations cease immediately on termination and none of the parties shall be liable to the other parties on account of such termination, provided that the remedies set forth in clause 6.8.1 shall be the Non-defaulting Party's sole and exclusive remedy for any claim made by Non-defaulting Party against the Defaulting Party for the termination of this Agreement pursuant to clause 6.6.3 (unless the termination of this Agreement is as a result of any fraud or wilful misconduct of the Defaulting Party).
7.Warranties¶
7.1.Holder Warranties. Each Holder, severally and not jointly, represents and warrants to the Buyer that each Holder Warranty is, solely with respect to himself/herself/itself, true, accurate and not misleading as at the date of this Agreement. Immediately before Completion, each Holder is deemed to, severally and not jointly, represent and warrant to the Buyer that each Holder Warranty is, solely with respect to himself/herself/itself, true, accurate and not misleading by reference to the facts and circumstances as at Completion. For this purpose only, where there is an express or implied reference in a Holder Warranty to the "date of this Agreement", that reference is to be construed as a reference to the Completion Date. Notwithstanding anything to the contrary in this Agreement, each Option Holder shall, severally and not jointly, represent and warrant to the Buyer that each Holder Warranty given by such Option Holder is, solely with respect to himself/herself/itself, true, accurate and not misleading as at the Completion Date only.
7.2.Warrantor Warranties. Save as Disclosed otherwise in the Disclosure Letter, each Warrantor, severally and not jointly, represents and warrants to the Buyer that each Warrantor Warranty is true, accurate and not misleading as at the date of this Agreement. Immediately before Completion and save as Disclosed otherwise in the Disclosure Letter, each Warrantor is deemed to, severally and not jointly, represent and warrant to the Buyer that each Warrantor Warranty is true, accurate and not misleading by reference to the facts and circumstances as at Completion. For this purpose only, where there is an express or implied reference in a Warrantor Warranty to the "date of this Agreement", that reference is to be construed as a reference to the Completion Date.
7.3.Buyer Warranties. The Buyer represents and warrants to the Company and each Holder that each Buyer Warranty is true, accurate and not misleading as at the date of this Agreement. Immediately before Completion, the Buyer is deemed to represent and warrant to the Company and each Holder that each Buyer Warranty is true, accurate and not misleading by reference to the facts and circumstances as at Completion. For this purpose only, where there is an express or implied reference in a Buyer Warranty to the "date of this Agreement", that reference is to be construed as a reference to the Completion Date.
7.4."Disclosed" Defined. Reference to any facts and circumstances being "Disclosed" in this Agreement shall be deemed to be a reference to them being fully, fairly, specifically and accurately disclosed with sufficient details to enable a reasonable buyer to be aware of, identify and determine, the existence, scope and impact of the
- 30 - | ||||||||
relevant fact or circumstances on the business, operations and prospects of the relevant Group Company.
7.5.Warranties Independent. Each Warranty is to be construed independently and (except where this Agreement provides otherwise) is not limited by a provision of this Agreement or another Warranty.
8.Pre-Completion Conduct¶
8.1.Conduct by Group Companies. Between the execution of this Agreement and Completion, the Company shall comply, and the Sellers and Warrantors shall procure that each of the Group Companies complies with, Schedule 10.
8.2.Conduct by Sellers. Between the execution of this Agreement and Completion, no Seller shall:
8.2.1enter into or be involved in any discussion or negotiation with any person except the Buyer in connection with the sale of any Share or any share of any Group Company, the sale or merger of any Group Company or the sale of the business or any part of the business of or the disposal of any of the assets or the business of any Group Company;
8.2.2enter into an agreement or arrangement with any person except the Buyer in connection with the sale of any Share or any share of any Group Company, the sale or merger of any Group Company or the sale of the business or any part of the business of or the disposal of any of the assets of any Group Company; or
8.2.3make available to any person except the Buyer any information relating to the sale of any Share or any share of any Group Company, the sale or merger of any Group Company or the sale of the business or any part of the business of or the disposal of any of the assets of any Group Company.
8.3.Notification. Between the execution of this Agreement and Completion, each Seller shall notify the Buyer immediately if he/she/it or any of his/her/its or his/her/its Affiliates' directors, employees or advisors becomes aware of a matter, fact or circumstance which constitutes or which would or might constitute a breach (whether repudiatory in nature or not) of clauses 7.1 and 7.2 and clause 8.1 or any other provision under this Agreement or which would or might cause a Warranty to be untrue, inaccurate or misleading if given in respect of the matters, facts or circumstances at the relevant time between signing and Completion.
8.4.Reasonable Access. Between the date of this Agreement and the Completion Date, upon reasonable prior notice, the Company shall, and shall cause its and the Group's respective officers, directors and employees, financial advisors, attorneys, accountants, consultants or other advisors, agents and representatives (such persons collectively, "Representatives") to, afford to the Buyer, its Affiliates and their respective Representatives reasonable access without undue interruption during normal business hours, consistent with Applicable Law, to the Group's officers,
- 31 - | ||||||||
employees, properties, offices, other facilities and books and records, and shall furnish the Buyer, its Affiliates and their respective Representatives with all financial, operating and other data and information as they may reasonably request.
9.Indemnities¶
9.1.Indemnification by Holders. Subject to the limitations set out in Schedule 9, the Holders ("Indemnifying Parties", each an "Indemnifying Party") shall, severally and not jointly, pro rata in accordance with their respective Indemnification Percentages, defend, hold harmless and indemnify the Buyer, and keep the Buyer indemnified, on demand against any Losses incurred by the Buyer and/or its Affiliates arising (directly or indirectly):
9.1.1out of any Relevant Claim; and
9.1.2from or related to:
(a)all Taxes (or damages resulting from their non-payment) relating any Group Company, and relating to the period up to and including the Completion Date that have not (i) been paid in full on or before the Completion Date; (ii) been fully covered by provisions in the Accounts that have been taken into account in the purchase price adjustments; or (iii) been fully covered by provisions in the Management Accounts, including, but not limited to:
i.any Liabilities for Taxes (whether incurred or assessed before or after Completion Date) in connection with matters (whether acts, omissions, events or business transactions) occurring up to and including Completion Date;
ii.any Liabilities for Taxes relating to capital, equity, income, profits and gains earned or received up to and including the Completion Date; For the avoidance of doubt, any increase in Tax Liabilities or reduction in Tax savings resulting from any loss, unavailability, or reduction in the amount of any Accounts Relief which would be given as a deduction in computing or as a set off against income, profits or gains, or as a credit against Tax, shall not constitute a Loss under this clause 9, to the extent that such loss, unavailability, or reduction is a result of any action or omission of any Holder, Warrantor or Group Company pursuant to this Agreement or any other Transaction Documents or pursuant to an express written request by the Buyer;
iii.any Liabilities for Taxes of another person as a result of its inclusion in a consolidated, combined or group Tax return up to and including the Completion Date; and
iv.all Taxes arising out of or in connection with any payment to or for the benefit of the Holders under this Agreement, including any
- 32 - | ||||||||
Taxes required to be withheld in connection with any payment to or for the benefit of the Holders under this Agreement to the extent they have not been withheld in accordance with this Agreement; and
(b)any Special Tax Claim.
(c)any claim by any Shareholder asserting non-compliance of or failure to adhere to co-sale rights provisions of the Company’s Memorandum and Articles of Association or the Shareholders’ Agreement.
9.2.Third Party Claims.
9.2.1Post Completion, if the Buyer becomes aware of any claim, action or demand being made by a third party against the Buyer or any Group Company which is likely to give rise to a Relevant Claim against an Indemnifying Party under this Agreement (a "Third Party Claim"), the Buyer shall, as soon as practicable and in any event within thirty (30) days of becoming aware of such Third Party Claim, give a written notice (the "Claim Notice") to the relevant Indemnifying Party of the Third Party Claim, describing in reasonable detail the nature of the Third Party Claim, and subject to any applicable confidentiality obligations and Applicable Law, provide a copy of all papers served with respect to such Third Party Claim (if any) and details on the basis for the Buyer's request for indemnification under this Agreement; provided that, no failure or delay in providing such Claim Notice shall constitute a waiver or otherwise modify the Buyer's right to indemnification hereunder, except that the Buyer shall not be entitled to claim, in respect of such Third Party Claim, any Losses which (1) arise directly out of the Buyer's failure to notify the relevant Indemnifying Party within the stipulated timeframe as set out in this clause 9.2.1 or (2) result in the relevant Indemnifying Party being substantially prejudiced by such failure or delay.
9.2.2Upon receipt of a Claim Notice with respect to a Third Party Claim, the relevant Indemnifying Parties shall, to the maximum extent permitted under Applicable Law, have the right to assume the defence of any Third Party Claim by notifying the Buyer in writing within 30 days of receipt of such Claim Notice, and upon delivery of such notice, such Indemnifying Parties shall have the right to fully control and settle the relevant proceeding, provided that any such settlement or admission of fault shall be permitted hereunder only with the written consent of the Buyer (such consent not to be unreasonably withheld, delayed or conditioned). Notwithstanding the foregoing, the Indemnifying Parties shall not be entitled to assume the defence, contest or control of any Third Party Claim if (i) the Third Party Claim relates to or arises in connection with any criminal action, (ii) the Third Party Claim seeks an injunction or equitable relief against the Buyer, (iii) the Third Party Claim is or would reasonably be expected to result in Losses in excess of the amounts available for indemnification pursuant to this clause 9, or (iv) the Indemnifying Parties have not acknowledged that such Third Party
- 33 - | ||||||||
Claim is subject to indemnification pursuant to this clause 9. If the Indemnifying Parties assume the defence of a Third Party Claim pursuant to this clause 9.2.2, they shall conduct such defence in good faith.
9.2.3If requested by the Indemnifying Parties and subject to clause 9.2.2, the Buyer shall, at the sole cost and expense of the Indemnifying Parties, and subject to the advice of the separate co-counsel of the Buyer (if any), cooperate reasonably with the Indemnifying Parties and its counsel in contesting any Third Party Claim which the Indemnifying Parties elect to contest, including in connection with the making of any related counterclaim against the Third Party asserting the Third Party Claim or any cross complaint against any person. The Buyer shall have the right to receive copies of all pleadings, notices and communications with respect to such Third Party Claim, other than any privileged communications between the Indemnifying Parties and its counsel, and shall be entitled, at its sole cost and expense, to retain separate co-counsel and participate in, but not control, any defence or settlement of any Third Party Claim assumed by the Indemnifying Parties pursuant to clause 9.2.2.
9.2.4In the event of a Third Party Claim for which the Indemnifying Parties elect not to, or are not entitled to, assume the defence, contest or control, or otherwise fail to make such an election within 30 days of receipt of the relevant Claim Notice or otherwise fail to continue the defence, the Buyer may, at its option, defend, settle, compromise, or pay for, such action or claim at the expense of the Indemnifying Parties.
9.2.5The Buyer shall not be precluded by any of the provisions of this clause 9.2 from claiming against the Indemnifying Parties in respect of any Loss suffered by the Buyer as a result of, or in connection with, a Third Party Claim subject to the limitations as set out in this clause 9 and Schedule 9.
9.3.Relevant Claims.
9.3.1If the Buyer has a Relevant Claim against an Indemnifying Party, it shall, as soon as practicable and in any event within thirty (30) days of becoming aware of the Relevant Claim, give a written notice (stating in reasonable detail the nature of the Relevant Claim and, if practicable, an estimate of the amount claimed) (the "Indemnity Notice") to the relevant Indemnifying Party and the Escrow Agent; provided that, no failure or delay in providing such Indemnity Notice shall constitute a waiver or otherwise modify the Buyer's right to indemnification hereunder, except that the Buyer shall not be entitled to claim, in respect of such Relevant Claim, any Losses which (1) arise directly out of the Buyer's failure to notify the relevant Indemnifying Party within the stipulated timeframe as set out in this clause 9.3.1 or (2) result in the relevant Indemnifying Party being substantially prejudiced by such failure or delay. If the Indemnifying Parties do not notify the Buyer within 30 days from its receipt of the Indemnity Notice that the Indemnifying Parties dispute such
- 34 - | ||||||||
Relevant Claim, the Indemnifying Parties shall be deemed to have accepted and agreed with such Relevant Claim.
9.3.2Where an Indemnity Notice is given under clause 9.3.1, without prejudice to the validity of the Relevant Claim or alleged Relevant Claim in question, the Buyer shall allow, and shall procure that the Group Companies allows, the relevant Indemnifying Party to investigate the matter or circumstance alleged to give rise to such Relevant Claim. For such purpose and at such Indemnifying Party's cost, the Buyer shall provide, and shall procure that the relevant Group Company provides, all reasonable information to such Indemnifying Party, including reasonable access to premises and personnel at reasonable office hours, and the right to examine and copy any accounts, documents and records of the Company, all to the extent reasonably requested by the Indemnifying Party's representative and necessary for the latter to assess the matter of the Relevant Claim in question and subject to appropriate arrangements being made to maintain commercial confidentiality and privilege, including without limitation execution of a confidentiality agreement in form satisfactory to the Buyer before any information is shared, and redaction of commercially sensitive or privileged information as solely determined by the Buyer, provided that any such request shall be with at least ten (10) Business Days prior written notice to Buyer and/or relevant Group Company, and shall not in any event disrupt or negatively impact business operation of relevant Group Company.
9.4.Payment of Indemnity Amount; Right to Set-off against Indemnity Escrow Fund.
9.4.1If the amount of Losses with respect to any Relevant Claim is agreed or deemed to be agreed to be owed to the Buyer in accordance with clause 9.3, subject always to the limitations set out in Schedule 9, the Buyer shall be entitled to (i) first, deduct an amount equal to such Losses from the Indemnity Escrow Fund in satisfaction of the Relevant Claim, by delivering a joint written instruction with Prenetics and the Holders' Representative to the Escrow Agent to pay such amount from the Indemnity Escrow Account to the Buyer's Bank Account, pursuant to the terms of the Escrow and Paying Agent Agreement; and (ii) second, if the then available Indemnity Escrow Fund is insufficient to cover the full amount of such Losses or if the Indemnity Escrow Fund has already been released to the Indemnifying Parties pursuant to clause 9.4.2, each Indemnifying Party shall, within ten (10) Business Days following the date the amount of Losses is determined, agreed or deemed agreed to be owed, pay to the Buyer such Indemnifying Party’s Indemnification Percentage of the excess amount; provided that, in case of any Holder Individual Claim, the Buyer shall not have the right to set-off any Losses against the Indemnity Escrow Fund; provided further that, the remaining provisions of this clause 9.4.1 shall apply to a Holder Individual Claim.
9.4.2Within three (3) Business Days following the Expiration Date, the Buyer, Prenetics and the Holders' Representative shall deliver a joint written instruction to the Escrow Agent to release the then available Indemnity
- 35 - | ||||||||
Escrow Fund in full to the Indemnifying Parties in accordance with their respective Indemnification Percentage, pursuant to the terms of the Escrow and Paying Agent Agreement.
9.5.Payment of Special Tax Indemnity Amount; Right to Set-off against Tax Indemnity Escrow Fund.
9.5.1If the amount of Losses with respect to any Special Tax Claim is agreed or deemed to be agreed to be owed to the Buyer in accordance with the same procedures set out in clause 9.3, the Buyer shall be entitled to (i) first, deduct an amount equal to such Losses from the Tax Indemnity Escrow Fund in satisfaction of the Tax Claim, by delivering a joint written instruction with Prenetics and the Holders' Representative to the Escrow Agent to pay such amount from the Tax Indemnity Escrow Account to the Buyer's Bank Account, pursuant to the terms of the Escrow and Paying Agent Agreement; and (ii) second, if the then available Tax Indemnity Escrow Fund is insufficient to cover the full amount of such Losses or if the Tax Indemnity Escrow Fund has already been released to the Indemnifying Parties pursuant to clause 9.5.2, each Indemnifying Party shall, within ten (10) Business Days following the date the amount of Losses is determined, agreed or deemed agreed to be owed, pay to the Buyer such Indemnifying Party’s Indemnification Percentage of the excess amount. Notwithstanding anything to the contrary herein, the Tax Indemnity Escrow Fund shall not be used or drawn to satisfy any Relevant Claim other than a Special Tax Claim.
9.5.2Within three (3) Business Days following the Special Tax Expiration Date, the Buyer, Prenetics and the Holders' Representative shall deliver a joint written instruction to the Escrow Agent to release the then available Tax Indemnity Escrow Fund in full to the Indemnifying Parties in accordance with their respective Indemnification Percentage, pursuant to the terms of the Escrow and Paying Agent Agreement.
10.Termination¶
10.1.Termination before Completion. This Agreement may be terminated prior to the Completion as between the Sellers and the Buyer:
10.1.1by mutual written consent between the Holders' Representative (on behalf of the Represented Holders), each Unrepresented Holder, and the Buyer;
10.1.2by written notice by the CP Non-defaulting Party, in accordance with clause 6.6; or
10.1.3by written notice by the Non-defaulting Party, in accordance with clause 6.6.3.
10.2.Effect of Termination. If this Agreement is terminated pursuant to clause 10.1, this Agreement shall have no further force and effect, except for provisions of clauses 1, 10 14-18, 20-22 which shall survive such termination indefinitely (other than as expressly provided therein), provided that nothing in clause 10.1 or this clause 10.2
- 36 - | ||||||||
shall be deemed to release any party from any Liability for any breach by such party of the terms of this Agreement or to impair the right of any party to compel specific performance by another party of its obligations under this Agreement.
11.Holders' Representative¶
11.1.Appointment. The Represented Holders and the Company hereby acknowledge and agree that, by virtue of the approval of the transactions contemplated by this Agreement by the Represented Holders and without further action of any of the Represented Holders or the Company, [***] is hereby irrevocably appointed as the Holders' Representative and shall have full power and authority, for the benefit of the Represented Holders as the exclusive agent and true and lawful attorney-in-fact to act on behalf of each Represented Holder under this Agreement, in connection with, and to facilitate the consummation of, the transactions contemplated by this Agreement. Such agency and appointment includes the power and authority, as the Holders' Representative may deem necessary or desirable in his sole discretion: (a) to negotiate, execute and deliver such waivers, consents and amendments (other than any written consent of the Represented Holders adopting this Agreement or appointing the Holders' Representative) under this Agreement and the consummation of the transactions contemplated by this Agreement; (b) to refrain from enforcing any right of the Represented Holders arising out of or in any way relating to this Agreement; provided, however, that no such failure to act, except as otherwise expressly provided in this Agreement, shall be deemed a waiver of any such right or interest by the Holders' Representative or the Represented Holders unless such waiver is a writing signed by the Holders' Representative or waiving party; (c) as the Holders' Representative, to enforce and protect the interests and rights of the Represented Holders and the rights and interests of such persons arising out of or in any manner relating to this Agreement and the transactions contemplated by this Agreement, and to take any and all actions under this Agreement for and on behalf of the Represented Holders, including, consenting to, compromising or settling any such claims, negotiating with the Buyer and its representative regarding such claims, and, in connection therewith, to (i) investigate, contest, defend or litigate any claim, action, proceeding or investigation initiated by the Buyer, against the Holders' Representative and/or any of the Represented Holders, and receive process on behalf of any or all Represented Holders therefor and compromise or settle, and give receipts, releases and discharges with respect to, any such claim, action, proceeding or investigation; (ii) assert any claim or institute any action, proceeding or investigation; (iii) settle or compromise any claims asserted under this Agreement; (iv) file any proofs of debt, claims and petitions; and (v) file and prosecute appeals from any decision, judgment or award rendered in any such action, proceeding or investigation, provided that the Holders' Representative shall not have any obligation to take any such actions, and shall not have any Liability for the failure to so take any such action; (d) assert any claim or institute any action, proceeding or investigation; and (e) to make, execute, and deliver all such other agreements, payments, receipts, guarantees, endorsements, notices, requests, instructions, certificates, letters, stock powers, and other writings, and, in general, to do any and all things and to take any and all action that the Holders' Representative, in its sole and absolute discretion, may deem necessary, proper or convenient in connection with or to carry out the transactions contemplated by this
- 37 - | ||||||||
Agreement or otherwise in connection with the subject matter of this Agreement. Notwithstanding the foregoing, the Holders' Representative shall have no obligation to act on behalf of the Represented Holders, except as expressly provided herein, and for purposes of clarity, there are no obligations of the Holders' Representative in any ancillary agreement, schedule, exhibit or the Disclosure Letter. The Holders' Representative may be changed by the holders of a majority in interest of the Represented Holders as determined by the Sale Shares of each Represented Holder upon not less than ten (10) days' prior written notice to all of the Represented Holders and to Buyer. If the Holders' Representative shall resign, a replacement shall be promptly named by the holders of a majority in interest of the Represented Holders as determined by the Sale Shares of each Represented Holder; if the Represented Holders fails to name a replacement of Holders' Representative within 10 days from the resignation, all Represented Holders agree that they shall apply with the court for a court-appointed representative. No bond shall be required of the Holders' Representative.
11.2.Expenses. The Represented Holders shall indemnify, defend and hold harmless the Holders' Representative from and against any and all losses, claims, damages, Liabilities, fees, costs, expenses (including fees, disbursements and costs of counsel and other skilled professionals and in connection with seeking recovery from insurers), judgments, fines or amounts paid in settlement (collectively, the "Holders' Representative Expenses") incurred without gross negligence or wilful misconduct on the part of the Holders' Representative and arising out of or in connection with the acceptance or administration of its duties hereunder. Such Holders' Representative Expenses may be recovered from any distribution or other amounts otherwise distributable to the Represented Holders and/or directly from the Represented Holders. The Represented Holders acknowledge that the Holders' Representative shall not be required to expend or risk its own funds or otherwise incur any financial Liability in the exercise or performance of any of its powers, rights, duties or privileges or pursuant to this Agreement, or the transactions contemplated hereby or thereby. Furthermore, the Holders' Representative shall not be required to take any action unless the Holders' Representative has been provided with funds, security or indemnities which, in its determination, are sufficient to protect the Holders' Representative against the costs, expenses and Liabilities which may be incurred by the Holders' Representative in performing such actions. The Holders' Representative shall be entitled to: (i) rely upon any signature believed by it to be genuine, and (ii) reasonably assume that a signatory has proper authorisation to sign on behalf of the applicable Represented Holder or other party.
11.3.Survival. All of the indemnities and immunities granted to the Holders' Representative under this Agreement shall survive Completion, the resignation or removal of the Holders' Representative, and/or any termination of this Agreement. All of the indemnities, immunities and powers granted to the Holders' Representative under this Agreement (i) are coupled with an interest, shall be irrevocable and shall survive the death, incompetency, bankruptcy or liquidation of any Represented Holder and shall be binding on any successor thereto and (ii) shall survive the consummation of the transactions contemplated by this Agreement. Any action taken by the Holders' Representative pursuant to the authority granted hereunder or otherwise under this
- 38 - | ||||||||
Agreement shall be effective and absolutely binding on each Represented Holder and such Represented Holder's successors as if expressly confirmed and ratified in writing by such Represented Holder notwithstanding any contrary action of or direction from such Represented Holder, and all defences which may be available to any Represented Holder to contest, negate or disaffirm the action of the Holders' Representative taken in good faith under this Agreement are waived.
11.4.Reliance by Buyer. The Buyer shall have the right to rely upon all actions taken or omitted to be taken by the Holders' Representative pursuant to this Agreement, all of which actions or omissions shall be legally binding upon the Represented Holders, and the Buyer shall have no obligation to monitor or supervise the performance of the obligations of the Holders' Representative. The Buyer shall not be liable to any of the Represented Holders for any action taken or omitted to be taken by the Holders' Representative.
11.5.Power of Attorney. With respect to the Escrow and Paying Agent Agreement only, each of the Unrepresented Holders irrevocably and unconditionally appoints the Holders' Representative as its exclusive agent and true and lawful attorney-in-fact attorney to negotiate, execute and deliver the Escrow and Paying Agent Agreement for and on behalf of such Unrepresented Holder, and to take any and all actions under the Escrow and Paying Agent Agreement for and on behalf of the Unrepresented Holders.
12.Undertakings by the Sellers¶
12.1.Non-Disparagement. Each Holder undertakes to the Buyer, acting for himself/herself/itself that he/she/it will not, and will procure that none of its Affiliates will, do any of the following things, whether directly or indirectly and whether alone or jointly with, or on behalf of, or as agent for, any other person or as shareholder, partner, director, principal, consultant, officer, agent, employee or otherwise, for a period of two (2) years starting on the date of this Agreement, do or say anything which is harmful to a Group Company's goodwill (subsisting as at the date of this Agreement) or which would reasonably be expected to lead a person who has dealt with a Group Company at any time during the twelve (12) months prior to the date of this Agreement to cease to deal with a Group Company on substantially equivalent terms to those previously offered or at all.
12.2.Non-Competition; Non-Solicitation.
Each Holder (other than any Financial Investor Seller and any Option Holder) (each a "Covenantor") undertakes to the Buyer, acting for himself/herself/itself that he/she/it will not, and will procure that none of its Affiliates will, do any of the following things, whether directly or indirectly and whether alone or jointly with, or on behalf of, or as agent for, any other person or as shareholder, partner, director, principal, consultant, officer, agent, employee or otherwise:
12.2.1for a period of one (1) year starting on the date of this Agreement, carry on or be engaged, concerned or interested in or assist a business which competes, directly or indirectly, with the Business of the Group as carried on as at the
- 39 - | ||||||||
date of this Agreement in a territory in which such Business is carried (a "Competing Business"); provided that, this clause 12.2.1 shall not prohibit or otherwise restrict any Covenantor from carrying on or being engaged, concerned or interested in or assisting an ongoing Competing Business of such Covenantor prior to the date of this Agreement (including, in the case of Prenetics, its interests in CircleDNA and Insighta).
12.2.2for a period of two (2) years starting on the date of this Agreement, in respect of the products or services of the Business of the Group, either seek to obtain orders from, or do business with, or encourage another person to obtain orders from or do business with, a person who has been a customer of the Group at any time during the twelve (12) months prior to the date of this Agreement for the products or services of such Business in its territory of operation;
12.2.3for a period of two (2) years starting on the date of this Agreement, solicit or contact with a view to his/her engagement or employment by another person, a director, officer, employee or manager of a Group Company or a person who was a director, officer, employee or manager of a Group Company at any time during the twelve (12) months prior to the date of this Agreement, in either case where the person in question either has any Protected Information or would be in a position to exploit a Group Company's trade connections, except where such solicitation or contact results from general job postings or public job advertisements; and
12.2.4for a period of two (2) years starting on the date of this Agreement, engage or employ a director, officer, employee or manager of a Group Company or a person who was a director, officer, employee or manager a Group Company at any time during the twelve (12) months prior to the date of this Agreement, in either case where the person in question either has any Protected Information or would be in a position to exploit a Group Company's trade connections, except where such engagement or employment results from general job postings or public job advertisements.
12.3.Release. From and after Completion, each Holder, on behalf of itself and its Affiliates, predecessors, successors, assigns, heirs, executors, legatees, administrators, beneficiaries and Representatives (the "Seller Releasing Parties"), fully, finally and irrevocably release, acquit and forever discharge the Group Companies and each of their respective Affiliates, predecessors, successors, assigns, heirs, executors, legatees, administrators, beneficiaries and Representatives (collectively, the "Seller Released Parties") from any and all commitments, Actions, claims, losses, Liabilities and costs and expenses of every kind and nature whatsoever, whether arising from any express, implied, oral or written agreement, contract, arrangement, commitment or otherwise, known or unknown, past, present or future, at law or in equity, contingent or otherwise, except those arising out of or in connection with this Agreement or any other Transaction Document (collectively, a "Potential Claim"), that such Seller Releasing Parties, or any of them, had, has or may have in the future against the Seller Released Parties, or any of them, for any matter, cause or thing relating to the Group
- 40 - | ||||||||
Companies and occurring at any time prior to Completion (the "Seller Released Matters").
12.3.1Each Holder irrevocably covenants to refrain from, and to cause any of the Seller Releasing Parties to refrain from, directly or indirectly asserting any Potential Claim or commencing, instituting or causing to be commenced, any Action of any kind against any of the Seller Released Parties, in any forum whatsoever (including any administrative agency), with respect to any of the Seller Released Matters.
12.3.2This Clause 12.3 may be pleaded by the Seller Released Parties as a full and complete defence and may be used as the basis for any injunction against any Action instituted or maintained against them in violation of clause 12.3. Each Holder waives any requirement of the posting of any bond in connection with such injunction. If any Potential Claim is brought or maintained by any Holder or any Seller Releasing Party against the Seller Released Parties in violation of clause 12.3, such Holder or the applicable Seller Releasing Party will be responsible for all costs and expenses, including reasonable attorneys' fees, incurred by the Seller Released Parties in defending the same.
12.4.Interpretation. For the avoidance of doubt, all references to "Group Company", "Group Companies" or "Group" in this clause 12 shall not include the Buyer or its Affiliate before or after the Completion.
13.Further Undertakings
14.Confidential Information¶
14.1.Protected Information. Each party undertakes to the other parties, that before and after Completion such party shall:
14.1.1not use or disclose to any person Protected Information he/she/it has or acquires;
14.1.2make every effort to erase from any computer under the control of any party any document, disk or file containing, reflecting or generated from any Protected Information, and following such erasure, not attempt to recover such material;
14.1.3make every effort to prevent the use or disclosure of Protected Information; and
14.1.4procure that each of his/her/its Affiliates complies with sub-clauses 14.1.1 to 14.1.3.
14.2.Permitted Disclosure. Clause 14.1 does not apply to disclosure of Protected Information:
14.2.1to the extent that it is generally known to the public not as a result of a breach of any duty of confidentiality;
- 41 - | ||||||||
14.2.2to a director, officer or employee of the Buyer or of a Group Company whose function requires him to have the Protected Information;
14.2.3to the extent that it is required to be disclosed by Applicable Law, by a rule of a listing authority by which any party's shares or shares of such party's holding company are listed, by a stock exchange on which such party's shares or shares of such party's holding company are listed or traded, or by a Government Authority or other authority with relevant powers to which the party disclosing the Protected Information is subject or submits, whether or not the requirement has the force of law provided that the disclosure shall so far as is practicable be made after consultation with the Buyer (if the disclosing party is any Holder) or the Holders (if the disclosing party is the Buyer) and after allowing such party the opportunity to contest such disclosure and after taking into account such party's reasonable requirements as to its timing, content and manner of making or despatch; or
14.2.4to an adviser for the purpose of advising such party in connection with the transactions contemplated by this Agreement and the Transaction Documents, provided that such disclosure is essential for these purposes and is on the basis that clause 14.1 applies to the disclosure by the adviser.
15.Announcements¶
15.1.Announcement. Subject to clause 15.2, no party may, before or after Completion, make, issue or send a public announcement, communication or circular concerning the transactions referred to in this Agreement and any other Transaction Document unless he/she/it has first obtained the other parties' prior written consent.
15.2.Permitted Announcement. Clause 15.1 does not apply to a public announcement, communication or circular, required by Applicable Law, by a rule of a listing authority by which the Buyer's or Holder's shares or shares of the Buyer's or Holder's holding company are listed, by a stock exchange on which the Buyer's or Holder's shares or shares of the Buyer's or Holder's holding company are listed or traded or by a Government Authority or other authority with relevant powers to which the Buyer or Holder or the Buyer's or Holder's holding company is subject or submits, whether or not the requirement has the force of law, provided that the public announcement, communication or circular shall so far as is practicable be made after consultation with the Buyer and after taking into account the reasonable requirements of the Buyer as to its timing, content and manner of making or despatch.
16.Costs¶
16.1.Costs and Expenses. Except where this Agreement or the relevant Transaction Document provides otherwise, each party shall pay its own fees, costs, expenses and Taxes relating to the negotiation, preparation, execution and performance by it of this Agreement and the other Transaction Documents.
- 42 - | ||||||||
16.2.Finder's Fee. In the event any party incurs any finder's fees (if any) in connection with the transactions contemplated under this Agreement and the other Transaction Documents, such party shall be solely responsible for any such fees.
16.3.Holders' Expenses Allocation. All costs and expenses incurred by the Holders in connection with the transactions contemplated under this Agreement, including but not limited to legal, accounting, advisory, and regulatory filing fees (the "Holders' Expenses") shall be borne by Prenetics, and each other Holder shall pay Prenetics its Pro-Rata Share of the Holders' Expenses. The parties hereby agree that such Holders' Expenses may be recovered by Prenetics from any distribution or other amounts otherwise distributable or payable to the other Holders pursuant to this Agreement and/or directly from the other Holders.
17.General¶
17.1.The failure to exercise or delay in exercising a right or remedy provided by this Agreement or by law does not impair or constitute a waiver of the right or remedy or an impairment of or a waiver of other rights or remedies. No single or partial exercise of a right or remedy provided by this Agreement or by law prevents further exercise of the right or remedy or the exercise of another right or remedy.
17.2.Except with respect to fraud, wilful misconduct or gross negligence, the remedies provided in this Agreement shall be the parties' sole and exclusive remedy for any and all breaches of the representations, warranties and/or covenants contained herein or in any certificate delivered pursuant to this Agreement or any claims arising relating to or arising from or in connection with this Agreement, the transactions contemplated by this Agreement or any other certificate delivered pursuant hereto, in all cases, except for claims based on fraud, wilful misconduct or gross negligence.
17.3.Except to the extent that they have been performed and except where this Agreement provides otherwise, the obligations contained in this Agreement remain in force after Completion.
17.4.If a party fails to pay a sum due from it under this Agreement on the due date of payment in accordance with the provisions of this Agreement, that party shall pay interest on the overdue sum from the due date of payment until the date on which its obligation to pay the sum is discharged at the rate per annum of one per cent (1%). Interest accrues and is payable from day to day.
17.5.All payments made by any Holder under this Agreement shall be made gross, free of right of counterclaim or set-off and without deduction or withholding of any kind other than any deductions or withholding required by Applicable Law.
17.6.If a party makes a deduction or withholding required by Applicable Law from a payment made pursuant to this Agreement for a breach of the Agreement or any payment by way of indemnity or of a compensatory nature under this Agreement, the sum due from the payer shall be increased to the extent necessary to ensure that, after the making of any deduction or withholding, the recipient receives a sum equal to the sum it would have received had no deduction or withholding been made.
- 43 - | ||||||||
17.7.If a payment made pursuant to this Agreement for a breach of the Agreement or any payment by way of indemnity or of a compensatory nature under this Agreement will be or has been subject to Tax in the hands of the payee (or would have been subject to Tax in the hands of the payee but for the availability of Relief), the payer shall pay to the recipient the amount (after taking into account Tax payable in respect of the amount or that would be payable but for the availability of Relief) that will ensure that the recipient receives and retains a net sum equal to the sum it would have received had the payment not been subject to Tax (or not been subject to Tax disregarding the availability of Relief).
17.8.Except as provided in clause 18.4, a person who is not a party to this Agreement has no right under the Third Parties Ordinance to enforce any term of this Agreement, but this does not affect any right or remedy of a third party which exists or is available apart from the Third Parties Ordinance.
17.9.Each of the parties agrees to perform (or procure the performance of) all such acts and things and/or to execute and deliver (or procure the execution and delivery of) all such documents, as may be required by Applicable Law or as may be necessary or reasonably requested by the Buyer for giving full effect to the Transaction Documents and securing to the Buyer the full benefit of the rights, powers and remedies conferred upon the Buyer by the Transaction Documents. Unless otherwise agreed, each party shall be responsible for its own costs and expenses incurred in connection with the provisions of this clause 17.9.
17.10.If, at any time, any provision of this Agreement is or becomes void, illegal, invalid or unenforceable in any respect, whether pursuant to any judgment or otherwise:
17.10.1that voidness, illegality, invalidity or unenforceability shall not affect the legality, validity or enforceability of any other provision of this Agreement; and
17.10.2the void, illegal, invalid or unenforceable provision shall be deemed never to have been a part of this Agreement.
18.Entire Agreement¶
In this clause 18, "Representation" means an assurance, commitment, condition, covenant, guarantee, indemnity, representation, statement, undertaking or warranty of any sort whatsoever (whether contractual or otherwise, oral or in writing, or made negligently or otherwise).
18.1.The Transaction Documents constitute the entire agreement between the parties. They supersede any previous agreements relating to the subject matter of the Transaction Documents, and set out the complete legal relationship of the parties arising from or connected with that subject matter.
18.2.Accordingly, the Buyer:
18.2.1represents and agrees that:
- 44 - | ||||||||
(a)no Holder or adviser to the Holders has made any Representation that the Buyer considers material which is not set out in the Transaction Documents; and
(b)it has not entered into the Transaction Documents in reliance on any Representation except those set out in the Transaction Documents,
and will not contend to the contrary; and
18.2.2for the avoidance of doubt, agrees that:
(a)no adviser to the Holders has any Liability to the Buyer for any Representation except set out where third parties have given opinions etc.;
(b)the Holders have no Liability of any kind to the Buyer for any Representation except in respect of those set out in the Transaction Documents; and
(c)its only rights and remedies in respect of any Representations are those rights and remedies set out in the Transaction Documents.
18.3.Likewise, each Holder:
18.3.1represents and agrees that:
(a)no adviser to the Buyer or provider of finance to the Buyer has made any Representation that such Holder considers material which is not set out in the Transaction Documents, and
(b)it has not entered into the Transaction Documents in reliance on any Representation except those set out in the Transaction Documents,
and will not contend to the contrary; and
18.3.2for the avoidance of doubt, agrees that:
(a)no adviser to the Buyer or provider of finance to the Buyer has any Liability to any Holder for any Representation except set out where third parties have given opinions etc.;
(b)the Buyer has no Liability of any kind to any Holder for any Representation except in respect of those set out in the Transaction Documents; and
(c)its only rights and remedies in respect of any Representations are those rights and remedies set out in the Transaction Documents.
- 45 - | ||||||||
18.4.Each Affiliate of the Buyer or any Holder, the advisers to the Buyer, the advisers to any Holder and providers of finance to the Buyer may enforce the terms of this clause 18 subject to and in accordance with the provisions of the Third Parties Ordinance.
18.5.Nothing in this clause 18 shall have the effect of limiting any Liability arising from fraud.
19.Assignment, Amendment and Modification¶
19.1.This Agreement is personal to each Holder. Accordingly, no Holder shall assign, transfer, declare a trust of the benefit of or in any other way alienate any of its rights under this Agreement whether in whole or in part.
19.2.Each Holder agrees that the benefit of every provision in this Agreement is given to the Buyer for itself and its successors in title and assigns. Accordingly, the Buyer (and its successors and assigns) may, without the consent of the Holders, assign the benefit of all or any of the Holders' obligations under this Agreement and/or any other benefit arising under or out of this Agreement.
19.3.This Agreement may not be amended, restated or otherwise modified except by a written instrument specifically referring to this Agreement that is executed by the Company, the Buyer and the Holders.
20.Notices¶
20.1.A notice or other communication under or in connection with this Agreement (a "Notice") shall be:
20.1.1in writing;
20.1.2in English; and
20.1.3delivered personally or sent by courier by an internationally recognised courier company (e.g. FedEx, DHL) or by email, to the party due to receive the Notice at its address set out in clause 20.3 or to such other address, person or email address as the party may specify by not less than seven (7) days' written notice to the other party received before the Notice was despatched.
20.2.In the absence of evidence of earlier receipt, a Notice shall be deemed to have been duly given if:
20.2.1delivered personally, when left at the address referred to in sub-clause 20.1.3;
20.2.2sent by courier, two (2) Business Days after posting it; and
20.2.3sent by email, when the email is sent.
20.3.The address referred to in sub-clause 20.1.3 is:
- 46 - | ||||||||
Name of party | Address | Email address | Marked for the attention of | |||||||||||
a Seller | As set out in column (A) in Part A of Schedule 1 opposite the name of such Seller | As set out in column (A) in Part A of Schedule 1 opposite the name of such Seller | As set out in column (A) in Part A of Schedule 1 opposite the name of such Seller | |||||||||||
an Option Holder | As set out in column (A) in Part B of Schedule 1 opposite the name of such Option Holder | As set out in column (A) in Part B of Schedule 1 opposite the name of such Option Holder | As set out in column (A) in Part B of Schedule 1 opposite the name of such Option Holder | |||||||||||
Holders' Representative | c/o the Company, [***] | [***] | [***] | |||||||||||
The Company | [***] | [***] | [***] | |||||||||||
The Buyer | [***] | [***] [***] | [***]; [***]. | |||||||||||
20.4.Each Represented Holder agrees and acknowledges that any Notice duly given under this clause 20 to the Holders' Representative shall be deemed to be given to all of the Represented Holders.
21.Governing Law¶
21.1.This Agreement is governed by, and shall be construed in accordance with, the laws of Hong Kong.
22.Dispute Resolution¶
22.1.Any dispute, controversy or claim arising in any way out of or in connection with this Agreement (including: (1) any issue regarding contractual, pre-contractual or non-contractual rights, obligations or Liabilities; and (2) any issue as to the existence, validity, breach or termination of this Agreement) (a "Dispute") shall be referred to and finally resolved by binding arbitration administered by the Hong Kong International Arbitration Centre ("HKIAC") in accordance with the HKIAC Administered Arbitration Rules in force when the Notice of Arbitration is submitted in accordance with such Rules (the "Rules"). The Rules are deemed to be incorporated by reference into this clause and as may be amended by the rest of this clause.
22.2.The arbitration shall be conducted as follows:
- 47 - | ||||||||
22.2.1The arbitration tribunal ("Tribunal") shall consist of three (3) arbitrators. The claimant(s) and the respondent(s) shall each nominate one (1) arbitrator, and the two (2) arbitrators thus appointed shall nominate the third arbitrator who shall be the presiding arbitrator; if the claimant(s) or the respondent(s) fails to appoint the arbitrator within fourteen (14) days of a request to do so from the other party(ies), or if the two (2) arbitrators fail to nominate the third arbitrator within fourteen (14) days after the appointment of the second arbitrator, the appointment shall be made, upon request of a party, by the HKIAC in accordance with the Rules.
22.2.2The seat of the arbitration shall be Hong Kong. This clause 22 shall be governed by the laws of Hong Kong.
22.2.3The language of the arbitration proceedings shall be English.
22.2.4Any award of the Tribunal shall be made in writing and shall be final and binding on the parties from the day it is made. The parties undertake to carry out any award without delay.
22.2.5The parties waive any right to apply to any court of law and/or other judicial authority to determine any preliminary point of law and/or review any question of law and/or the merits, insofar as such waiver may validly be made. The parties shall not be deemed, however, to have waived any right to challenge any award on the ground that the Tribunal lacked substantive jurisdiction and/or on the ground of serious irregularity affecting the Tribunal, the proceedings or the award to the extent allowed by the law of the seat of arbitration. Nothing in this clause 22 shall be construed as preventing any party from seeking conservatory or interim relief from any court of competent jurisdiction.
23.Counterparts¶
This Agreement may be executed in any number of counterparts, each of which when executed and delivered is an original and all of which together evidence the same agreement.
- 48 - | ||||||||
Schedule 1
SELLERS AND OPTION HOLDERS
SELLERS AND OPTION HOLDERS
Part A: Sellers
[***]
Part B: Option Holders
[***]
Part C: Unrepresented Holders
[***]
- 49 - | ||||||||
Schedule 2
INFORMATION ABOUT THE COMPANY
AND THE SUBSIDIARIES
INFORMATION ABOUT THE COMPANY
AND THE SUBSIDIARIES
Part A
THE COMPANY
THE COMPANY
Name of company and registered number | ACT Genomics Holdings Company Limited (No. MC-336039) | ||||
Place of incorporation | Cayman Islands | ||||
Company Type | Exempted Company with Limited Liability | ||||
Date of incorporation | 20 April 2018 | ||||
Directors | 1.YEUNG Danny Sheng Wu 2.LO Hoi Chun, Stephen 3.[***] 4.[***] 5.[***] | ||||
Company registered office provider | Maples Corporate Services Limited | ||||
Registered Office | PO Box 309, Ugland House, Grand Cayman, KY1-1104, Cayman Islands | ||||
Issued Share Capital | ●[***] | ||||
Shareholders | Prenetics (73.3%) | ||||
Accounting Reference Date | 31 December | ||||
Part B
THE SUBSIDIARIES
THE SUBSIDIARIES
- 50 - | ||||||||
Name of company and registered number | ACT Genomics Co., Ltd. 行動基因生技股份有限公司 (No. 54650959) | ||||
Place of incorporation | Taiwan (R.O.C.) | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 25 November 2013 | ||||
Directors / Representatives | 1.[***] 2.[***] 3.YEUNG Danny Sheng Wu | ||||
Supervisor | [***] | ||||
Company secretary | N/A | ||||
Registered Office | 3F., No. 345, Xinhu 2nd. Rd., Neihu Dist., Taipei, Taiwan 台北市內湖區新湖二路345號3樓 | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 51 - | ||||||||
Name of company and registered number | ACT Genomics (Hong Kong) Limited 行動基因(香港)股份有限公司 (No. 2243121) | ||||
Place of incorporation | Hong Kong | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 28 May 2015 | ||||
Directors | 1.[***] 2.[***] 3. YEUNG Danny Sheng Wu | ||||
Company secretary | [***] | ||||
Registered Office | Unit 803-807, 8/F, Building 15W, No. 15 Science Park West Avenue, Hong Kong Science Park, Pak Shek Kok, N.T., Hong Kong | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 52 - | ||||||||
Name of company and registered number | ACT Genomics (Singapore) Pte. Ltd. 行動基因(新加坡)私人有限公司 (No. 201536840C) | ||||
Place of incorporation | Singapore | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 6 October 2015 | ||||
Directors | 1.[***] 2. [***] 3. YEUNG Danny Sheng Wu 4. [***] | ||||
Company secretary | [***] | ||||
Registered Office | 1 Phillip Street, Royal One Philip, Singapore 048692 | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 53 - | ||||||||
Name of company and registered number | ACT Genomics (Japan) K.K. アクトゲノミクス日本株式会社公司 (No. 0104-01-130615) | ||||
Place of incorporation | Japan | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 29 March 2017 | ||||
Directors | 1. YEUNG Danny Sheng Wu 2. [***] 3.[***] (Director change in progress) | ||||
Company secretary | N/A | ||||
Registered Office | TTD Building, No. 18, Sanada Ichome, Minato-ku, Tokyo 东京都港区三田一丁目2番18 号TTD ビル | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 54 - | ||||||||
Name of company and registered number | ACT Genomics (IP) Limited 行動基因(智財)有限公司 (No. 2839434) | ||||
Place of incorporation | Hong Kong | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 11 June 2019 | ||||
Directors | 1.[***] 2.[***] 3.YEUNG Danny Sheng Wu | ||||
Company secretary | [***] | ||||
Registered Office | Unit 10, 9/F, Island Place Tower, Island Place, 510 King’s Road, North Point, Hong Kong | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
Name of company and registered number | ACT Genomics HK Lab Limited (No. 2848931) | ||||
Place of incorporation | Hong Kong | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 5 July 2019 | ||||
Directors | 1.YEUNG Danny Sheng Wu 2.TZANG Lawrence 3.[***] | ||||
Company secretary | [***] | ||||
Registered Office | Unit 10, 9/F, Island Place Tower, Island Place, 510 King’s Road, North Point, Hong Kong | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 55 - | ||||||||
Name of company and registered number | MC Diagnostics Limited (No. 5843597) | ||||
Place of incorporation | England and Wales | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 12 June 2006 | ||||
Directors | 1.YEUNG Danny Sheng Wu 2.[***] 3.[***] 4.[***] 5.[***] | ||||
Company secretary | N/A | ||||
Registered Office | 1 Hawkshead Rd Croft Technology Park, Bromborough, Wirral, United Kingdom, CH62 3RJ | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
Name of company and registered number | AM Robotics 2018 Ltd (No. 11173912) (in the process of being struck off in accordance with Applicable Law) | ||||
Place of incorporation | England and Wales | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 29 January 2018 | ||||
Directors | 1.[***] 2.[***] | ||||
Company secretary | N/A | ||||
Registered Office | 1 Hawkshead Rd Croft Technology Park, Bromborough, Wirral, England, CH62 3RJ | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 56 - | ||||||||
Name of company and registered number | Sanomics Holdings Limited (No. 1972173) | ||||
Place of incorporation | British Virgin Islands | ||||
Company Type | British Virgin Islands Business Company Limited by Shares | ||||
Date of incorporation | 6 March 2018 | ||||
Directors | 1.[***] 2.YEUNG Danny Sheng Wu 3.[***] | ||||
Company registered office provider | Vistra Corporate Services | ||||
Registered Office | Vistra Corporate Services Centre, Wickhams Cay II, Road Town, Tortola, VG1110, British Virgin Islands | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 57 - | ||||||||
Name of company and registered number | Sanomics Limited善覓有限公司 (No. 2206533) | ||||
Place of incorporation | Hong Kong | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 26 February 2015 | ||||
Directors | 1.[***] 2.YEUNG Danny Sheng Wu 3.[***] 4.TZANG Lawrence | ||||
Company secretary | [***] | ||||
Registered Office | 12 Unit 306, 3rd Floor, Science Park West Avenue, Phase 3, NT Hong Kong Special Administrative Region of the People’s Republic of China | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 58 - | ||||||||
Name of company and registered number | ACT Genomics (Thailand) Limited (No. 0105561133747) | ||||
Place of incorporation | Thailand | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 6 August 2018 | ||||
Directors | 1.[***] 2.YEUNG Danny Sheng Wu 3.[***] 4.[***] | ||||
Company secretary | N/A | ||||
Registered Office | No.4, 4/5 Unit 1503, Unit 1509, Central Tower, 15/F, Ratchadamri Rd, Pathum Wan, Bangkok 10330, Thailand | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
Name of company and registered number | ACT Medical Instrument Co., Ltd.行動基因醫療器材股份有限公司 (No. 90874222) | ||||
Place of incorporation | Taiwan (R.O.C.) | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 12 April 2021 | ||||
Directors | [***] | ||||
Company secretary | N/A | ||||
Registered Office | 10F.-1, No. 6, Ln. 180, Sec. 6, Minquan E. Rd., Neihu Dist., Taipei City 114, Taiwan (R.O.C.) 台北市內湖區民權東路6段180巷6號10樓之1 | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 59 - | ||||||||
Name of company and registered number | CERBACT ASIA HOLDINGS PTE. LTD. (No. 202124327Z) | ||||
Place of incorporation | Singapore | ||||
Company Type | Private Company Limited by Shares | ||||
Date of incorporation | 12 July 2021 | ||||
Directors | [***] [***] [***] [***] [***] [***] | ||||
Company secretary | [***] [***] | ||||
Registered Office | 380 Jalan Besar #06-06 ARC 380 Singapore (209000) | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 60 - | ||||||||
Name of company and registered number | Cerbact Asia Holdings Pte. Ltd. Taiwan Branch 新加坡商識動亞洲生技股份有限公司台灣分公司 (No. 90430831) | ||||
Place of incorporation | Taiwan | ||||
Company Type | Branch Company | ||||
Date of incorporation | 29 November 2021 | ||||
Directors | [***] | ||||
Branch manager | [***] | ||||
Company secretary | N/A | ||||
Registered Office | 6F, NO 345, Xinhu 2nd Road, Neihu Dist., Taipei Taiwan 台北市內湖區新湖二路345號6樓 | ||||
Issued Share Capital | [***] | ||||
Shareholders | [***] | ||||
Accounting Reference Date | 31 December | ||||
- 61 - | ||||||||
Schedule 3
DETERMINATION OF COMPLETION STATEMENTS
DETERMINATION OF COMPLETION STATEMENTS
1.For the purposes of this Schedule 3, each Holder agrees and acknowledges that the Holders' Representative shall have full power and authority, for the benefit of the Holders as the exclusive agent and true and lawful attorney-in-fact to act on behalf of each Holder under this Schedule 3.
2.As soon as reasonably practicable after the Completion Date, the Holders' Representative shall, on behalf of all Holders, prepare the Group's consolidated profit and loss statement, balance sheet and cashflow statement as of the Completion Date (which, for the avoidance of doubt, shall include all the line items set out in Part A of Schedule 4) (the "Draft Completion Accounts") and the Holders' Representative shall, on behalf of the Holders and at the cost of the Holders, engage KPMG to review and audit the Draft Completion Accounts in accordance with the provisions of this Schedule 3 and on the basis of the Accounting Policies. Each of the Buyer and the Company agrees to furnish or cause to be furnished to the Holders' Representative and/or KPMG, as soon as practicable, all such information and assistance (including access to books and records of the Group Companies) as may be reasonably requested by the Holders' Representative and/or KPMG in connection with the preparation, review and/or audit of the Draft Completion Accounts. The Holders' Representative shall use its/his/her commercially reasonable efforts to procure that KPMG shall complete its audit of the Draft Completion Accounts no later than sixty (60) Business Days after its engagement (such audited Draft Completion Accounts, the "Audited Completion Accounts").
3.The Holders' Representative shall, within five (5) Business Days after the receipt of the Audited Completion Accounts, prepare and deliver to the Buyer the completion statement substantially in the form as set out in Part B of Schedule 4 (the "Draft Completion Statement"), together with the Audited Completion Accounts.
4.The Buyer shall have thirty (30) Business Days after the receipt of the Audited Completion Accounts and the Draft Completion Statement to challenge any amounts set out in the Draft Completion Statement by giving a written notice (the "Buyer Notice") to the Holders' Representative, setting out the reasons for the disagreement in reasonable detail including each disputed item, the amount in dispute and the basis for such dispute. If the Buyer does not deliver a Buyer Notice within the prescribed timeline in accordance with this paragraph 4 of this Schedule 3, the Draft Completion Statement shall be final and binding on the parties all purposes.
5.If the Buyer gives a Buyer Notice under paragraph 4 of this Schedule 3, the Buyer and the Holders' Representative shall attempt in good faith to reach agreement in respect of the matters set out in the Buyer Notice (and, if such agreement is reached, the Draft Completion Statement as amended pursuant to such agreement shall be final and binding on the parties for all purposes). If the Buyer and the Holders' Representative are unable to agree on one or more the matters set out in the Buyer Notice within ten (10) days after the date of the Buyer Notice, either the Buyer or the Holders' Representative may, by written notice to the other, require that the Draft Completion Statement be referred to the Reporting Accountants.
- 62 - | ||||||||
6.The Reporting Accountants shall be engaged by the Buyer and the fees, costs and expenses for such appointment shall be borne by the Buyer.
7.Except to the extent that the Buyer and the Holders' Representative agree otherwise, the Reporting Accountants shall determine their own procedure but:
7.1.apart from procedural matters and as otherwise set out in this Agreement, they shall determine only:
7.1.1.whether any of the arguments for an alteration to the Draft Completion Statement put forward in respect of matters specified in the Buyer Notice is correct in whole or in part (unless such matters have been agreed between the Holders' Representative and the Buyer); and
7.1.2.if so, what alterations (if any) should be made to the Draft Completion Statement;
7.2.in making their determination, they shall refer to the Audited Completion Accounts and apply the Accounting Policies;
7.3.they shall make their determination pursuant to paragraph 7.1 of this Schedule 3 as soon as is reasonably practicable and in any event no later than fifteen (15) Business Days after their engagement;
7.4.the procedure of the Reporting Accountants shall:
7.4.1.give the Buyer and the Holders' Representative a reasonable opportunity to make oral representations and representations in writing to them;
7.4.2.require that each party supplies the other with a copy of any representations in writing at the same time as they are made to the Reporting Accountants; and
7.4.3.permit each party to be present while oral submissions are being made by the other party;
8.The Reporting Accountants shall act as experts and not as arbitrators and their determination of any matter falling within their jurisdiction shall be final and binding on the parties, save in the event of fraud of the Buyer, the Sellers or the Reporting Accountants or manifest error of the Reporting Accountants (in which event the relevant part of their determination shall be void). In particular, without limitation:
8.1.their determination of an amount for any matter shall be an amount within the range demarcated by the lowest and highest limits of the positions specified by the Buyer and the Holders' Representative;
8.2.their determination shall be deemed to be incorporated into the Draft Completion Statement, which shall then be final and binding on the parties for the purposes of this Schedule 3, save as stated above in the event of fraud or manifest error; and
- 63 - | ||||||||
8.3.their determination of any fact which they have found it necessary to determine pursuant to paragraph 7 of this Schedule 3 shall be final and binding on the parties for the purposes of this Schedule 3.
9.The parties shall co-operate with the Reporting Accountants and comply with their reasonable requests made in connection with the carrying out of their duties pursuant to their engagement under the terms of this Agreement.
10.Subject to paragraph 11 of this Schedule 3, nothing in this Schedule 3 shall entitle a party or the Reporting Accountants to access to any information or document which is protected by legal professional privilege, or which has been prepared by the other party or its accountants or other professional advisers with a view to assessing the merits of any claim or argument.
11.A party shall not be entitled by reason of paragraph 10 of this Schedule 3 to refuse to supply such part or parts of documents as contain only the facts on which the Relevant Claim or argument is based.
12.Each party and the Reporting Accountants shall, and shall procure that its accountants and other advisers shall, keep all information and documents provided to them pursuant to this Schedule 3 confidential and shall not use them for any purpose, except for disclosure or use in connection with the preparation and/or review of the Draft Completion Accounts or the Draft Completion Statement, and/or the agreement or determination of the Draft Completion Statement, as applicable, the proceedings of the Reporting Accountants or any other matter arising out of this Agreement or in defending any claim or argument or alleged claim or argument relating to this Agreement or its subject matter.
13.When the Draft Completion Statement is accepted, agreed or finally determined at any stage in accordance with the procedures set out in this Schedule 3:
13.1.the Draft Completion Statement as so accepted, agreed or determined shall constitute the Completion Statement for the purposes of this Agreement and shall (in the absence of fraud or manifest error) be final and binding on the parties; and
13.2.the Actual Cash, the Actual Indebtedness, the Actual Net Debt, the Actual Working Capital Amount and the Actual Working Capital Adjustment shall be as set out in the Completion Statement.
14.Except as expressly provided otherwise in this Schedule 3, the Sellers on the one hand and the Buyer on the other hand shall each bear their own costs, expenses and Taxes arising out of the preparation and/or review of the Draft Completion Accounts and the Draft Completion Statement, and the agreement or determination of the Draft Completion Statement, as applicable.
- 64 - | ||||||||
Schedule 4
FORM OF DRAFT COMPLETION ACCOUNTS AND DRAFT COMPLETION STATEMENT
FORM OF DRAFT COMPLETION ACCOUNTS AND DRAFT COMPLETION STATEMENT
PART A
FORM OF DRAFT COMPLETION ACCOUNTS
FORM OF DRAFT COMPLETION ACCOUNTS
[***]
- 65 - | ||||||||
PART B
FORM OF DRAFT COMPLETION STATEMENT
FORM OF DRAFT COMPLETION STATEMENT
[***]
- 66 - | ||||||||
Schedule 5
ACCOUNTING POLICIES
ACCOUNTING POLICIES
[***]
- 67 - | ||||||||
Schedule 6
COMPLETION REQUIREMENTS
COMPLETION REQUIREMENTS
1.Sellers' and Option Holders' obligations
1.1At Completion, each Seller shall deliver or procure to be delivered to the Buyer (unless otherwise indicated below, copies of each item):
1.1.1the Instrument(s) of Transfer in respect of the Sale Shares in favour of the Buyer or its nominee(s) duly executed by each Seller;
1.1.2as evidence of the authority of each person executing a document referred to in this Schedule 6 on any Seller's behalf (other than a Seller that is an individual):
(a)a copy of the minutes of a duly held meeting of the directors of such Seller (or a duly constituted committee thereof) authorising the execution by such Seller of the document and, where such execution is authorised by a committee of the board of directors of such Seller, a copy of the minutes of a duly held meeting of the directors constituting such committee or the relevant extract thereof; or
(b)a copy of the power of attorney conferring the authority.
1.1.3a copy of the written consent or waiver duly executed by each of Prenetics, the Company Key Management and the Remaining Majority Holder (as defined in the Memorandum and Articles of Association of the Company effective at date of this Agreement) waiving their rights to appoint directors of the Company under Article 29.3 of the Memorandum and Articles of Association of the Company effective at date of this Agreement.
1.2At Completion, each Option Holder shall deliver or procure to be delivered to the Buyer (unless otherwise indicated below, copies of each item):
1.2.1the deed of accession to this Agreement in favour of the Buyer or its nominee(s) duly executed by each Option Holder; and
1.2.2the instrument(s) of transfer in respect of the Option Shares in favour of the Buyer or its nominee(s) duly executed by each Option Holder.
1.3Further, Prenetics and the Holders' Representative (on behalf of all Represented Holders) shall procure and ensure that the Company duly carry out its obligations outlined in paragraph 2 of this Schedule 6.
2.Company's obligations
2.1At Completion, the Company shall deliver or procure to be delivered to the Buyer (unless otherwise indicated below, copies of each item):
2.1.1evidence in a form satisfactory to the Buyer (by way of a certificate signed by the chief executive officer of the Company) of satisfaction of the Conditions;
- 68 - | ||||||||
2.1.2a true copy of the updated register of members of the Company, reflecting the Buyer as the registered holder of all Sale Shares, certified by the registered office provider of the Company;
2.1.3a true copy of the updated register of directors of the Company, reflecting the director changes outlined in paragraphs 2.1.13(c) and 2.1.13(d) of this Schedule 6, certified by the registered office provider of the Company (which shall be duly submitted for filing with competent Government Authority no later than three (3) Business Days after the Completion Date);
2.1.4a true and complete copy of the Amended Articles duly adopted by all necessary action of the directors and/or the shareholders of the Company (which shall be duly submitted for filing with competent Government Authority no later than three (3) Business Days after the Completion Date);
2.1.5a true copy of the certificate of incumbency in respect of the Company issued by the registered office provider of the Company no later than five (5) Business Days before the Completion Date;
2.1.6a true copy of the certificate of good standing in respect of the Company issued by the Registrar of Companies of the Cayman Islands no later than five (5) Business Days before the Completion Date;
2.1.7a duly signed letter to the registered office provider of the Company instructing and authorising the registered office provider that its client of record is now the Buyer or its nominee(s);
2.1.8a duly signed letter to the registered agent of [***] instructing and authorising the registered agent that its client of record is now the Buyer or its nominee(s);
2.1.9the common seal (if any), the certificate of incorporation and the statutory corporate registers of the Company (or confirmation that all of such items are held by the registered office provider of the Company);
2.1.10new share certificate(s) for the Sale Shares in the name of the Buyer;
2.1.11the Disclosure Letter duly executed by the Warrantors;
2.1.12the Deed of Release duly executed by Prenetics and acknowledged by the Company;
2.1.13a copy of the written resolutions of the board of directors of the Company (or the minutes of a duly held meeting of the board of the directors of the Company), including the affirmative vote or consent of at least four members of the board of directors of the Company, approving:
(a)the entry by the Company into this Agreement and the other Transaction Documents to which it is a party and the transactions contemplated hereunder and thereunder
- 69 - | ||||||||
(b)the transfer of the Sale Shares, and the registration of the Buyer as the holder of the Sale Shares (including without limitation, instruction to the registered office provider of the Company to update the register of members of the Company to reflect the transfer of the Sale Shares, to cancel any share certificate(s) for the Sale Shares in the name of the Seller(s) and new share certificate(s) to be issued for the Sale Shares in the name of the Buyer);
(c)the appointment of the persons nominated by the Buyer as directors of the Company (to be identified by the Buyer prior to Completion) with effect from Completion;
(d)the resignations of the relevant directors of the Company (to be identified and agreed between the Company and the Buyer prior to Completion) with effect from Completion;
(e)the adoption of the Amended Articles;
(f)the Redesignation of Class; and
(g)the entry by the Company into the Amended Shareholders' Agreement;
provided that, if required by the Buyer by written notice no later than ten (10) Business Days before the Completion Date, board resolutions of each Group Company other than the Company of similar effect shall be passed to deal with any matter referred to in paragraph 2.1.13 as applicable to the relevant Group Company, copies of which shall be delivered to the Buyer at Completion;
2.1.14a copy of the written resolutions of the Shareholders (or the minutes of a duly held meeting of the Shareholders), including separate meetings of each class of Shareholders, certified by the chief executive officer of the Company, approving:
(a)the entry by the Company into this Agreement and the other Transaction Documents to which it is a Party and the transactions contemplated hereunder and thereunder; and
(b)the adoption of the Amended Articles; and
(c)the adoption of the Redesignation of Class;
2.1.15resignations in the agreed form from the relevant directors and if applicable, company secretary of each Group Company (to be identified and agreed between the Company and the Buyer prior to Completion) expressed to take effect on the Completion Date;
2.1.16a copy of each bank mandate of each Group Company and copies of statements of each bank account of each Group Company made up to a date not earlier than two (2) Business Days before the Completion Date.
- 70 - | ||||||||
2.2Further, the Company shall ensure that:
2.2.1the company chop, common seal (if any) of each Group Company, each register, statutory book, minute book, financial and accounting books and other books required to be kept by each Group Company under Applicable Laws made up to the Completion Date, and each certificate of incorporation and certificate of incorporation on change of name, business registration certificate, and all documents and papers in connection with the affairs of each Group Company and all documents of title to assets of each Group Company; and
2.2.2share certificates for all issued shares in the capital of each Group Company (other than the Company),
shall be kept at the principal business office of the Company at Completion for inspection by the Buyer or its advisers.
3.Buyer's obligations
3.1At Completion, the Buyer shall:
3.1.1deliver or procure to be delivered to the Holders' Representative the instrument(s) of transfer in respect of the Sale Shares duly executed by the Buyer;
3.1.2pay, or cause to be paid, the Initial Consideration in accordance with clause 3.2; and
3.1.3deliver or procure to be delivered to the Holders' Representative, as evidence of the authority of each person executing a document referred to in this Schedule 6 on the Buyer's behalf:
(a)a copy of the minutes of a duly held meeting of the directors or written board resolutions of the Buyer (or a duly constituted committee thereof) authorising the execution by the Buyer of the document and, where such execution is authorised by a committee of the board of directors of the Buyer a copy of the minutes of a duly held meeting of the directors constituting such committee or the relevant extract thereof or written resolutions of such committee; or
(b)a copy of the power of attorney conferring the authority.
- 71 - | ||||||||
Schedule 7
HOLDER WARRANTIES AND WARRANTOR WARRANTIES
HOLDER WARRANTIES AND WARRANTOR WARRANTIES
Part A: Holder Warranties
1.Title and Capacity
1.1Authority and Capacity
1.1.1Such Holder (to the extent it is a company) is validly existing and is a company duly incorporated and in good standing (where such concept is applicable) under the law of its jurisdiction of incorporation.
1.1.2Such Holder has the legal right and full power and authority and has taken all action necessary to enter into, deliver and perform this Agreement and the other Transaction Documents to be executed by it.
1.1.3The Transaction Documents to which such Holder is a party will, when executed, constitute valid and binding obligations on such Holder in accordance with their respective terms.
1.1.4The execution, delivery and performance by such Holder of this Agreement and the other Transaction Documents (to the extent that such Holder is a party to such Transaction Document) do not and will not result in a breach of, constitute a default under or given any person rights of termination, amendment, acceleration or cancellation under (a) any provision of its constitutional documents (to the extent it is a company), (b) any Applicable Laws or regulations or any order, judgment or decree of any court, arbitral tribunal or Government Authority, and/or (c) any agreement or arrangement to which it is party or by which its assets are bound.
1.2No Consent
No consent, approval, order or authorization of, or registration, declaration or filing with, any Government Authority or any other person is required on the part of such Holder in order to enable such Holder to execute this Agreement and the other Transaction Documents to which it is a party, and perform its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby.
1.3Sale Shares
1.3.1Such Holder:
(a)is the legal and beneficial owner of the Sale Shares set out opposite its name in column (B) in Part A or Part B of Schedule 1 (as applicable); and
(b)has the right to exercise all voting and other rights over the Sale Shares set out opposite its name in column (B) in Part A or Part B of Schedule 1 (as applicable).
- 72 - | ||||||||
1.3.2The Sale Shares of such Holder have been properly and validly issued and are each fully paid.
1.3.3There are no Encumbrances, and there is no agreement, arrangement or obligation to create or give an Encumbrance, in relation to the Sale Shares of such Holder. No person has claimed to be entitled to an Encumbrance in relation to any of such Sale Shares.
Part B: Warrantor Warranties
2.Corporate
2.1Group Companies
2.1.1The Group Companies are exempted limited liability companies, limited companies or companies limited by shares (as applicable) (i) duly incorporated under the laws of their respective jurisdiction of incorporation and have been in continuous existence since incorporation, and (ii) duly qualified or licensed as to do business in each jurisdiction where the character of the properties and assets occupied, owned, leased or operated by it or the nature of its business makes such qualification or licensing necessary.
2.1.2There are no Encumbrances, and there is no agreement, arrangement or obligation to create or give an Encumbrance, in relation to any of the shares or unissued shares in the capital of each Group Company.
2.1.3Other than this Agreement and the Shareholders' Agreement, there is no agreement, arrangement or obligation requiring the creation, allotment, issue, transfer, redemption or repayment of, or the grant to a person of the right (conditional or not) to require the allotment, issue, transfer, redemption or repayment of, a share in the capital of a Group Company (including, in each case, an option or right of pre-emption or conversion).
2.1.4No Group Company has any interest in, or has agreed to acquire, any share capital or other security in any other company other than the Group Companies.
2.1.5Save for the Company, AM Robotics 2018 Ltd. (which is in the process of being struck off in accordance with Applicable Law as of the date of this Agreement), ACT Genomics Co., Ltd., ACT Thailand and SG JV, all allotted and issued shares in the capital of each Group Company are legally and beneficially owned by a Group Company alone, has been properly allotted and issued and is fully paid or credited as fully paid.
2.1.6The structure and the details of the Group Companies as set out in Schedule 2 (i) are true, accurate and not misleading as of the date of this Agreement, and (ii) shall be true, accurate and not misleading immediately before the Completion Date except for changes between the date of this Agreement and
- 73 - | ||||||||
the Completion as agreed by the Buyer or that are otherwise immaterial. Except as set out in Schedule 2, no Group Company has any other subsidiary.
2.1.7All actions carried out in connection with any corporate restructuring or reorganisation undertaken by any Group Company were in compliance with any and all Applicable Laws in all material respects. No objection has been received, nor is there any expectation that an objection will be received, from any person (including any credit, contract counterparty, customer or supplier of the Group) and no Group Company shall be liable for loss of any kind (including Tax Liability) arising out of or in connection with such restructuring or reorganisation.
2.2Constitution, Registers and Returns
2.2.1Each of the Group Companies is operating and has always operated its business in accordance with its memorandum and articles of association at the relevant time.
2.2.2Each register, minute book and other book which each of the Group Companies is required by law to keep has been properly kept (except for minor inaccuracies that are inconsequential in nature) and contains a true, accurate and not misleading record of the matters which it is required by Applicable Laws to record. No notice has been received that a register or book is incorrect or should be rectified (other than changes to the members' English names and addresses in the Company's register of members).
2.2.3All returns, particulars, resolutions and other documents required to be delivered or registered by the Group Companies to or with any Government Authority have been properly prepared and delivered except for minor non-compliances that are inconsequential in nature.
2.2.4No Group Company has given a power of attorney or other authority by which a person may enter into an agreement, arrangement or obligation on such Group Company's behalf (other than an authority for a director, other officer or employee to enter into an agreement in the usual course of that person's duties).
2.3Power and Authority; Enforceability
2.3.1Each of the Group Companies has the legal right and full power and authority and has taken all action necessary to enter into and perform this Agreement and the other Transaction Documents to be executed by it.
2.3.2This Agreement has been duly executed by the Company. The Transaction Documents are, or when executed by a Group Company will be, valid and binding obligations of such Group Company, enforceable against such Group Company in accordance with their respective terms, subject to the effect of (i) applicable bankruptcy, winding-up, insolvency, arrangement, reorganization, moratorium or other Applicable Laws of general application now or hereafter
- 74 - | ||||||||
in effect relating to rights of creditors generally, and (ii) the discretion that a court may exercise in the granting of equitable remedies such as specific performance and injunction.
2.4No Consent
No consent, approval, order or authorization of, or registration, declaration or filing with, any Government Authority or any other person is required on the part of the Group Companies in order to enable such Group Company to execute this Agreement and the other Transaction Documents to which it is a party, and perform its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby.
2.5No Conflict
Neither the execution nor the performance of this Agreement by the Company or any other Transaction Document to which it is a party will conflict with, result in a breach of, give rise to an event of default under, require the consent of a person under, enable a person to terminate, or relieve a person from an obligation under any agreement or arrangement to which any Group Company is a party or any legal or administrative requirement by which any Group Company is bound.
3.Information
3.1All information contained in this Agreement and the Disclosure Letter (including any annex thereof) is true, accurate and not misleading in all material respects, and, to each Warrantor's knowledge, there is no fact, matter or circumstances which renders any such information untrue, inaccurate or misleading.
4.Accounts
4.1Accounts
4.1.1The Accounts have been prepared:
(a)in accordance with Applicable Law and with the IFRS at the Last Accounting Date; and
(b)subject to sub-paragraph (a) above, on a basis consistent with that adopted in preparing the Accounts for each of the financial years or periods to which the Accounts relate.
4.1.2The Accounts show a true and fair view of the assets, Liabilities and state of affairs of each of the Group Companies as at the Last Accounting Date and of the profits and losses and cash flow of each of the Group Companies for the financial years ended on the Last Accounting Date.
- 75 - | ||||||||
4.1.3Debts and liabilities
The Accounts fully Disclose and provide adequately for all bad and doubtful debts, all Liabilities (actual, contingent or otherwise) and all financial commitments of each of the Group Companies existing as at the Last Accounting Date except for minor inaccuracies that are inconsequential in nature.
4.1.4Extraordinary and exceptional items
The results shown by the audited profit and loss account of each of the Group Companies in the Accounts as of the Last Accounting Date have not (except as Disclosed in those accounts) been affected by any extraordinary, exceptional or non-recurring item or by another fact or circumstance making the profit or loss for a period covered by any of those accounts unusually high or low.
4.1.5Provision for Tax
The Accounts reserve or provide in accordance with applicable standards, principles and the IFRS for all Tax liable to be assessed on each of the Group Companies, or for which it is or may become accountable, for all periods starting on or before the Last Accounting Date (whether or not the relevant Group Company has or may have a right of reimbursement against another person). The Accounts reserve in accordance with applicable standards, principles and the IFRS for all material contingent or deferred Liabilities to Tax for all periods starting on or before the Last Accounting Date.
4.1.6Depreciation
(a)The bases and rates of depreciation and amortisation used in the Accounts were the same as those used for each of the financial years to which the Accounts relate.
(b)The rates of depreciation and amortisation used in Accounts for the financial years to which the Accounts relate were sufficient to ensure that each fixed asset of the relevant Group Company will be written down to nil by the end of its useful life.
4.1.7Filing of Accounts
Each of the Accounts has been duly filed with the appropriate body in each of the relevant jurisdictions the Group Companies operate in or to which they are subject on a timely basis. For all periods covered by the Accounts, the Group Companies have not received any material written complaint, allegation, assertion or claim regarding the accounting or auditing practices, procedures, methodologies or internal accounting controls of any of the Group Companies, including any material written complaint, allegation, assertion or claim that any of the Group Companies have engaged in improper accounting or auditing practices.
- 76 - | ||||||||
4.1.8No Other Liabilities
The Group Companies have no Liabilities other than: (a) Liabilities reflected on the Accounts, (b) Liabilities which have arisen after the Last Accounting Date in the Ordinary Course of Business, (c) executory obligations under contracts to which the Group Companies is a party, other than in respect of any breach thereof, (d) Transaction Expenses and Indebtedness to be included in the calculation of the Purchase Price, and (e) Liabilities described on Disclosure Letter.
4.2Management Accounts
4.2.1The Management Accounts have been prepared with all due care and attention and in accordance with accounting policies used in preparing the Accounts applied on a consistent basis and fairly present and Disclose with reasonable accuracy the assets, Liabilities, financial position and state of affairs of the Group as at (as applicable) the Management Accounts Date.
4.2.2The Management Accounts are fair and not misleading and do not misstate the assets and Liabilities of each of the Group Companies as at the Management Accounts Date and the profits and losses and cash flow of each of the Group Companies for the period concerned.
4.2.3The Management Accounts fully Disclose and provide adequately for all bad and doubtful debts, all Liabilities (actual, contingent or otherwise) and all financial commitments existing at the Management Accounts Date except for minor inaccuracies that are inconsequential in nature.
4.2.4The Management Accounts are not affected by any unusual, exceptional or non-recurring items.
4.2.5As at the Management Accounts Date, no material Liability, obligation or commitment of any nature whatsoever (asserted or unasserted, known or unknown, absolute or contingent, accrued or unaccrued, matured or unmatured or otherwise) existed in relation to any Group Company which was not required to, or permitted not to, be included in the (as applicable) Management Accounts in accordance with applicable accounting policies.
5.Changes Since the Last Accounting Date
Since the Last Accounting Date:
5.1the Group's business has been operated in the usual way so as to maintain it as a going concern;
5.2there has been no material change or interruption to the Group's business or operations, no material change in the level of borrowing or in the working capital requirements of the Group, and no material reduction in the cash balance or debtors of the Group;
- 77 - | ||||||||
5.3there has been no Material Adverse Effect on the Group as a whole and, to each Warrantor's knowledge, there are no facts or circumstances likely to give rise to such a Material Adverse Effect on the Group as a whole;
5.4no Group Company has declared, paid or made a dividend or distribution (whether in cash or otherwise) to its members;
5.5no Group Company has created, allotted, issued, sold, transferred, acquired, repaid, redeemed, leased, subleased, pledged, encumbered, hypothecated, disposed of or granted share or loan capital or any other securities;
5.6no Group Company has changed its accounting reference date or accounting procedures, policies, principles or practices unless such changes are required pursuant to Applicable Law;
5.7no Group Company has assumed or incurred any material Liability (including any contingent Liability), obligation, commitment or expenditure other than in the Ordinary Course of Business;
5.8Save as contemplated under this Agreement, no Group Company has written off or released debts owed to it;
5.9Save as contemplated under this Agreement for write-off, no Group Company has acquired or disposed of or agreed to acquire or dispose of any material asset, or interest in any share, debenture or any other security of any entity, in each case other than in the Ordinary Course of Business;
5.10no Liability or contingent Liability for Tax has arisen otherwise than as a result of trading activities of the Group in the Ordinary Course of Business;
5.11no material supplier or customer has ceased or substantially reduced its trade with the Group or materially altered the terms under which such suppliers or customers trade to the Group Companies' material disadvantage;
5.12other than pay raises consistent in the Group Companies' Ordinary Course of Business, no change has been made in the emoluments or other terms of employment of any of the employees and the Group Companies have not paid any bonus or special remuneration of more than (a) US$[***] per annum to any person, or (b) US$[***] per annum to all persons in the aggregate; and
5.13no Group Company has made an agreement or arrangement or undertaken an obligation to do any of the foregoing.
6.Financial Obligations
6.1Except as Disclosed in the Accounts, the Group Companies do not have outstanding and have not agreed to create or incur loan capital, borrowings or indebtedness in the nature of borrowings.
- 78 - | ||||||||
6.2None of the Group Companies is party to or is liable under any guarantee, indemnity or other agreement to secure or incur a financial or other obligation with respect to another person's obligation, nor has any Group Company factored any of its debts, or engaged in financing of a type which would not be required to be shown or reflected in the Accounts.
6.3To each Warrantor's knowledge, no event has occurred, or has been alleged either orally or by notice in writing to a Group Company, to have occurred which:
6.3.1constitutes an event of default, or otherwise gives rise to an obligation to repay, under an agreement relating to borrowing or indebtedness in the nature of borrowing (or will do so with the giving of notice or lapse of time or both); or
6.3.2will lead to an Encumbrance constituted or created in connection with borrowing or indebtedness in the nature of borrowing, a guarantee, an indemnity or other obligation of any Group Company becoming enforceable (or will do so with the giving of notice or lapse of time or both).
7.Assets
7.1The Properties
7.1.1None of the Group Companies owns any premises, buildings, land or other real property right.
7.1.2All of the premises, buildings, land or other property rights occupied or otherwise used by the Group have been Disclosed in the Disclosure Letter.
7.2Leases
Where any Property is leased by a Group Company:
7.2.1the requisite details have been Disclosed in the Disclosure Letter and are true and accurate;
7.2.2there is no fact or circumstance (and, to each Warrantor's knowledge, no fact or circumstance will within six (6) months starting on the date of this Agreement occur or arise) which:
(a)could entitle or require a person (including a landlord or licensor) to forfeit or enter on, or take possession of, or occupy, the relevant Property;
(b)could restrict or terminate the Group Company's continued and uninterrupted possession or occupation of the relevant Property; or
(c)could prevent or restrict the relevant Property's development for which planning permission has been or is expected to be obtained;
- 79 - | ||||||||
7.2.3a rent or fee payable in respect of the relevant Property is not as at the date of this Agreement being reviewed;
7.2.4the lease of such Property does not contain an unusually onerous covenant or condition including a covenant or condition allowing the landlord or licensor unreasonably to withhold or delay consent to an assignment of the whole or to an application to carry out a non-structural obligation;
7.2.5the lease of such Property is valid, binding, enforceable and in full force and effect, and the applicable Group Company holds a valid and existing leasehold interest under such Property leases free and clear of any Encumbrances. To each Warrantor's knowledge, there has been no actual termination of any such lease agreement due to any material breach on the part of the applicable Group Company of any covenants, conditions and agreements contained in the agreement; and
7.2.6all of the rent payable under such Property lease has been paid up to date.
7.3Condition of Property
To each Warrantor's knowledge:
7.3.1there is no material deficiency which requires correction in the state or condition of any building or other structure on or forming part of any Property; and
7.3.2no flooding, subsidence or other material defect of any kind (including a design or construction defect) affects or has affected any Property.
7.4Properties subject to Leases and Other Agreements
None of the Group Companies has leased, subleased, licensed or granted occupancy rights in any parcel or any portion of any parcel of the Group's Properties to any other person and no other person has any rights to the use, occupancy or enjoyment thereof pursuant to any lease, sublease, licence, occupancy or other agreement, and none of the Group Companies has assigned its interest under any lease or sublease listed in the Disclosure Letter to any third party. Where any Property is the subject of any lease or other agreement relating to its occupation or use for the benefit of any person other than a Group Company, the requisite details have been Disclosed in the Disclosure Letter.
7.5Assets
7.5.1All material assets included in the Management Accounts or acquired by a Group Company since the Management Accounts Date, other than the Properties, the rights in Intellectual Property and any assets disposed of or realised in the Ordinary Course of Business, and excepting rights and retention of title arrangements arising by operation of law in the Ordinary Course of Business, are (i) legally and beneficially owned solely by the relevant Group
- 80 - | ||||||||
Company; (ii) where capable of possession, in the possession or under the control of the relevant Group Company, and (iii) free from Encumbrances.
7.5.2Each of the Group Companies owns or has the right to use such asset used in and is necessary for the effective operation of its business.
7.5.3No Group Company is party to, nor is liable under, a lease or hire, hire purchase, credit sale or conditional sale or similar agreement.
7.5.4All inventory, machinery, vehicles, equipment and other tangible assets owned, possessed or used by the Group Companies are in good condition and working order and have been regularly and properly maintained in all material respects in accordance with generally accepted industry practice, except for normal wear and tear, and are adequate for the uses to which they are being put. None is in need of material renewal or replacement or surplus to such Group Company's requirements, except for routine or regular maintenance works.
7.5.5Each Group Company's inventory or stock (subject to normal wear and tear) is of satisfactory quality and saleable in the Ordinary Course of Business.
7.5.6Each material asset of the Group, which is normal to have maintained by independent or specialist contractors, has been regularly maintained to a good technical standard and in accordance with safety regulations required to be observed in relation to it.
7.6Debts
7.6.1No debt shown in the Accounts, the Management Accounts or a Group Company's accounting records is overdue by more than twelve (12) weeks or is the subject of an arrangement.
7.6.2No Group Company has released a material debt shown in the Accounts, the Management Accounts or its accounting records so that the debtor has paid or will pay less than the debt's book value. None of the material debts shown in the Accounts, the Management Accounts or a Group Company's accounting records has been deferred, subordinated or written off or become irrecoverable to any extent. To each Warrantor's knowledge, each of those debts will realise its book value in the usual course of collection.
8.Intellectual Property and Information Technology
8.1General
8.1.1The Disclosure Letter contains true, accurate and not misleading details in all material respects of all the Intellectual Property which are used by the Group for the operation of the Business in respect of which any Group Company is registered as the owner or is the applicant for registration (the "Registered Owned IP").
- 81 - | ||||||||
8.1.2The trademarks, patents and domain names listed in the Disclosure Letter constitute all Registered Owned IP that is necessary for the operation of the Business of the Group.
8.1.3Except for the Disclosed Registered Owned IP, no member of the Group has rights to (whether as owner, licensee, licensor or otherwise) any registered or applied for Intellectual Property that is material to the operation of the Group's Business.
8.1.4All of the Registered Owned IP is valid and enforceable and nothing has been done or omitted to be done by which it may cease to be valid or enforceable, or affect the ownership of any Business IP.
8.1.5All renewal and maintenance fees and Taxes due up to and including the date of this Agreement in respect of each of the Registered Owned IP have been paid in full and on time. All other actions due or required to maintain and protect the Registered Owned IP have been taken on time.
8.1.6The Business IP as Disclosed in the Disclosure Letter comprise all of the Intellectual Property which is necessary for the operation of the Group's Business.
8.1.7To each Warrantor's knowledge, each Group Company has taken all steps necessary or desirable for the fullest protection of all Business IP. Each Group Company has not disposed of, agreed to dispose of or, except for the licenses granted in the Ordinary Course of Business as disclosed in the Disclosure Letter, granted to any person any rights over any Business IP.
8.1.8Except for licenses granted in the Ordinary Course of Business as disclosed in the Disclosure Letter, no Group Company has granted or is obliged to grant any licence, assignment, Encumbrance, consent, undertaking or other right in respect of the Business IP or agreed to any restriction on use or any disclosure obligation or entered into any co-existence agreement in respect of the Business IP.
8.1.9None of the Business IP or any Group Company's ownership or rights in the Business IP will be adversely affected by the transactions contemplated by this Agreement.
8.1.10Other than any commercially available off-the-shelf software, there is no Intellectual Property used or required for the operation of the Group's Business that is licensed from third parties.
8.1.11All Intellectual Property which is or is likely to be material to the Business of the Group, including all Intellectual Property developed by employees or consultants of the Group, or by a third party for the Group, is legally and beneficially vested exclusively in a Group Company.
- 82 - | ||||||||
8.1.12There is no outstanding obligation or restriction owed by a Group Company that may impact the Business or freedom of the Group to develop other works in the Ordinary Course of Business.
8.1.13All agreements to which any of the Group Companies is a party, or by which it is bound relating to the Business IP, are Disclosed in the Disclosure Letter.
8.2Trade secrets
8.2.1The Group has taken all commercially reasonable and necessary steps to maintain the secrecy of all its Confidential Information, trade secrets and know-how used in the operation of the Business of the Group.
8.2.2All Confidential Information, trade secrets and know-how used or in connection with the Business of the Group have been kept confidential and have not been disclosed to third parties (other than parties who have signed written confidentiality undertakings in respect of such information or such disclosure is required pursuant to Applicable Law or order from Government Authorities). To each Warrantor's knowledge, no person has breached an obligation of confidence which it owes to the Group with respect to any Confidential Information.
8.3Infringement
8.3.1To each Warrantor's knowledge, there is not, and has not at any time been, an infringement, misappropriation, misuse, violation or other unauthorised use of any of the Business IP. No such proceedings (including any litigation, arbitration, or other dispute resolution mechanism) have been threatened, and no Group Company has received any notice relating to such proceedings. To each Warrantor's knowledge, no fact or circumstance exists which might give rise to proceedings of that type.
8.3.2None of the Business IP is the subject of any opposition, invalidation, revocation or cancellation proceedings or counterclaim or any other proceedings or counterclaim (including any litigation, arbitration, or other dispute resolution mechanism) concerning its validity, enforceability, or the relevant Group Company's title to such right. No such proceedings have been threatened, and no Group Company has received any notice relating to such proceedings. To each Warrantor's knowledge, no fact or circumstance exists which might give rise to proceedings of that type.
8.3.3There is no and has not in the last three (3) years been any civil, criminal, arbitration, administrative or other proceedings or dispute in any jurisdiction concerning any of the Business IP.
8.3.4To each Warrantor's knowledge, the activities of each Group Company do not infringe, nor have they infringed, misappropriated, misused, violated or otherwise made use without authorisation of any third party Intellectual Property. No such proceedings (including any litigation, arbitration, or other
- 83 - | ||||||||
dispute resolution mechanism) have been threatened, and no Group Company has received any notice relating to such proceedings. To each Warrantor's knowledge, no fact or circumstance exists which might give rise to proceedings of that type.
8.4Information Technology and Data Protection
8.4.1Each element of the Business IT is owned by, or used under an agreement with or validly licensed to, the relevant Group Company. The Disclosure Letter contains true, accurate and not misleading details of all such Business IT and relevant agreements and/or licences in all material respects. Each of the Group Companies has complied with all the terms and conditions of all licences and agreements to which it is a party or by which it is bound relating to such Business IT licensed from third parties in all material respects.
8.4.2Each Group Company has prudent procedures, including adequate disaster recovery arrangements, in place to ensure the security of the Business IT and the data stored on it.
8.4.3To each Warrantor's knowledge, no Group Company has been or is in breach of any data security breach reporting or notification requirement under any Applicable Laws.
8.4.4In the last three (3) years, the Business IT has not suffered any breakdowns or security breaches that have materially affected or are materially affecting the Group's Business and to each Warrantor's knowledge, no circumstances exist which are likely to give rise to such breakdown, breach or disruption.
8.4.5The Business IT is in good working order and functions efficiently in accordance with all applicable specifications in all material respects.
8.4.6The Business IT is the subject of warranty and/or maintenance arrangements which are adequate for the requirements of each Group Company's Business in all material respects.
8.4.7The present capacity of the Business IT is sufficient in order to satisfy the current requirements of the Business of the Group with regard to data processing and communications.
8.4.8Each Group Company has necessary and reasonable security measures in place in order to keep processed personal data strictly confidential, to the extent required by Applicable Laws. No claims or proceedings have been raised or pending and, to each Warrantor's knowledge, no claims or proceedings have been threatened with regard to processing of personal data by any Group Company.
8.4.9Each Group Company is in material compliance with Applicable Laws and any guidance notes or guidelines relating to personal data that are issued from time to time by the Government Authorities.
- 84 - | ||||||||
8.4.10Each Group Company has established and maintained a system of internal controls that is reasonably designed to ensure compliance with all material requirements of Applicable Laws relating to personal data.
8.4.11No Group Company has received any complaints from any individual regarding the Group Company's processing of his/her personal data or has compensated or been required to compensate any individual under Applicable Laws in relation to personal data, no claim for such compensation is outstanding and, to each Warrantor's knowledge there is no reason to believe that any circumstances exist which might lead to any claim for compensation being made.
8.4.12The consummation of the transactions contemplated under this Agreement will not give rise to any breach of Applicable Laws including Applicable Laws relating to personal data.
9.Contracts
9.1Contracts
No Group Company is party to or subject to any contract which:
9.1.1is not in the Ordinary Course of Business;
9.1.2is not on an arm's length basis;
9.1.3restricts such Group Company's freedom to operate the whole or part of its Business or to use or exploit any of its assets;
9.1.4cannot be complied with on time or without undue or unusual expenditure of money or effort by such Group Company; or
9.1.5is of a long-term nature that is incapable of termination in accordance with its terms by such Group Company on six (6) months' notice or less.
9.2Validity of Contracts
9.2.1Section 9.2 of the Disclosure Letter includes all contracts, commitments, undertakings, arrangements or obligations, which are relating to the Business of the Group in effect as of the date hereof and to which the Group Company is a party (each a "Material Contract"), and which:
(a)relate to revenue or provide for a Liability of the Company equivalent to more than USD 100,000 in aggregate or per annum;
(b)grant exclusive rights to any party thereto;
(c)contain any provision that refers to a change of control of the Group Company;
- 85 - | ||||||||
(d)restrict the Group Company from engaging in or competing in any line of business or with any person;
(e)constitute a partnership, joint-venture or similar agreement or arrangement material for the Business of the Group;
(f)constitute agreements or arrangements with any Seller or any of their respective Affiliates, or any director or officer of any of their Affiliates; or
(g)constitute agreements or arrangements otherwise material to the Group.
9.2.2All of the Material Contracts are valid, binding and enforceable on the parties thereto in accordance with their respective terms. To each Warrantor's knowledge, no fact or circumstance exists which might invalidate or give rise to a ground for termination, avoidance or repudiation of any Material Contract. No party with whom a Group Company has entered into a Material Contract has given notice of its intention to terminate, or has sought to repudiate or disclaim, the relevant Material Contract.
9.2.3Neither a Group Company (on the one hand) nor any party with whom a Group Company (on the other hand) has entered into a Material Contract is in material breach of such Material Contract. To each Warrantor's knowledge, no fact or circumstance exists which might give rise to a breach of this type.
9.2.4Each Material Contract is legally binding and enforceable in accordance with its respective terms.
9.2.5In respect of the sale and purchase of the Sale Shares in accordance with this Agreement, no consent is required from any third party that is a party to any Material Contract.
9.3Joint Ventures etc.
Other than in relation to SG JV and CERBACT Asia Holdings Pte. Ltd. Taiwan Branch, no Group Company is, or has agreed to become, a member of any joint venture, consortium, partnership or other unincorporated association.
10.Employees and Benefits
10.1General
10.1.1The Disclosure Letter contains true, accurate and not misleading details in relation to the Group Companies, of:
(a)the total number of each Group Company's employees including those who are on maternity or other statutory leave or other long-term leave of absence and who have or may have a right to return to work with a Group Company;
(b)the salary and other benefits, period of continuous employment, grade and location of each Group Company's employees;
- 86 - | ||||||||
(c)the standard terms of the contract of employment of each director, other officer and employee of each Group Company; and
(d)the terms of all consultancy agreements under which services are provided to the Company by an individual who is either a party to such a contract or has a controlling interest in a legal entity which is party to such a contract.
10.1.2True, accurate and not misleading copies or details have been Disclosed of:
(a)any employee handbook or similar document issued by or on behalf of a Group Company (whether issued in paper or electronic form) and all policies relating to the provision of benefits applicable to each grade or category of employee; and
(b)each bonus, commission, profit sharing, share option, share incentive or other incentive schemes for any director, officer or employee including details of numbers of employees eligible to participate, maximum and target bonus opportunities and details of amounts accrued to date as at the date of this Agreement.
10.1.3Neither any Group Company nor any employee is in material breach of the relevant employment contracts. No fact or circumstance exists which might give rise to a breach of this type.
10.1.4Each employment contract with the directors, officers and employee is legally binding and enforceable in accordance with its respective terms.
10.1.5There is no, and in the past three (3) years there has been no, trade union, works council or other body representing any of the employees of any Group Company vis-a-vis the relevant Group Company. No Group Company is involved in, or has during the twelve (12) months prior to the date of this Agreement been involved in, any strike, lock-out or industrial dispute with, or in respect of, its employees.
10.1.6No Group Company has provided or agreed to provide a gratuitous payment or benefit to a current or former director, officer or employee or to any of their dependants.
10.1.7No Group Company is liable (on an actual or a contingent basis) to make any payment or provide any benefit to any of its current or former directors, officers or employees, (or any dependant of such person) in connection with the proposed termination or suspension of employment or services, or variation of any contract of employment.
10.1.8Each Group Company is, and has been prior to the date of this Agreement, in compliance with all applicable employment and labour laws and regulations, including without limitation provisions thereof relating to wages, hours, employee benefits, mandatory provident fund contributions, employees'
- 87 - | ||||||||
compensation insurance and collective bargaining, and is not liable or delinquent for any arrear of wages, Tax or penalty for failure to comply with the foregoing.
10.1.9No Group Company has any outstanding Liability in excess of US$[***] for breach or termination of an employment contract between it and its current or former directors, officers or employees.
10.1.10 No Group Company has:
(a)adopted, amended, or committed to any new benefit plan, pension, or incentive scheme;
(b)incurred any undisclosed Liability for employee compensation or benefits; or
(c)any outstanding or threatened claims related to compensation or employment matters.
10.2Termination
Save as otherwise Disclosed in the Disclosure Letter, no employee of any Group Company with an annual salary exceeding US$[***] has given or received notice during the past six (6) months to terminate his or her employment and as of the date of this Agreement no such person has ceased to be employed or engaged by any Group Company.
10.3Pension Schemes
Other than in accordance with the terms of the statutory pension schemes applicable in the jurisdiction of each Group Company, the Group Companies have not paid, and no proposal has been announced to enter into or establish, any agreement or arrangement for the payment or contribution towards a pension, allowance or lump sum or other similar benefit on retirement, death, illness, disability or termination of employment (whether voluntary or not) for the benefit of its current or former directors or officers or employees (or their respective dependents).
11.Legal Compliance
11.1Licences and Consents
11.1.1All licences, consents and authorisations material to the Business of the Group have been obtained, are in force and are being complied with in all material respects. All actions required for the relevant renewal or extension of each such material licence, consent and authorisation have been taken. All schemes (such as Personal Information file security maintenance plans) required for the Business of each Group Company have been established and are in compliance with Applicable Laws in all material respects.
11.1.2No written notification has been received by the Group in the past three (3) years that any of such licences, consents or authorisations is likely to be
- 88 - | ||||||||
suspended, modified or revoked and no such licences, consents or authorisations will be breached, revoked, suspended, cancelled, varied or not renewed (whether as a result of the execution or performance of this Agreement or any document to be executed at or before Completion or otherwise).
11.2Compliance with law
11.2.1Each of the Group Companies and its directors, officers and employees has conducted the Business of the Group and dealt with the Group's assets in accordance with all Applicable Laws in all material respects.
11.2.2There is not and has not been any investigation, enquiry or disciplinary proceeding instigated by or involving any Government Authority (including the TIC, Taiwan Ministry of Health and Welfare, and Taiwan Food and Drug Administration) concerning or involving a Group Company or any of its directors, officers and employees, and none is pending or threatened. To each Warrantor's knowledge, no fact or circumstance exists which might give rise to an investigation, enquiry or proceedings of that type.
11.2.3There is no outstanding judgment, order, decree, arbitral award or decision of a court, tribunal, arbitrator or Government Authority against any Group Company or any of its directors, officers and employees.
11.2.4each Group Company has complied with all corporate governance requirements in all material respects, including but not limited to: (a) validly adopting and filing its articles of association with the companies registry; (b) duly passing and recording all necessary board and shareholder resolutions; and (c) maintaining all required corporate records in accordance with Applicable Laws.
11.2.5No Group Company is in material breach of any corporate governance obligations, and no notices of non-compliance, penalties, or disputes related to corporate governance are pending or threatened.
11.3Anti-Bribery and Sanctions
11.3.1None of the Group Companies, the Sellers, nor their respective agents, employees, and other persons acting on their behalf, have taken or will take any action that would cause the Group or any member of the Buyer's Group to be in violation of anti-corruption laws, anti-money laundering laws or sanctions laws.
11.3.2Without limiting the generality of the foregoing, none of the Group Companies, the Sellers, nor their respective agents, employees, and other persons acting on their behalf, have taken or will take any action in furtherance of a payment, offer, promise to pay, or authorisation or ratification of a payment of any gift, money or anything of value to:
- 89 - | ||||||||
(a)a Government Official; or
(b)any person or entity while knowing or having reasonable grounds to believe that all or a portion of that payment will be passed on to a Government Official,
to influence any act or decision of any Government Official in his or her official capacity to secure an improper advantage (e.g., to obtain a Tax rate lower than permitted by law) in order to obtain or retain business.
11.3.3No Group Company is subject to any investigation by, or request for information from, law enforcement officials regarding a violation or potential violation of anti-corruption laws, anti-money laundering laws or sanctions laws by a Group Company.
11.3.4No Group Company has received any allegation or conducted any internal investigation related to a violation or potential violation of anti-corruption laws, anti-money laundering laws or sanctions laws by a Group Company.
11.3.5To each Warrantor's knowledge, there are no facts or circumstances that would lead a reasonable person to believe that there is a likelihood that any of the officers, directors, employees or agents of the Group Companies has made any payment in violation of any anti-corruption laws, anti-money laundering laws or sanctions laws on behalf of or for the benefit of a Group Company.
11.3.6The Group has established and continues to maintain reasonable internal controls and procedures for monitoring cash payments and managing the book-keeping practices of the Group to the extent required by Applicable Law.
11.3.7None of the current officers, directors, employees or agents of the Group Companies are or were in the past five (5) years Government Officials.
11.3.8The Group has:
(a)maintained its books and records in a manner that, in reasonable detail, accurately and fairly reflects the transactions and disposition of its assets; and
(b)maintained a system of internal accounting controls sufficient to provide reasonable assurances that:
(i)transactions are executed and access to assets is given only in accordance with management's authorisation,
(ii)transactions are recorded as necessary to permit preparation of periodic financial statements and to maintain accountability of corporate assets; and
- 90 - | ||||||||
(iii)recorded assets are compared with existing assets at reasonable intervals and appropriate action is taken with respect to any discrepancies between recorded and actual assets.
11.3.9No director or officer of the Group has (i) made or caused to be made false or misleading statements to an accountant, or (ii) attempted to coerce or fraudulently influence, an accountant in connection with any audit, review or examination of the financial statements of the Group.
11.3.10Notwithstanding any other provision of this Agreement to the contrary, nothing herein shall (i) require the Buyer to make any payment that it reasonably believes will constitute a violation of any anti-corruption laws, anti-money laundering laws or sanctions laws or (ii) prohibit the Buyer, in its sole discretion, from reporting any actual or possible violation of the anti-corruption laws, anti-money laundering laws or sanctions laws to law enforcement officials.
12.Litigation
12.1Current Proceedings
No Group Company nor any of the directors, officers and employees of the Group is, or has in the past three (3) years been, involved, whether as claimant or defendant or other party, in any claim, legal action, proceedings, suit or litigation in excess of US$[***] (other than as claimant in the collection of debts arising in the ordinary course of its business).
12.2Pending or Threatened Proceedings
No written notification has been received by a Group Company in the past three (3) years to the effect that a claim, legal action, proceedings, suit or litigation in excess of US$[***] is pending or threatened by or against a Group Company or any of the directors, officers and employees of the Group and, to each Warrantor's knowledge, no fact or circumstance exists which will or would reasonably be likely to give rise to a claim, legal action, proceedings, suit or litigation involving a Group Company or any of the directors, officers and employees of the Group, which will or would reasonably be likely to result in a Liability exceeding US$[***].
13.Insurance
13.1General
Each of the Group Companies has at all material times been and is as at the date of this Agreement adequately insured against professional negligence, accident, damage, injury, third party loss, loss of profits and all other reasonably foreseeable risks to which a person operating the types of business operated by each of the Group Companies is exposed. The insurance policies are sufficient for compliance by each of the Group Companies with the requirements of Applicable Laws and of all contracts to which any Group Company is a party in all material respects.
- 91 - | ||||||||
13.2Details on Policies
In respect of each of the insurances referred to in paragraph 13.1 above:
13.2.1it is valid and enforceable and is not void or voidable;
13.2.2the relevant Group Company has not done anything or omitted to do anything which might:
(a)make any of the insurance policies void or voidable; or
(b)prejudice the ability to effect insurance on the same or better terms in the future;
13.2.3all premiums have been duly paid to date;
13.2.4no Group Company has received any written notification that such insurances are not valid or enforceable;
13.2.5to each Warrantor's knowledge, there are no special or unusual limits, terms, exclusions or restrictions in any of such policies;
13.2.6to each Warrantor's knowledge, the premiums payable are not in excess of the normal rates; and
13.2.7the relevant Group Company has not done anything or omitted to do anything, and there is nothing, which might result in an increase in the premium payable under any such policies.
13.3Claims
13.3.1No claims have been made, no claim is outstanding and, to each Warrantor's knowledge, no fact or circumstance exists which will or would reasonably be likely to give rise to a claim under any of the insurance policies.
13.3.2to each Warrantor's knowledge, no event, act or omission has occurred which requires notification under any of the insurance policies.
13.3.3None of the insurers under any of the insurance policies has given any notice in writing to refuse or to the effect that it intends to refuse, indemnity in whole or in part in respect of any claims under the insurance policies.
13.3.4Nothing has been done or omitted to be done, and there is nothing, which will or would reasonably be likely to entitle the insurers under any of the insurance policies to refuse indemnity in whole or in part in respect of any claims under the insurance policies.
- 92 - | ||||||||
14.Tax
14.1Returns, Information and Clearances
All returns, computations, notices and information which are or have been required to be made or given by a Group Company for any Taxation purpose (i) have been made or given within the requisite periods and on a proper basis and are up to date and correct and (ii) none of them is the subject of any dispute with or investigation by any Tax Authority.
14.2Taxation Claims, Liabilities and Reliefs
14.2.1To each Warrantor's knowledge, there is no Liability to Taxation in respect of which a claim for breach of warranties could be made.
14.2.2All Taxation of any nature whatsoever for which a Group Company is liable and/or which is assessed or imposed by any Tax Authority, or for which a Group Company is liable to account, has been duly and punctually paid, withheld or has been accrued in the Accounts or Management Accounts.
14.2.3All material documents to which a Group Company is a party upon which stamp duty is payable have been duly stamped and there are no circumstances or transactions to which any Group Company is or has been a party such that a Liability to stamp duty or any penalty in respect of such duty may arise for any Group Company.
14.2.4The Tax losses of each of the Group Companies, as Disclosed in the most recent corporate income tax return submitted to the relevant Tax Authority for each of the Group Companies are correct and up-to-date in all material respects.
14.2.5Each of the Group Companies:
(a)is not liable for any penalty, surcharge, fine or interest in connection with Tax in respect of any period up to and including Completion;
(b)has made all deductions and withholdings in respect of or on account of any Taxation from all payments made by it, which deductions and withholdings it was obliged to make, and has accounted to the relevant Tax Authority for all amounts so deducted or withheld;
(c)is not and does not expect to be involved in a dispute in relation to Tax in respect of any period up to and including Completion;
(d)is not and has not been subject to any investigation or enquiry by a Tax Authority concerning its Tax affairs and none is pending or threatened, and to each Warrantor's knowledge, no fact or circumstance exists which will or would reasonably be likely to give rise to an investigation or enquiry of that type;
- 93 - | ||||||||
(e)is not a party to or otherwise involved in any transaction, agreement or arrangement which will, or is reasonably likely to, subject it to Tax in respect of an amount deemed for Tax purposes to be income or gains of such Group Company where such amount is not reflected as income or gains of such Group Company in the Accounts or Management Accounts; and
(f)is not a party to any transaction or series of transactions of which the main purpose, or one of the main purposes, was or could reasonably be likely to constitute the avoidance of, or deferral or a reduction in the Liability for, Tax.
14.2.6Each of the Accounts and Management Accounts have made full provision or reserve for all Tax liable to be assessed on each of the Group Companies or for which it may be or may after that date become liable, with respect to Tax on profits, gains, income and receipts, benefits and other items subject to Tax for any period ending on or before, and for any transactions, or events or situations occurring (or deemed to have occurred for Taxation purposes) on or before, the Last Accounting Date or the Management Accounts Date (as the case may be).
14.2.7There is no order, decree or decision of any Tax Authority outstanding or pending against any Group Company.
14.2.8Each of the Group Companies has maintained sufficient records to support all returns lodged, filed or submitted to the relevant Tax Authority relating to Taxation (or which may be legally required to be filed, lodged or submitted) and to comply with all Applicable Laws and regulations.
14.2.9No Tax incentive, exemption or Relief has been claimed by any Group Company in the two (2) years prior to the date of this Agreement without such Group Company having satisfied the conditions for such Tax incentive, exemption or Relief. No Tax Authority has operated or agreed to operate any special arrangement (being an arrangement which is not based on relevant legislation or any published practice) in relation to any Group Company's affairs.
14.3Arm's-length dealings
No Group Company is or has been a party to or otherwise involved in any transaction, agreement or arrangement otherwise than by way of a bargain at arm's length, or any transaction, agreement or arrangement (whether or not by way of a bargain at arm's length) under which it has been or is or may be required to make any payment for any goods, services or facilities provided to it which is in excess of the market value of such goods, services or facilities or under which it has been, or is or may be required to provide goods, services or facilities for a consideration which is less than the market value of such goods, services or facilities and/or in consequence of which it is or will be liable to Tax in respect of an amount deemed for Tax purposes to be income
- 94 - | ||||||||
or gains of the Group Company but not actually income or gains of the Group Company.
14.4Company Residence
Each of the Group Companies has been resident for Taxation purposes in its place of incorporation or other jurisdiction by operation of any double tax agreement and nowhere else at all times since its incorporation and has, and has had, no branch, agency, place of business or permanent establishment outside such place or jurisdiction which may result in that Group Company being subject to Tax in that other place or jurisdiction.
15.Insolvency
15.1.1No Group Company is insolvent under the laws of its jurisdiction of incorporation or unable to pay its debts as they fall due nor has any Group Company commenced negotiations with one (1) or more of its creditors with a view to rescheduling or restructuring any of its indebtedness.
15.1.2There are no proceedings in relation to any compromise or arrangement with creditors or any winding-up, bankruptcy or other insolvency proceedings concerning any Group Company.
15.1.3To each Warrantor's knowledge, no steps have been taken to enforce any security over any assets of any Group Company.
15.1.4No Group Company has received a notice to the effect that an administrator has been or will be appointed in relation to the relevant Group Company. No notice has been given or filed with the court of an intention to appoint an administrator and no petition or application has been presented or order made for the appointment of an administrator in respect of any Group Company.
15.1.5No receiver or administrative receiver has been appointed, nor any notice given to the relevant Group Company of an intention to appoint, any such person, over the whole or part of any Group Company's business or assets.
15.1.6No Group Company has proposed or agreed to a composition, compromise, assignment or arrangement with any of its creditors.
15.1.7No distress, execution, attachment, sequestration or other process has been levied on an asset of a Group Company which remains undischarged.
15.1.8No action is being taken by a court to dissolve a Group Company and/or strike it off the relevant register.
15.1.9No Group Company is, in any jurisdiction, subject to or threatened by any other procedures or steps which are analogous to those set out above.
- 95 - | ||||||||
16.Brokerage or Commissions
No person is entitled to receive a finder's fee, brokerage or other commission from a Group Company in connection with the transactions contemplated, directly or indirectly, by this Agreement.
- 96 - | ||||||||
Schedule 8
BUYER WARRANTIES
BUYER WARRANTIES
1.Title and Capacity
1.1The Buyer is validly existing and in good standing (if such concept is applicable) and is a company with limited liability duly incorporated under the laws of Taiwan.
1.2The Buyer has the legal right and full power and authority and has taken all corporate action necessary to enter into and perform this Agreement and the other Transaction Documents to be executed by it.
1.3The Transaction Documents to which the Buyer is a party will, when executed, constitute valid and binding obligations on the Buyer in accordance with their respective terms.
1.4The execution, delivery and performance by the Buyer of this Agreement and the other Transaction Documents (to the extent that the Buyer is a party to such Transaction Documents) do not and will not result in a breach of, constitute a default under, require any consent under, or given any person rights of termination, amendment, acceleration or cancellation under (a) any provision of its constitutional documents, (b) any Applicable Laws or regulations or any order, judgment or decree of any court, arbitral tribunal or Government Authority which would materially delay or hinder the Buyer's consummation of the transaction contemplated by this Agreement, except for such consents described in Schedule 11 to this Agreement, and/or (c) any agreement or arrangement to which it is party or by which its assets are bound which would materially delay or hinder the Buyer's consummation of the transaction contemplated by this Agreement.
2.Compliance with Law
2.1The Buyer has not been notified of any investigation, enquiry or disciplinary proceeding instigated by any Government Authority concerning the Buyer that could materially delay or hinder the Buyer's consummation of the transaction contemplated by this Agreement.
2.2There is no outstanding judgment, order, decree, arbitral award or decision of a court, tribunal, arbitrator or Government Authority against the Buyer that could materially delay or hinder the Buyer's consummation of the transaction contemplated by this Agreement.
3.Sufficient Funds
The Buyer's cash on hand and other financial resources available to the Buyer, will, in the aggregate, be sufficient for the Buyer to pay the Purchase Price payable pursuant to this Agreement.
- 97 - | ||||||||
4.Solvency
After giving effect to the consummation of the transactions contemplated by this Agreement and the other Transaction Documents, the Buyer will not be insolvent under the laws of the Taiwan or unable to pay its debts as they fall due.
5.Disclosure of Information
The Buyer has had an opportunity to ask questions and receive answers from the Sellers regarding the terms and conditions of the offering of the Sale Shares, and the business, properties, prospects and financial condition of the Company.
- 98 - | ||||||||
Schedule 9
LIMITATIONS ON LIABILITY
LIMITATIONS ON LIABILITY
1.LIMITATION ON QUANTUM
1.1Basket. Notwithstanding anything to the contrary in this Agreement but subject to paragraph 6, none of the Indemnifying Parties shall be liable in respect of any Warranty Claim:
1.1.1unless the amount recoverable from the Indemnifying Parties in respect of such Warranty Claim exceeds US$[***]. For this purpose, all Warranty Claims arising out of the same, related or similar subject matter, facts, events or circumstances shall be aggregated and form a single Warranty Claim; and
1.1.2unless the aggregate amount recoverable from the Indemnifying Parties in respect of such Warranty Claim and any other Warranty Claims exceeds US$[***] (the "Basket"),
in which case, the Indemnifying Parties shall be liable for such aggregated amount and not just the amount in excess of the Basket.
1.2Cap. Notwithstanding anything to the contrary in this Agreement but subject to paragraph 6, the total amount that the Buyer is entitled to recover from each Indemnifying Party shall be limited to:
1.2.1in respect of a Holder Warranty Claim, a Fundamental Warranty Claim, or a Tax Warranty Claim, [***]% of the Purchase Price received by such Indemnifying Party,
1.2.2in respect of a Warrantor Warranty Claim (other than a Fundamental Warranty Claim or a Tax Warranty Claim), [***]% of the Purchase Price received by such Indemnifying Party; and
1.2.3in respect of any Relevant Claim other than a Warranty Claim, [***]% of the Purchase Price received by such Indemnifying Party,
for the avoidance of doubt, subject always to the limitations set out in this Schedule 9, each Indemnifying Party (i) shall be liable to Buyer in respect of a Relevant Claim for any breach of the Warrantor Warranties made by any Warrantor, but (ii) shall only be liable in respect of a Relevant Claim for any breach of the Holder Warranties made by himself/herself/itself in respect of himself/herself/itself, or for non-performance of his/her/its own obligations under this Agreement.
2.LIMITATION PERIOD
2.1No Indemnifying Party shall be liable:
2.1.1in respect of any Holder Warranty Claim, Fundamental Warranty Claim, or any Relevant Claim other than a Warranty Claim (other than a Tax Warranty Claim or a Fundamental Warranty Claim), unless the Indemnity Notice for
- 99 - | ||||||||
such Warranty Claim is given to the Indemnifying Party on or before the sixth (6th) year anniversary of the Completion Date;
2.1.2in respect of any Warrantor Warranty Claim (other than a Tax Warranty Claim or a Fundamental Warranty Claim), unless the Indemnity Notice for such Warranty Claim is given to the Indemnifying Party on or before the eighteenth (18th) month anniversary of the Completion Date;
2.1.3in respect of any Tax Warranty Claim or Special Tax Claim, unless the Indemnity Notice for such Tax Warranty Claim or Special Tax Claim is given to the Indemnifying Party on or before 90 days after the expiration of the applicable statutory limitation period for Taxes.
2.2So long as a Relevant Claim is notified within the applicable time period described above, such claim shall survive and the Indemnifying Parties shall remain liable until such claim has been finally resolved in accordance with the terms of this Agreement.
2.3To the maximum extent permissible under Applicable Laws, no Indemnifying Party shall rely on the Limitation Ordinance (Cap. 347, Laws of Hong Kong) or similar statutes under Applicable Laws, laches or any other time-based defence in respect of any Warranty Claim.
3.SPECIFIC LIMITATIONS
3.1No Indemnifying Party shall be liable in respect of any Relevant Claim to the extent that:
3.1.1the Losses arising from such Relevant Claim are covered by an insurance policy of a Group Company and payment is made by the insurer to the relevant Group Company (for the avoidance of doubt, any gap amount not fully covered and paid by the insurer shall still be liable by Indemnifying Party);
3.1.2the matter giving rise to such Relevant Claim is a direct result of the passing of, or any change in, any Applicable Law after the date of this Agreement which is not actually or prospectively in effect as at the date of this Agreement;
3.1.3the matter giving rise to such Relevant Claim is a direct result of any change in accounting or Tax policies, bases or practices binding upon a Group Company or the Buyer, introduced after the date of this Agreement;
3.1.4the matter giving rise to such Relevant Claim is a direct result of any increase in the rates of Tax announced after the date of this Agreement or any imposition of Tax not actually in effect on the date of this Agreement; or
3.1.5the matter giving rise to such Relevant Claim is a direct result of (a) voluntary actions taken by the Buyer after the date of this Agreement, (b) any actions taken by the Warrantors, Holders or Group Companies at the express direction
- 100 - | ||||||||
of the Buyer, or (c) any actions taken pursuant to a binding commitment made by a Group Company prior to the date of this Agreement which has been Disclosed to the Buyer.
4.NO DOUBLE RECOVERY
The Buyer may not recover from the Indemnifying Parties under this Agreement more than once in respect of the same Loss suffered.
5.MITIGATION
Nothing in this Schedule 9 restricts or limits the Buyer's common law duty to mitigate any Losses suffered by it as a result of a Relevant Claim.
6.GENERAL
Nothing in this Schedule 9 restricts or limits any Liability of any Indemnifying Party in respect of a Relevant Claim arising as a result of any fraud, wilful misconduct, gross negligence, or reckless misrepresentation on the part of such Indemnifying Party.
- 101 - | ||||||||
Schedule 10
ACTION PENDING COMPLETION
ACTION PENDING COMPLETION
Each of the Group Companies shall:
1.carry on its business in the Ordinary Course of Business in accordance with all Applicable Laws and shall not make (or agree to make) any payment other than routine payments in the Ordinary Course of Business and usual course of trading;
2.take all commercially reasonable and necessary steps to preserve and protect its assets and not create, or agree to create or amend, an Encumbrance over any asset or redeem, or agree to redeem, an existing Encumbrance over any asset;
3.not take any action which is inconsistent with the provisions of the Transaction Documents or the consummation of the transactions contemplated by the Transaction Documents, which would materially and adversely affect the Company's ability to perform its obligations under the Transaction Documents and the consummation of the transactions contemplated by the Transaction Documents;
4.not create, allot, issue, acquire, repay or redeem any share or loan capital or agree, arrange or undertake to do any of those things or acquire or agree to acquire, an interest in a corporate body or merge or consolidate with a corporate body or any other person, enter into any demerger transaction or participate in any other type of corporate reconstruction;
5.not adopt a plan of complete or partial liquidation, dissolution, merger, consolidation or recapitalization;
6.not establish any subsidiary or enter into any joint venture or partnership;
7.preserve sufficient working capital for its day-to-day business operation in the Ordinary Course of Business;
8.not acquire or dispose of, or agree to acquire or dispose of, any revenues, assets, business or undertakings except in the Ordinary Course of Business or assume or incur, or agree to assume or incur, any Liability, obligation or expense (actual or contingent) except in the Ordinary Course of Business;
9.not make, or agree to make, capital expenditure exceeding in total US$100,000 (or its equivalent at the time) or incur, or agree to incur, a commitment or commitments involving capital expenditure exceeding in total US$100,000 (or its equivalent at the time), except in accordance with this Agreement and the Transaction Documents;
10.not pass a shareholders' resolution, or make any distribution, except in accordance with this Agreement and the Transaction Documents;
11.not make any amendments to the constitutional documents of any Group Company, except in accordance with this Agreement and the Transaction Documents;
- 102 - | ||||||||
12.continue, without amendment, each of the insurance policies of the Group and not do, or omit to do, anything which might:
12.1make any such policy void or voidable;
12.2entitle any of the insurers under any such policy to refuse indemnity in relation to particular claims in whole or in part; or
12.3result in an increase in the premium payable under any such policies;
13.not enter into, amend or terminate any long-term, onerous, unusual or material agreement, arrangement or obligation, other than in the Ordinary Course of Business;
14.not amend the terms and conditions of employment or engagement of a director, other officer or employee of any Group Company or provide, or agree to provide, a gratuitous payment or benefit to a director, officer or employee of any Group Company (or any of their dependants) or employ, engage or terminate the employment or engagement of, a person, except in accordance with this Agreement and the Transaction Documents;
15.not employ or make any offer to employ any person with annual base compensation in excess of US$100,000;
16.not amend, or agree to amend, the terms of its borrowing or indebtedness in the nature of borrowing or create, incur, or agree to create or incur, borrowing or indebtedness in the nature of borrowing (except pursuant to facilities Disclosed in the Disclosure Letter where the borrowing or indebtedness in the nature of borrowing does not exceed the amount available to be drawn by a Group Company under those facilities), except in accordance with this Agreement and the Transaction Documents;
17.not give, or agree to give, a guarantee, indemnity or other agreement to secure, or incur financial or other obligations with respect to, another person's obligation;
18.not enter into an agreement, arrangement or obligation (whether legally enforceable or not) in which a Holder, a director or former director of a Group Company or a person connected with any of them is interested, except in accordance with this Agreement and the Transaction Documents;
19.not establish an employee incentive scheme or enter into any obligation or arrangement of any employee incentive scheme, or communicate to a current or former employee a plan, proposal or an intention to establish any employee incentive scheme or enter into any employee incentive scheme;
20.not start litigation or arbitration proceedings;
21.not compromise or settle litigation or arbitration proceedings or any Action, demand or dispute or waive a right in relation to litigation or arbitration proceedings;
22.not change its accounting policies or its auditors;
- 103 - | ||||||||
23.not change residence for Tax purposes or otherwise establish a taxable presence in any jurisdiction where the Group Company is not currently subject to Tax;
24.not enter into any transaction or arrangement outside the Ordinary Course of Business which would give rise to a Tax Liability for the Group Company;
25.not make a Tax election, amend a Tax return or submit a Tax return which is inconsistent with past practice, in each case to the extent that any such election, amendment or inconsistency could reasonably be expected to give rise to or increase a Liability to Tax or utilise any Relief of any Group Company;
26.not settle or compromise any Tax dispute with a Tax Authority; and
27.not enter into any agreement or arrangement (conditional or otherwise) to do any of the foregoing.
- 104 - | ||||||||
Schedule 11
LIST OF CONSENTS, APPROVALS AND WAIVERS
LIST OF CONSENTS, APPROVALS AND WAIVERS
[***]
- 105 - | ||||||||
Schedule 12
TAXES AND LIABILITIES
TAXES AND LIABILITIES
Part A: Settlement of related party loans/transactions; payment of withholding taxes
[***]
Part B: Tax Liabilities subject to Tax Indemnification Escrow
[***]
- 106 - | ||||||||
Appendix I
FORM OF DEED OF RELEASE
FORM OF DEED OF RELEASE
[***]
- 107 - | ||||||||
Appendix II
AMENDED ARTICLES
AMENDED ARTICLES
[***]
- 108 - | ||||||||
Appendix III
FORM OF ESCROW AND PAYING AGENT AGREEMENT
FORM OF ESCROW AND PAYING AGENT AGREEMENT
[***]
- 109 - | ||||||||
Appendix IV
FORM OF INSTRUMENT OF TRANSFER
FORM OF INSTRUMENT OF TRANSFER
[***]
- 110 - | ||||||||
IN WITNESS whereof the Parties have caused this Agreement to be duly executed as a deed on the date first above written.
THE BUYER
EXECUTED AND DELIVERED AS A DEED for and on behalf of DELTA ELECTRONICS, INC. by its directors | ) | ______________________________ Name: Capacity: | ||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ______________________________ Name: Capacity: | |||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
By executing this Deed each signatory warrants that such signatory is duly authorized to execute this document on behalf of DELTA ELECTRONICS, INC. | |||||
[Project Apex – Signature Page to Share Purchase Agreement]
IN WITNESS whereof the Parties have caused this Agreement to be duly executed as a deed on the date first above written.
THE HOLDERS' REPRESENTATIVE
EXECUTED AND DELIVERED AS A DEED By [***] | ) | ______________________________ | ||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||

[Project Apex – Signature Page to Share Purchase Agreement]
IN WITNESS whereof the Parties have caused this Agreement to be duly executed as a deed on the date first above written.
THE COMPANY
EXECUTED AND DELIVERED AS A DEED for and on behalf of ACT GENOMICS HOLDINGS COMPANY LIMITED by its directors | ) | ______________________________ Name: Capacity: | ||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ______________________________ Name: Capacity: | |||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
By executing this Deed each signatory warrants that such signatory is duly authorized to execute this document on behalf of ACT GENOMICS HOLDINGS COMPANY LIMITED. | |||||
[Project Apex – Signature Page to Share Purchase Agreement]
IN WITNESS whereof the Parties have caused this Agreement to be duly executed as a deed on the date first above written.
THE SELLERS
[IF SELLER IS AN ENTITY]
EXECUTED AND DELIVERED AS A DEED for and on behalf of ____________________________________________ by its directors | ) | ______________________________ Name: Capacity: | ||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | _______________________________Name: Capacity: | |||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
By executing this Deed each signatory warrants that such signatory is duly authorized to execute this document on behalf of ______________________. | |||||
[IF SELLER IS A NATURAL PERSON]
[Project Apex – Signature Page to Share Purchase Agreement]
EXECUTED AND DELIVERED AS A DEED By ___________________________ | ) | ______________________________ | ||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||
) | ||||||||

[Project Apex – Signature Page to Share Purchase Agreement]
Exhibit 4.28
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. Certain schedules have been excluded from this exhibit because they are both (i) not material and (ii) not otherwise disclosed in the exhibit or our annual report on Form 20-F for the fiscal year ended December 31, 2025. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
ASSET PURCHASE AGREEMENT
by and among
PRENETICS GLOBAL LIMITED,
EUROPA SPORTS PARTNERS, LLC,
HUBMATRIX PARTNERS, LLC,
IM8 GLOBAL LIMITED,
[***],
and
and
[***]
Dated as of January 1, 2026
TABLE OF CONTENTS
ASSET PURCHASE AGREEMENT
This Asset Purchase Agreement (this “Agreement”), dated as of January 1, 2026, is entered into among Europa Sports Partners, LLC, a Delaware limited liability company (“Europa Sports Seller”), and Hubmatrix Partners, LLC, a Delaware limited liability company (“Hubmatrix Seller”), Prenetics Global Limited, a Cayman Islands limited liability company (“Seller Parent”, and together with Europa Sports Seller and Hubmatrix Seller, the “Sellers”), IM8 Global Limited, a Cayman Islands company (“IM8”), [***], a Delaware limited liability company (“Buyer”), and [***], a Delaware corporation (“Buyer Parent”).
RECITALS
Sellers are engaged in the business of providing third-party logistics, fulfillment, warehousing, and distribution services to direct-to-consumer and e-commerce brands, including inventory management, order fulfillment, shipping, and related logistics services, together with all associated assets, operations, customer relationships, contracts, and goodwill as conducted by the Sellers as of the date hereof (the “Business”).
Sellers wish to sell and assign to Buyer, and Buyer wishes to purchase and assume from Sellers, substantially all the assets, and certain specified liabilities, of the Business, subject to the terms and conditions set forth herein.
Buyer is a wholly-owned subsidiary of Buyer Parent.
In consideration of the mutual covenants and agreements hereinafter set forth and for other good and valuable consideration, the receipt and sufficiency of which are hereby acknowledged, the parties hereto agree as follows:
Article I
PURCHASE AND SALE
PURCHASE AND SALE
Section 1.01Purchase and Sale of Assets. Subject to the terms and conditions set forth herein, at the Closing, Sellers shall sell, convey, assign, transfer, and deliver to Buyer, and Buyer shall purchase from Sellers, all of Sellers’ right, title, and interest in, to, and under all of the tangible and intangible assets, properties, and rights of every kind and nature and wherever located (other than the Excluded Assets), which relate to, or are used or held for use in connection with, the Business (collectively, the “Purchased Assets”) free and clear of any Encumbrances, including the following:
(a)all inventory, finished goods, raw materials, work in progress, packaging, supplies, parts, and other inventories related to the operations of Hubmatrix;
(b)all contracts, leases, licenses, instruments, notes, commitments, undertakings, indentures, joint ventures, and all other agreements, commitments, and legally binding arrangements, whether written or oral, including customer agreements, vendor agreements, warehouse agreements, partner agreements, and equipment lease agreements (the “Assigned Contracts”), including as set forth on Section 1.01(b) of the Disclosure Schedules;
(c)all Intellectual Property Assets (as defined herein);
(d)all furniture, fixtures, equipment, machinery, tools, vehicles, office equipment, supplies, computers, information technology hardware, cables, telephones, and other tangible personal property, including as set forth on Section 3.09 of the Disclosure Schedules (the “Tangible Personal Property”);
(e)all rights to any Actions of any nature available to or being pursued by Sellers to the extent related to the Business, the Purchased Assets or the Assumed Liabilities, whether arising by way of counterclaim or otherwise;
(f)all prepaid expenses, credits, advance payments, claims, security, refunds, rights of recovery, rights of set-off, rights of recoupment, deposits, charges, sums, and fees (including any such item relating to the payment of Taxes);
(g)all of Sellers’ rights under warranties, indemnities, and all similar rights against third parties to the extent related to any Purchased Assets;
(h)the real property leases set forth on Section 1.01(h) of the Disclosure Schedules (the “Assigned Real Property Leases”);
(i)all insurance benefits, including rights and proceeds, arising from or relating to the Business, the Purchased Assets or the Assumed Liabilities, including as set forth on Section 1.01(i) of the Disclosure Schedules;
(j)originals or, where not available, copies, of all books and records, including books of account, ledgers, and general, financial, and accounting records, machinery and equipment maintenance files, customer lists, customer purchasing histories, price lists, distribution lists, supplier lists, production data, quality control records and procedures, customer complaints and inquiry files, research and development files, records, and data (including all correspondence with any federal, state, local, or foreign government or political subdivision thereof, or any agency or instrumentality of such government or political subdivision, or any arbitrator, court, or tribunal of competent jurisdiction (collectively, “Governmental Authority”), sales material and records, strategic plans and marketing, and promotional surveys, material, and research (“Books and Records”); and
(k)all goodwill and the going concern value of the Purchased Assets and the Business.
For the avoidance of doubt, the inclusion of any reference in this Section 1.01 to any schedule or list is for identification and organizational purposes only, and no Purchased Asset shall be excluded from the Purchased Assets solely by reason of not being listed or identified on any schedule.
Section 1.02Excluded Assets. Buyer expressly understands and agrees that it is not acquiring, and Sellers are not selling or assigning, any assets of the Buyer not included in the Purchased Assets, or any of the following assets, each of which as measured and existing up to and including the Closing Date (the “Excluded Assets”):
(a)All cash, securities and cash equivalents of Sellers;
(b)The shared contracts of Sellers that are related to multiple divisions of its business and are not exclusively related to the Business (the “Shared Contracts”) as set forth in Section 1.02(b) of the Disclosure Schedules;
(c)All accounts receivable held by Sellers (“Accounts Receivable”)
(d)All bank accounts of Sellers;
(e)All Benefit Plans and trusts or other assets attributable thereto; and
(f)All inventory, finished goods, raw materials, work in progress, packaging, supplies, parts, and other inventories of the Sellers related to the Europa Sports distribution business.
2
Section 1.03Assumed Liabilities.
(a)Subject to the terms and conditions set forth herein, Buyer shall assume and agree to pay, perform, and discharge only the following Liabilities of Sellers (collectively, the “Assumed Liabilities”), and no other Liabilities:
(i)all Liabilities in respect of the Assigned Contracts listed on Section 1.01(b) of the Disclosure Schedules but only to the extent that such Liabilities are required to be performed after the Closing Date; and
(ii)all other Liabilities arising out of or relating to Buyer’s ownership or operation of the Business and the Purchased Assets (which for the avoidance of doubt shall include the Assigned Real Property Leases) on or after the Closing.
(b)Notwithstanding any provision in this Agreement to the contrary, Buyer shall not assume and shall not be responsible to pay, perform, or discharge any Liabilities of Sellers or any of their Affiliates of any kind or nature whatsoever other than the Assumed Liabilities (the “Excluded Liabilities”). For the avoidance of doubt, any Liability arising out of an Assumed Contract that is not listed on Schedule 1.01(b) will be an Excluded Liability except to the extent Buyer expressly agrees after the Closing Date that it will assume such Liability.
Section 1.04Share Consideration.
(a)Purchase Price. The aggregate consideration for the Purchased Assets shall consist of (1) the issuance of the Closing Shares, having an aggregate value of $[***], (2) subject to being earned pursuant to Section 1.04(c) and Section 1.04(d), the Contingent Shares, having an aggregate value of up to $[***] (items (1) and (2), the “Purchase Price”), payable solely in duly authorized, validly issued, fully paid and non-assessable shares of Non-Voting Common Stock, par value $[***] per share, of Buyer Parent (“Common Stock”), and (3) Buyer Parent’s assumption of the Assumed Liabilities. For the avoidance of doubt, the maximum number of shares that may be issued to Sellers as part of the Purchase Price shall be [***].
(b)Closing Consideration. At the Closing, Buyer Parent shall issue to IM8 or an Affiliate designated by the Seller Parent [***] shares of Common Stock (the “Closing Shares”); provided, however, that the Closing Shares shall be subject to forfeiture and shall not vest unless and until the [***] Contingency is satisfied. If the [***] Contingency is not satisfied, the Closing Shares shall be automatically forfeited and cancelled as of the [***] Contingency Failure Date without any consideration or payment to Sellers, and Buyer Parent shall update, and Sellers hereby authorize Buyer Parent to update, its books and records to reflect such forfeiture and cancellation as of such date. In addition, if (i) the [***] Agreement does not remain in full force and effect for a continuous period of six (6) months from the Closing Date, or if there is a material breach by [***] (including, for the avoidance of doubt, [***]’s non-payment of fees) or any termination during such six-month period; or (ii) [***] provides notice during such six-month period of its intent to terminate the [***] Agreement, in each case, other than in the case of a [***] Breach Termination, then from and after the date of such termination or breach, Seller Parent shall reimburse Buyer Parent for [***] percent ([***]%) of all rent, operating expenses, taxes, insurance, and other costs and expenses payable by Buyer Parent under the [***] Lease and the [***] Lease (collectively, the “Lease Costs”), which reimbursement obligation shall continue with respect to each lease until the termination or expiration of such lease. Seller Parent shall pay such reimbursement amounts to Buyer Parent within fifteen (15) days after receipt of an invoice from Buyer Parent, together with reasonable supporting documentation. Any failure by Seller Parent to timely pay such reimbursement amounts shall constitute a material breach of this Agreement and shall entitle Buyer Parent to all remedies available at law or in equity, including indemnification under Article VII.
(c)Shipment Consideration.
3
(i)Upon IM8 US’s shipment of its 600,000th Qualified Order, Buyer Parent shall issue to IM8 or an Affiliate designated by the Seller Parent a number of shares of Common Stock equal to the Adjusted Contingent Tranche Value, divided by the Buyer Parent Share Value, rounded down to the nearest whole share (the “600k Shipment Shares”). For the avoidance of doubt, if less than 600,000 Qualified Orders are shipped, no 600k Shipment Shares shall be issued.
(ii)Upon IM8 US’s shipment of its 1,000,000th Qualified Order (inclusive of the 600,000 Qualified Orders referenced in clause (i)), Buyer Parent shall issue to IM8 or an Affiliate designated by the Seller Parent a number of shares of Common Stock equal to the Adjusted Contingent Tranche Value, divided by the Buyer Parent Share Value, rounded down to the nearest whole share (the “1M Shipment Shares”, and together with the 600k Shipment Shares, the “Shipment Shares”). For the avoidance of doubt, if less than 1,000,000 Qualified Orders are shipped, no 1M Shipment Shares shall be issued.
(d)Referral Revenue Consideration.
(i)If, at any time during the Referral Measurement Period, the cumulative Referral Revenue equals or exceeds $[***], then Buyer Parent shall issue to IM8 or an Affiliate as designated by the Seller Parent a number of shares of Common Stock equal to the Adjusted Contingent Tranche Value, divided by the Buyer Parent Share Value, rounded down to the nearest whole share (the “$[***]M Referral Shares”). For the avoidance of doubt, if cumulative Referral Revenue is less than $[***], then no $[***]M Referral Shares shall be issued.
(ii)If, at any time during the Referral Measurement Period, the cumulative Referral Revenue equals or exceeds $[***], then Buyer Parent shall issue to IM8 or an Affiliate as designated by the Seller Parent a number of shares of Common Stock equal to the Adjusted Contingent Tranche Value, divided by the Buyer Parent Share Value, rounded down to the nearest whole share (the “$[***]M Referral Shares”, and together with the $[***]M Referral Shares, the “Referral Shares”). For the avoidance of doubt, if cumulative Referral Revenue is less than $[***], then no $[***]M Referral Shares shall be issued.
(e)Reporting. Following the Closing, (x) through the end of calendar year 2026, Buyer Parent shall deliver to Seller Parent, within thirty (30) days after the end of each fiscal quarter, a written report setting forth in reasonable detail the cumulative number of orders shipped by IM8 pursuant to the IM8 Agreement as tracked by Buyer Parent’s Order Management System during such fiscal quarter and since January 1, 2026, together with a calculation showing Buyer Parent’s good faith determination of whether any issuance of Shipment Shares has been triggered pursuant to Section 1.04(c), and (y) until the expiration of the Referral Measurement Period, Buyer Parent shall deliver to Seller Parent Quarterly Reports in accordance with the Referral Agreement. Following the Closing, through the date that is twelve (12) months after the Closing Date, Buyer Parent shall deliver to Seller Parent, within thirty (30) days after the end of each three-month period, a written report setting forth in reasonable detail (i) the cumulative fees paid by [***] under the [***] Agreement during the period from the Closing Date through the end of such three-month period, (ii) the status of the [***] Agreement (including whether it remains in full force and effect and whether any breach, default, or termination notice has been given or received), and (iii) if applicable and if it has been determined by Buyer Parent, the [***] Contingency Failure Date. Within thirty (30) days following the twelve (12) month anniversary of the Closing Date, Buyer Parent shall deliver to Seller Parent a final written report certifying whether or not the [***] Contingency has been satisfied, and if applicable, the [***] Contingency Failure Date which determination shall be final and binding on the parties absent manifest error.
(f)Defined Terms. As used in this Agreement, the following terms shall have the meanings set forth below:
4
(i)“Adjusted Contingent Tranche Value” means, with respect to any issuance of Contingent Shares, the Contingent Tranche Value as reduced, if applicable, pursuant to Section 7.05.
(ii)“Buyer Parent Share Value” means (i) with respect to the Closing Shares, $[***], and (ii) with respect to any other Consideration Shares, the price per share paid by investors for the most recently issued capital stock of Buyer Parent for cash in a bona fide arm’s-length transaction consummated prior to the applicable determination date, but in no event shall the Buyer Parent Share Value be less than $[***].
(iii)“Consideration Shares” means the Closing Shares and the Contingent Shares.
(iv)“Contingent Shares” means the Shipment Shares and the Referral Shares.
(v)“Contingent Tranche Value” means, with respect to a Tranche, the notional value of $[***].
(vi)“Qualified Order” means an order made by a customer during the period beginning January 1, 2026, and ending December 31, 2026, pursuant to the IM8 Agreement.
(vii)“Quarterly Report” shall have the meaning given to such term in the Referral Agreement.
(viii)“Referral Measurement Period” means the period beginning on the Closing Date and ending on the date that is eighteen (18) months thereafter.
(ix)“Referral Revenue” shall have the meaning given to such term in the Referral Agreement.
(x)“Tranche” means each issuance of Contingent Shares pursuant to Section 1.04(c) or Section 1.04(d).
(g)Consideration Shares Issued to Affiliate. To the extent Seller’s Parent designates an Affiliate to receive all or any portion of the Consideration Shares issuable hereunder, such Affiliate shall, as a condition to the issuance of such Consideration Shares, be required to execute and deliver to Buyer’s Parent a written instrument making the representations and warranties set forth in Section 3.18, all of which must be true and correct as of the date of issuance of such Consideration Shares.
Section 1.05Allocation of Purchase Price. The Purchase Price and the Assumed Liabilities shall be allocated among the Purchased Assets for all purposes (including Tax and financial accounting) in accordance with an allocation schedule prepared by Buyer following the Closing in accordance with Section 1060 of the Internal Revenue Code of 1986, as amended (the “Allocation Schedule”). The Allocation Schedule shall be deemed final unless Sellers notify Buyer in writing that Sellers object to one or more items reflected in the Allocation Schedule within thirty (30) days after the delivery of the Allocation Schedule to Sellers. In the event of any such objection, Sellers and Buyer shall negotiate in good faith to resolve such dispute; provided, however, that if Sellers and Buyer are unable to resolve any dispute with respect to the Allocation Schedule within thirty (30) days after the delivery of the Allocation Schedule to Sellers, such dispute shall be resolved by KPMG or another impartial regionally recognized firm of independent certified public accountants mutually appointed by Sellers and Buyer. The fees and expenses of such accounting firm shall be borne equally by Sellers and Buyer. Buyer and Sellers shall file all returns, declarations, reports, information returns and statements, and other documents relating to Taxes (including amended returns and claims for refund) (“Tax Returns”) in a manner consistent with the Allocation Schedule.
5
Section 1.06Withholding Tax. Buyer Parent shall be entitled to deduct and withhold from the Consideration Shares such amounts as Buyer Parent may be required to deduct and withhold under any provision of Tax Law, and may satisfy such obligation by reducing the number of shares of Common Stock otherwise deliverable to Sellers by a number of shares having an aggregate Buyer Parent Share Value (determined as of the date of issuance of the applicable shares) equal to the amount required to be deducted and withheld. Buyer Parent shall provide Sellers with written notice of its intent to withhold at least ten (10) days prior to the Closing (or, if later, prior to the issuance of such shares) together with a written explanation substantiating the requirement to deduct or withhold, and the parties shall use commercially reasonable efforts to cooperate to mitigate or eliminate any such withholding to the maximum extent permitted by Law. All such withheld amounts shall be treated as delivered to Sellers hereunder.
Section 1.07Third-Party Consents.
(a)To the extent that Sellers’ rights under any Purchased Asset may not be assigned to Buyer without the consent of another Person (a “Required Consent”) and such Required Consent has not been obtained on or prior to the Closing, this Agreement shall not constitute an agreement to assign such Purchased Asset if an attempted assignment would (i) constitute a breach thereof, (ii) be unlawful, or (iii) otherwise be ineffective. Buyer and Sellers shall use commercially reasonable efforts to obtain any such Required Consents as promptly as practicable; provided, however, that neither Buyer nor Sellers shall be required to pay any material consideration in connection therewith.
(b)If any Required Consent is not obtained, or if any attempted assignment would be ineffective or would impair Buyer’s rights under the Purchased Asset in question so that Buyer would not, in effect, acquire the benefit of such Purchased Asset, then Buyer and Sellers shall use commercially reasonable efforts to enter into such arrangements as may be necessary to provide Buyer, to the extent permitted under applicable Law, with the economic and, to the extent reasonably practicable, operational equivalent of the transfer of such Purchased Asset. Such arrangements shall include the prompt remittance by Sellers to Buyer of any income, proceeds, or other monies received by Sellers in connection with such Purchased Asset, together with the transfer to Buyer of any related Assumed Liabilities, in each case as of the Closing.
Article II
CLOSING
CLOSING
Section 2.01Closing. Subject to the terms and conditions of this Agreement, the consummation of the transactions contemplated by this Agreement (the “Closing”) shall take place remotely by exchange of documents and signatures (or their electronic counterparts), at 10 a.m. Eastern Time, simultaneously with the execution of this Agreement, or at such other time or place or in such other manner as Sellers and Buyer may mutually agree upon in writing. The date on which the Closing is to occur is herein referred to as the “Closing Date.”
Section 2.02Closing Deliverables.
(a)At the Closing, Sellers, as applicable, shall deliver or cause to be delivered to Buyer Parent and Buyer the following:
(i)an executed copy of a bill of sale, assignment and assumption agreement, substantially in the form attached hereto as Exhibit B (the “Bill of Sale, Assignment and Assumption Agreement”), transferring the Tangible Personal Property included in the Purchased Assets to Buyer and effecting the assignment to and assumption by Buyer of the Purchased Assets and the Assumed Liabilities ;
(ii)one or more executed assignments, substantially in the form attached hereto as Exhibit C (each, an "Intellectual Property Assignment"), transferring all of Sellers’ right, title and interest in and to the Intellectual Property Assets to Buyer;
6
(iii)executed Landlord Consents, substantially in the forms attached hereto as Exhibits D-1 and D-2,
(iv)the consents of the Persons listed on Schedule 2.02(a)(vii);
(v)an employee leasing agreement in respect of post-Closing services to be provided by the Sellers or their Affiliates to the Buyer, substantially in the form attached hereto as Exhibit E, duly executed by Sellers (the “Employee Leasing Agreement”);
(vi)an executed Referral Agreement, substantially in the form attached hereto as Exhibit F (the “Referral Agreement”)
(vii)a certificate of the Secretary (or equivalent officer) of Europa Sports Seller certifying as to (A) the resolutions of the sole member of Europa Sports Seller, which authorize the execution, delivery, and performance of the Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and (B) the names and titles of the officers of Europa Sports Seller authorized to sign this Agreement and the other Transaction Documents;
(viii)a certificate of the Secretary (or equivalent officer) of Hubmatrix Seller certifying as to (A) the resolutions of the sole member of Hubmatrix Seller, which authorize the execution, delivery, and performance of the Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and (B) the names and titles of the officers of Hubmatrix Seller authorized to sign this Agreement and the other Transaction Documents;
(ix)a certificate of the Secretary (or equivalent officer) of Seller Parent certifying as to (A) the extracts of resolutions of the board of directors of Seller Parent, which authorize the execution, delivery, and performance of the Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and (B) the names and titles of the officers of Seller Parent authorized to sign this Agreement and the other Transaction Documents;
(x)an executed Joinder to Buyer Parent’s Right of First Refusal and Co-Sale Agreement, in the form attached hereto as Exhibit G;
(xi)an executed Adoption Agreement to Buyer Parent’s Voting Agreement, in the form attached hereto as Exhibit H; and
(xii)such other customary instruments of transfer or assumption, filings, or documents, in form and substance reasonably satisfactory to Buyer, as may be required to give effect to the transactions contemplated by this Agreement.
(b)At the Closing, Buyer or Buyer Parent, as applicable, shall deliver to Sellers the following:
(i)the Closing Shares;
(ii)an executed copy of the Bill of Sale, Assignment and Assumption Agreement;
(iii)an executed copy of each Intellectual Property Assignment;
(iv)an executed copy of the Employee Leasing Agreement;
(v)an executed copy of the Referral Agreement;
7
(vi)a certificate of the Secretary (or equivalent officer) of Buyer certifying as to (A) the resolutions of the sole Member of Buyer, which authorize the execution, delivery, and performance of this Agreement and the Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and (B) the names and titles of the officers of Buyer authorized to sign this Agreement and the other Transaction Documents; and
(vii)a certificate of the Secretary (or equivalent officer) of Buyer Parent certifying as to (A) the resolutions of the board of directors of Buyer Parent, which authorize the execution, delivery, and performance of this Agreement and the Transaction Documents and the consummation of the transactions contemplated hereby and thereby, and (B) the names and titles of the officers of Buyer Parent authorized to sign this Agreement and the other Transaction Documents.
Article III
REPRESENTATIONS AND WARRANTIES OF SELLER
REPRESENTATIONS AND WARRANTIES OF SELLER
Sellers jointly and severally represent and warrant to Buyer and Buyer Parent that, except as set forth on the disclosure schedules delivered by the Sellers to the Buyer and Buyer Parent concurrently herewith (the “Disclosure Schedules”), the statements contained in this ARTICLE III are true and correct as of the date hereof.
Section 3.01Organization and Authority of Sellers. Europa Sports Seller and Hubmatrix Seller are both limited liability companies duly organized, validly existing, and in good standing under the Laws of Delaware. Seller Parent is a limited liability company duly organized, validly existing, and in good standing under the Laws of the Cayman Islands. Sellers have full corporate power and authority to enter into this Agreement and the other Transaction Documents to which Sellers are a party, to carry out their obligations hereunder and thereunder, and to consummate the transactions contemplated hereby and thereby. Sellers are duly licensed or qualified to do business and are in good standing in each jurisdiction in which the ownership of the Purchased Assets or the operation of the Business as currently conducted makes such licensing or qualification necessary, except where the failure to be so licensed, qualified or in good standing would not have a Material Adverse Effect. The execution and delivery by Sellers of this Agreement and any other Transaction Document to which Sellers are a party, the performance by Sellers of their obligations hereunder and thereunder, and the consummation by Sellers of the transactions contemplated hereby and thereby have been duly authorized by all requisite corporate, board, and shareholder action on the part of Sellers. This Agreement and the Transaction Documents have been duly executed and delivered by Sellers, and assuming due authorization, execution and delivery by Buyer, each constitute a legal, valid, and binding obligation of Sellers enforceable against Sellers in accordance with their respective terms, subject to the Enforceability Exceptions.
Section 3.02No Conflicts or Consents. Except as set out in Schedule 3.02 of the Disclosure Schedule, the execution, delivery, and performance by Sellers of this Agreement and the other Transaction Documents to which Sellers are a party, and the consummation of the transactions contemplated hereby and thereby, do not and will not: (a) violate or conflict with any provision of the certificate of incorporation, by-laws, or other governing documents of Sellers or any of their subsidiaries; (b) violate or conflict with any provision of any Law or Governmental Order applicable to Sellers or any of their subsidiaries, the Business, or the Purchased Assets; (c) require the consent, notice, declaration, or filing with or other action by any Person or require any permit, license, or Governmental Order; (d) violate or conflict with, result in the acceleration of, or create in any party the right to accelerate, terminate, modify, or cancel any Contract to which Sellers or any of their subsidiaries is a party or by which Sellers or any of their subsidiaries or the Business is bound or to which any of the Purchased Assets are subject (including any Assigned Contract); or (e) result in the creation or imposition of any charge, claim, pledge, equitable interest, lien, security interest, restriction of any kind, or other encumbrance (“Encumbrance”) on the Purchased Assets.
Section 3.03Financial Statements; Books and Records; Accounts Receivable.
8
(a)Sellers have delivered to Buyer copies of: (i) the audited balance sheet and related statement of income and cash flows for the Business for the fiscal year ended December 31, 2023, and the unaudited balance sheet and related statement of income and cash flows for the Business for the fiscal year ended December 31, 2024 (collectively, the “Annual Financial Statements”); (ii) the unaudited balance sheet and related statements of income and cash flows for the Business as of September 30, 2025 for the 9- month period then ended (the “Interim Financial Statements”, collectively with the Annual Financial Statements, the “Financial Statements”). The Financial Statements (i) have been prepared in accordance with GAAP, applied on a consistent basis throughout the periods covered thereby, (ii) fairly present the financial position, results of operations and cash flows of the Business as at the dates and for the periods indicated therein, and (iii) are consistent with the books and records of the Business maintained in the ordinary course of business.
(b)The books of account and other business records of the Business, all of which have been previously delivered to Buyer and its representatives, are accurate and complete in all material respects.
(c)All receivables reflected in the balance sheet in the Interim Financial Statements (the “Balance Sheet”), or which have arisen from the conduct of the Business since the date of the Balance Sheet (the “Balance Sheet Date”), are valid and have arisen only from bona fide, arms-length transactions entered into in the ordinary course of business consistent with past practices, and are not subject to any known defense, offset or any counterclaim. Such receivables, net of reserves, are fairly presented in accordance with GAAP, consistently applied, in the Financial Statements, are Purchased Assets and will be fully collected within two (2) years of the original invoice date without resorting to litigation and without offset or counterclaim. Sellers have not accepted any prepayment or other payment for products to be delivered or services to be performed by the Business on or after the Closing Date. Sellers do not have any issued and outstanding invoices for payments due in consideration for services not yet rendered or goods not yet delivered by the Business as of the Closing Date.
Section 3.04Undisclosed Liabilities. Sellers, together with their subsidiaries, have no material Liabilities with respect to the Business, except (a) those which are adequately reflected or reserved against in the Balance Sheet as of the Balance Sheet Date, and (b) those which have been incurred in the ordinary course of business consistent with past practice since the Balance Sheet Date.
Section 3.05Absence of Certain Changes, Events, and Conditions. Since the Balance Sheet Date: (i) Sellers have conducted the Business only in the ordinary course of business consistent with past practices; and (ii) there has not been any event, change, occurrence or circumstance that has had, or is reasonably likely to have, individually or in the aggregate, a Material Adverse Effect on the Business. Except as contemplated by this Agreement or as set forth on Section 3.05 of the Disclosure Schedules, since the Balance Sheet Date, with respect to the Business, Sellers have not:
(a)incurred any Debt relating to the Business, other than trade accounts payable in the ordinary course of business consistent with past practices;
(b)changed any accounting principles, methods or practices, or the manner in which the books and records relating to the Business are kept, or its practices with regard to the booking of sales, receivables, payables or accrued expenses or materially altered its payment or collection practices;
(c)(i) granted any severance, continuation or termination pay to any employee of the Business; (ii) entered into any employment, deferred compensation or other similar agreement (or any amendment to any such existing agreement) with any employee of the Business; (iii) increased, amended, or changed compensation, bonus or other benefits payable or potentially payable to current or former employees of the Business, other than that as may be required by Law; (iv) adopted any new or changed the terms of any existing bonus, pension, insurance, health or other benefit plan applicable to the employees of the Business; or (v) represented to any current or former employee of the Business that Sellers, Buyer or any other Person would
9
continue to maintain or implement any benefit or would continue to employ such employee after the Closing Date;
(d)suffered any damage, destruction or loss (whether or not covered by insurance) to any material properties or assets of the Business or disposed of any such assets of the Business other than inventory in the ordinary course of business consistent with past practices;
(e)except in the ordinary course of business consistent with past practices and levels, granted customers of the Business any rebates, price concessions, discounts or allowances, materially altered its pricing or payment terms or agreed to any material reduction in discounts received from suppliers or any material increase in the price of raw materials;
(f)purchased, leased or otherwise acquired (whether by merger, consolidation or other business combination, purchase of securities, purchase of assets or otherwise) any material portion of the Business or assets of any other Person for use in the Business;
(g)made, changed or revoked any material Tax election, elected or changed any material method of accounting for Tax purposes, settled any Legal Proceeding in respect of Taxes or entered into any Contract in respect of Taxes with any Governmental Authority, in each case, to the extent relating to the Business;
(h)terminated or closed any facility, business or operation that is part of the Business;
(i)written up or down any material properties or assets of the Business or materially revalued any of its inventory or altered its inventory management or valuation policies or practices relating to the Business;
(j)cancelled, waived or compromised any Debt, right or claim of the Business having a value of more than $[***] (individually) or an aggregate value in excess of $[***];
(k)sold, assigned, transferred or granted any Intellectual Property used in the Business, entered into any settlement regarding the breach or infringement of any Intellectual Property used in the Business, or taken any action (or, to Sellers’ Knowledge, failed to take any action) that has resulted in, or would reasonably be likely to result in, the loss, lapse, abandonment, invalidity or enforceability of any of its Intellectual Property used in the Business;
(l)made any capital expenditures or capital additions or betterments for the Business in excess of an aggregate of $[***];
(m)made any purchase commitment outside the ordinary course of business consistent with past practice, or made any advances to any Person, other than to employees of the Business in the ordinary course of business consistent with past practice; or
(n)committed or agreed to do any of the foregoing with respect to the Business.
Section 3.06Assigned Contracts.
(a)Section 1.01(b) of the Disclosure Schedules sets forth a complete list, as of the date hereof, of each of the Assigned Contracts. (i) Each Assigned Contract is in full force and effect and is a valid and binding agreement of the Sellers, enforceable against the Sellers in accordance with its terms, subject to the Enforceability Exceptions, (ii) the Sellers are not in breach of or default under any Assigned Contract to which it is a party, and, to the Knowledge of the Sellers, no other party to any such Contract is in breach thereof or default thereunder, (iii) the Sellers have not received from any counterparty any written notice or written claim of default by the Sellers under any Assigned Contract, (iv) to the Knowledge of the Sellers, no event has occurred that, with or without notice or lapse of time or both, would result in a breach or default
10
under any Assigned Contract by the Sellers, and (v) the Sellers have not nor, to the Knowledge of the Sellers, has any other party provided or received any notice of any intention to terminate or not renew any Assigned Contract. Complete and correct copies of each Assigned Contract (including all modifications, amendments, and supplements thereto and waivers thereunder) have been made available to the Buyer. There are no disputes pending or, to the Knowledge of the Sellers, threatened under any Assigned Contract.
(b)Section 3.06(b) of the Disclosure Schedules sets forth with respect to the Business: (i) a list of top ten (10) customers for the nine months ended September 30, 2025 (collectively, the “Material Customers”), and (ii) the amount of consideration paid by each Material Customer during such periods. Sellers have not received any notice, and have no reason to believe, that any of the Material Customers has ceased, or intends to cease after the Closing, to use the goods or services of the Business or to otherwise terminate or materially reduce its relationship with the Business.
Section 3.07Title to Purchased Assets. Sellers have good and valid title to all the Purchased Assets, free and clear of Encumbrances except for Permitted Encumbrances.
Section 3.08Sufficiency of Assets. The Purchased Assets are sufficient, as of Closing, for the continued conduct of the Business in the same manner as conducted prior to the Closing and constitute all of the rights, property, and assets necessary to conduct the Business as currently conducted.
Section 3.09Tangible Personal Property. Each item of Tangible Personal Property included in the Purchased Assets is structurally sound, is in good working order and repair, and is adequate for the uses to which it is being put in the operation of the Business, and no item of Tangible Personal Property is in need of maintenance or repairs except for ordinary, routine maintenance and repairs that are not material in nature or cost.
Section 3.10Real Property.
(a)Sellers do not own or hold, and have never owned or held (i) any real property (including ground leases) used in the Business; (ii) a freehold or other ownership interest (either directly or indirectly) in any real property used in the Business; or (iii) any option or right of first refusal or first offer to acquire any real property. Section 3.10(a) of the Disclosure Schedules sets forth a complete list of all leases of real property used in the Business by Sellers (each, a “Real Property Lease,” and the real property subject to the Real Property Leases being referred to herein as the “Leased Real Property”). The Leased Real Property comprises all of the real property used in the operation of the Business.
(b)With respect to the Leased Real Property, except as set forth on Section 3.10(b) of the Disclosure Schedules: (i) Sellers are the owner and holder of all of the leasehold estates in the Leased Real Property purported to be granted by the Real Property Leases; (ii) the Real Property Leases are in full force and effect; (iii) neither the Sellers, nor any of their Affiliates, nor, to the Knowledge of the Sellers, the landlord under such lease, is in default thereunder; (iv) to the Knowledge of the Sellers, no event has occurred that, with or without notice or lapse of time or both, would result in a breach or default by the Sellers or any of its Affiliates under such lease; (v) Sellers’ possession and quiet enjoyment of the Leased Real Property has not been disturbed and there are no disputes with respect to such Real Property Lease; (vi) no security deposit or portion thereof deposited with respect to such Real Property Lease has been applied in respect of a breach of or default under such Real Property Lease that has not been redeposited in full; (vii) Sellers do not owe, or will owe in the future, any brokerage commissions or finder’s fees with respect to such Real Property Lease; and (viii) there are no Liens on the estate or interest created by such Real Property Lease.
(c)There are no parties (other than Sellers) in possession of any portion of the Leased Real Property.
11
(d)No portion of the Leased Real Property is subject to any pending condemnation or eminent domain Legal Proceeding or other Legal Proceeding by any Governmental Authority and, to the Knowledge of Sellers, there is no threatened condemnation or eminent domain Legal Proceeding or other Legal Proceeding with respect thereto.
(e)The physical condition of the Leased Real Property is free from any material defect and sufficient to permit the continued conduct of the Business, subject to the provision of usual and customary maintenance and repair performed in the ordinary course with respect to similar properties of like age and construction, and no repairs, replacements or regularly scheduled maintenance relating to any of the Leased Real Property has been deferred.
(f)Access to the Leased Real Property is from public streets and roads adjoining the Leased Real Property, is not limited or restricted and is adequate to service the Leased Real Property as currently used.
(g)All water, storm and sanitary sewer, gas, electric, telephone and drainage facilities, and all other utilities required by any Law or necessary for the current use and operation of the Leased Real Property are fully operable and are adequate to service the Leased Real Property as currently used in the Business.
(h)The classification of each parcel of Leased Real Property under applicable zoning Laws permits the use and occupancy of such parcel and the operation of the Business. Sellers’ use or occupancy of the Leased Real Property or any portion thereof or the operation of the Business is not dependent on a “permitted non-conforming use” or “permitted non-conforming structure” or similar variance, exemption or approval from any Governmental Authority.
(i)The current use and occupancy of the Leased Real Property and the operation of the Business as currently conducted thereon do not violate any easement, covenant, condition, restriction or similar provision in any instrument of record or other unrecorded agreement affecting such Leased Real Property. Sellers have not received any notice of violation of the foregoing.
(j)Except as disclosed in Section 3.10(j) of the Disclosure Schedules, on the Closing Date, Sellers will not be indebted to any contractor, laborer, mechanic, materialman, architect or engineer for work, labor or services performed or rendered, or for materials supplied or furnished, in connection with the Leased Real Property which any such person could claim a Lien against the Leased Real Property.
(k)There are no management, maintenance, service or other contracts with respect to the Leased Real Property which cannot be terminated on not more than 30 days’ notice without penalty or fee.
(l)Sellers have not received any notice that the current use and occupancy of the Leased Real Property and the operation of the Business violate any easement, covenant, condition, restriction or similar provision in any instrument of record or other unrecorded agreement affecting such Leased Real Property.
Section 3.11Intellectual Property.
(a)Section 3.11(a) of the Disclosure Schedules contains a correct, current and complete list of: (i) all Intellectual Property Registrations; (ii) all unregistered Trademarks included in the Intellectual Property Assets; (iii) all proprietary Software included in the Intellectual Property Assets; and (iv) all other Intellectual Property Assets that are used or held for use in the conduct of the Business as currently conducted or proposed to be conducted.
(b)Section 3.11(b) of the Disclosure Schedules contains a correct, current and complete list of all Intellectual Property Agreements that are material to the Business, excluding shrink-wrap, click-wrap or similar agreements for commercially available off-the-shelf software.
12
Sellers have provided Buyer with true and complete copies (or in the case of any oral agreements, a complete and correct written description) of all such Intellectual Property Agreements, including all modifications, amendments and supplements thereto and waivers thereunder. Each Intellectual Property Agreement is valid and binding on Sellers in accordance with its terms and is in full force and effect, except as such enforceability may be limited by bankruptcy, insolvency, reorganization, moratorium or similar Laws affecting creditors’ rights generally and by general principles of equity (regardless of whether enforcement is sought in a proceeding at law or in equity). Neither Sellers nor, to Sellers’ Knowledge, any other party thereto is, or is alleged to be, in breach of or default under, or has provided or received any notice of breach of, default under, or intention to terminate (including by non-renewal), any Intellectual Property Agreement.
(c)Sellers are the sole and exclusive legal and beneficial, and with respect to the Intellectual Property Registrations, record, owners of all right, title and interest in and to the Intellectual Property Assets, and have the valid and enforceable right to use all other Intellectual Property used or held for use in or necessary for the conduct of the Business as currently conducted, free and clear of Encumbrances other than Permitted Encumbrances. Sellers have entered into binding, valid and enforceable written Contracts with each current and former employee and independent contractor who is or was involved in or has contributed to the invention, creation, or development of any Intellectual Property during the course of employment or engagement with Sellers whereby such employee or independent contractor (i) acknowledges Sellers’ exclusive ownership of all Intellectual Property Assets invented, created or developed by such employee or independent contractor within the scope of his or her employment or engagement with Sellers; (ii) grants to Sellers a present, irrevocable assignment of any ownership interest such employee or independent contractor may have in or to such Intellectual Property, to the extent such Intellectual Property does not constitute a "work made for hire" under Applicable Law; and (iii) irrevocably waives any right or interest, including any moral rights, regarding such Intellectual Property, to the extent permitted by applicable Law. Sellers have provided Buyer with true and complete copies of all such Contracts. To Sellers’ Knowledge, all assignments and other instruments necessary to establish, record, and perfect Sellers’ ownership interest in the Intellectual Property Registrations have been validly executed, delivered, and filed with the relevant Governmental Authorities and authorized registrars.
(d)Neither the execution, delivery, or performance of this Agreement, nor the consummation of the transactions contemplated hereunder, will result in the loss or impairment of or payment of any additional amounts with respect to, or require the consent of any other Person in respect of, the Buyer's right to own or use any Intellectual Property Assets in the conduct of the Business as currently conducted. Immediately following the Closing, all Intellectual Property Assets will be owned or available for use by Buyer on the same or substantially similar terms as they were owned or available for use by Sellers immediately prior to the Closing.
(e)The Intellectual Property Assets are valid and enforceable, and all Intellectual Property Registrations are subsisting and in full force and effect. Sellers and their subsidiaries have taken all commercially reasonable steps to maintain and enforce the Intellectual Property Assets and to preserve the confidentiality of all Trade Secrets included in the Intellectual Property Assets, including by requiring all Persons having access thereto to execute binding, written non-disclosure agreements. All required filings and fees related to the Intellectual Property Registrations have been timely submitted with and paid to the relevant Governmental Authorities and authorized registrars in the ordinary course of business. Sellers have provided Buyer with true and complete copies of all material file histories, documents, certificates, office actions, correspondence, assignments, and other instruments relating to the Intellectual Property Registrations.
(f)The conduct of the Business as currently and formerly conducted, including the use of the Intellectual Property Assets in connection therewith, and the products, processes, and services of the Business have not infringed, misappropriated, or otherwise violated the Intellectual Property rights of any Person. To Sellers’ Knowledge, no Person has infringed, misappropriated, or otherwise violated any Intellectual Property Assets.
13
(g)There are no Actions (including any opposition, cancellation, revocation, review, or other proceeding), whether settled, pending or threatened in writing (including in the form of offers to obtain a license): (i) alleging any infringement, misappropriation, or other violation of the Intellectual Property of any Person by Sellers or any of their subsidiaries in the conduct of the Business; (ii) challenging the validity, enforceability, registrability, patentability, or ownership of any Intellectual Property Assets; or (iii) by Sellers or any other Person alleging any infringement, misappropriation, or other violation by any Person of any Intellectual Property Assets. Neither Sellers nor any of their subsidiaries is subject to any outstanding or prospective Governmental Order (including any motion or petition therefor) that does or could reasonably be expected to restrict or impair the use of any Intellectual Property Assets.
(h)In the past twelve (12) months, there has been no malfunction, failure, continued substandard performance, denial-of-service, or other cyber incident, including any cyberattack, or other impairment of the Business IT Systems that has resulted or is reasonably likely to result in disruption or damage to the Business. Sellers and each of their subsidiaries have taken all commercially reasonable steps to safeguard the confidentiality, availability, security, and integrity of the Business IT Systems, including implementing and maintaining appropriate backup, disaster recovery, and Software and hardware support arrangements.
(i)Sellers and each of their subsidiaries have complied with all Laws in all material respects and complied in all respects with all internal or publicly posted policies, notices, and statements concerning the collection, use, processing, storage, transfer, and security of personal information in the conduct of the Business. In the past twelve (12) months, neither Sellers nor any of their subsidiaries have (i) experienced any actual, alleged, or suspected data breach or other security incident involving personal information in its possession or control or (ii) been subject to or received any notice of any audit, investigation, complaint, or other Action by any Governmental Authority or other Person concerning the Company's collection, use, processing, storage, transfer, or protection of personal information or actual, alleged, or suspected violation of any applicable Law concerning privacy, data security, or data breach notification, in each case in connection with the conduct of the Business, and there are no facts or circumstances that could reasonably be expected to give rise to any such Action.
Section 3.12Employee Benefit Matters. Section 3.12 of the Disclosure Schedules contains a true and complete list of each pension, benefit, retirement, compensation, consulting, profit-sharing, deferred compensation, incentive, bonus, performance award, phantom equity, stock or stock-based, change in control, retention, severance, vacation, paid time off (PTO), medical, vision, dental, disability, welfare, Code Section 125 cafeteria, fringe-benefit and other similar agreement, plan, policy, program or arrangement (and any amendments thereto), in each case whether or not reduced to writing and whether funded or unfunded, including each "employee benefit plan" within the meaning of Section 3(3) of ERISA, whether or not tax-qualified and whether or not subject to ERISA, which is or has been maintained, sponsored, contributed to, or required to be contributed to by Sellers for the benefit of any current or former employee, officer, director, retiree, independent contractor or consultant of the Business or any spouse or dependent of such individual, or under which Sellers or any of their ERISA Affiliates have or may have any Liability, or with respect to which Buyer or any of its Affiliates would reasonably be expected to have any Liability, contingent or otherwise (as listed on Section 3.122 of the Disclosure Schedules, each, a “Benefit Plan”).
Section 3.13Employment Matters.
(a)Section 3.13(a) of the Disclosure Schedules contains a list of all persons who are employees, independent contractors or consultants of the Business as of the date hereof, including any employee who is on a leave of absence of any nature, paid or unpaid, authorized or unauthorized, and sets forth for each such individual the following: (i) name; (ii) title or position (including whether full-time or part-time); (iii) hire or retention date; (iv) current annual base compensation rate or contract fee; and (v) commission, bonus or other incentive-based compensation. Except as set forth on Section 3.13(a) of the Disclosure Schedules, as of the date hereof all compensation, including wages, commissions, bonuses, fees and other compensation, payable to all employees, independent contractors or consultants of the Business for services
14
performed on or prior to the date hereof have been paid in full, and there are no outstanding agreements, understandings or commitments of Sellers with respect to any compensation, commissions, bonuses or fees.
(b)Sellers are and have been in compliance in all material respects with all applicable Laws pertaining to employment and employment practices to the extent they relate to employees, volunteers, interns, consultants and independent contractors of the Business, including all Laws relating to labor relations, equal employment opportunities, fair employment practices, employment discrimination, harassment, retaliation, reasonable accommodation, disability rights or benefits, immigration, wages, hours, overtime compensation, child labor, hiring, promotion and termination of employees, working conditions, meal and break periods, privacy, health and safety, workers’ compensation, leaves of absence, paid sick leave and unemployment insurance. To Sellers’ Knowledge, all individuals characterized and treated by Sellers as consultants or independent contractors of the Business are properly treated as independent contractors under all applicable Laws. To Sellers’ Knowledge, all employees of the Business classified as exempt under the Fair Labor Standards Act and state and local wage and hour laws are properly classified. Sellers are in compliance with and have complied with all immigration laws, including Form I-9 requirements and any applicable mandatory E-Verify obligations. There are no Actions against Sellers pending, or to the Sellers’ Knowledge, threatened to be brought or filed in writing, by or with any Governmental Authority or arbitrator in connection with the employment of any current or former applicant, employee, consultant, volunteer, intern or independent contractor of the Business, including, without limitation, any charge, investigation or claim relating to unfair labor practices, equal employment opportunities, fair employment practices, employment discrimination, harassment, retaliation, reasonable accommodation, disability rights or benefits, immigration, wages, hours, overtime compensation, employee classification, child labor, hiring, promotion and termination of employees, working conditions, meal and break periods, privacy, health and safety, workers' compensation, leaves of absence, paid sick leave, unemployment insurance or any other employment related matter arising under applicable Laws.
(c)Sellers have no Contracts with any Governmental Authority. Sellers have not been debarred, suspended or otherwise made ineligible from doing business with the United States government or any government contractor.
Section 3.14Legal Proceedings; Governmental Orders.
(a)Except as disclosed in Section 3.14 of the Disclosure Schedules. during the past three (3) years, there have been no claims, actions, causes of action, demands, lawsuits, arbitrations, inquiries, audits, notices of violation, proceedings, litigation, citations, summons, subpoenas, or investigations of any nature, whether at law or in equity (collectively, “Actions”) pending or, to Sellers’ knowledge, threatened in writing against or by Sellers or any of their subsidiaries: (i) relating to or affecting the Business, the Purchased Assets, or the Assumed Liabilities; or (ii) that challenge or seek to prevent, enjoin, or otherwise delay the transactions contemplated by this Agreement. To Sellers’ Knowledge, no event has occurred or circumstances exist that may give rise to, or serve as a basis for, any such Action.
(b)There are no outstanding Governmental Orders against, relating to, or affecting the Business or the Purchased Assets. No event has occurred or circumstances exist that may constitute or result in (with or without notice or lapse of time) a violation of any such Governmental Order.
Section 3.15Compliance with Laws; Permits.
(a)During the past three (3) years, Sellers have been in compliance in all material respects with all Laws applicable to the conduct of the Business as currently conducted or the ownership and use of the Purchased Assets.
15
(b) (i) The Sellers have all material Permits that are necessary to conduct the Business as currently conducted, (ii) all such Permits are in full force and effect, (iii) the Business is not being conducted in violation or default of such Permits and (iv) the Sellers are not in receipt of any written notification that any Governmental Authority is threatening to revoke any such Permit.
Section 3.16Environmental Matters.
(a)The Sellers are in compliance with all applicable Environmental Laws in all material respects to the extent relating to the Business and the premises leased under the Assigned Real Property Lease.
(b)The Sellers have obtained and is in compliance with all material Environmental Permits to conduct the Business as currently conducted;
(c)Since the Balance Sheet Date, the Sellers have not received a written order, complaint or penalty in connection with the Business that alleges a material violation of any Environmental Law, the substance of which has not been resolved;
(d)As of the date hereof, there is no judicial proceeding pending or, to the Knowledge of the Sellers, threatened by a third party against the Sellers in connection with the Business alleging a material liability under any Environmental Law; and
(e)As of the date hereof, the Sellers are not investigating or remediating releases of Hazardous Substances at any of the real property subject to the Assigned Real Property Lease pursuant to any Environmental Law.
Section 3.17Taxes.
(a)All material Tax Returns required to have been filed with respect to the Purchased Assets have been timely filed (taking into account any extension of time to file granted or obtained), and all such Tax Returns are correct and complete in all material respects;
(b)All Taxes shown to be payable on such Tax Returns have been paid;
(c)There are no Encumbrances (other than Permitted Encumbrances) for Taxes with respect to the Purchased Assets;
(d)Sellers have not waived any statute or period of limitations with respect to any Tax or agreed, or been requested by any Governmental Authority to agree, to any extension of time with respect to any Tax due with respect to the Purchased Assets or the Business for any Tax period that remains open;
(e)Sellers have (with respect to the Business and the Purchased Assets) complied with all information reporting and back up withholding requirements, and have maintained all required records with respect thereto, in connection with amounts paid or owing to any employee, customer, creditor, member, stockholder, independent contractor, or other third party; and
(f)There is no claim with respect to the Purchased Assets for any material Taxes, and no assessment, deficiency, or adjustment has been asserted, proposed, or, to the Knowledge of the Sellers, threatened with respect to any material Taxes or material Tax Returns with respect to the Purchased Assets. No material Tax Proceedings or administrative or judicial proceedings are being conducted, pending or, to the Knowledge of the Sellers, threatened with respect to the Purchased Assets.
Section 3.18Investment in Shares.
16
(a)Each Receiving Seller is an accredited investor as defined in Rule 501(a) of Regulation D promulgated under the Securities Act of 1933, as amended.
(b)The Consideration Shares will be acquired for investment for the Receiving Sellers’ own account, not as a nominee or agent, and not with a view to the resale or distribution of any part thereof, and Receiving Sellers have no present intention of selling, granting any participation in, or otherwise distributing the same. By executing this Agreement, Sellers further represent that Receiving Sellers do not presently have any contract, undertaking, agreement or arrangement with any Person to sell, transfer or grant participations to such Person or to any third Person, with respect to any portion of the Consideration Shares. Receiving Sellers have not been formed for the specific purpose of acquiring the Consideration Shares.
(c)Sellers and Receiving Sellers have had an opportunity to discuss Buyer Parent’s business, management, financial affairs and the terms and conditions of the offering of the Consideration Shares with Buyer Parent’s management and have had an opportunity to review Buyer Parent’s facilities.
(d)Receiving Sellers understand that the Consideration Shares have not been, and will not be, registered under the Securities Act, by reason of a specific exemption from the registration provisions of the Securities Act which depends upon, among other things, the bona fide nature of the investment intent and the accuracy of Sellers’ representations as expressed herein. Receiving Sellers understand that the Consideration Shares are “restricted securities” under applicable U.S. federal and state securities laws and that, pursuant to these laws, Receiving Sellers must hold the Consideration Shares indefinitely unless they are registered with the Securities and Exchange Commission and qualified by state authorities, or an exemption from such registration and qualification requirements is available. Receiving Sellers acknowledge that Buyer Parent has no obligation to register or qualify the Consideration Shares. Receiving Sellers further acknowledge that if an exemption from registration or qualification is available, it may be conditioned on various requirements including, but not limited to, the time and manner of sale, the holding period for the Consideration Shares, and on requirements relating to Buyer Parent which are outside of Receiving Sellers’ control, and which Buyer Parent is under no obligation and may not be able to satisfy. Receiving Sellers understand that this offering is not intended to be part of the public offering, and that Receiving Sellers will not be able to rely on the protection of Section 11 of the Securities Act.
(e)Receiving Sellers understand that no public market now exists for the Consideration Shares, and that Buyer Parent has made no assurances that a public market will ever exist for the Consideration Shares.
(f)Receiving Sellers understand that the Consideration Shares and any securities issued in respect of or exchange for the Consideration Shares, may be notated with one or all of the following legends:
(i)“THE SHARES REPRESENTED HEREBY HAVE NOT BEEN REGISTERED UNDER THE SECURITIES ACT OF 1933, AND HAVE BEEN ACQUIRED FOR INVESTMENT AND NOT WITH A VIEW TO, OR IN CONNECTION WITH, THE SALE OR DISTRIBUTION THEREOF. NO SUCH TRANSFER MAY BE EFFECTED WITHOUT AN EFFECTIVE REGISTRATION STATEMENT RELATED THERETO OR AN OPINION OF COUNSEL IN A FORM SATISFACTORY TO THE COMPANY THAT SUCH REGISTRATION IS NOT REQUIRED UNDER THE SECURITIES ACT OF 1933.”
(ii)“THE SALE, PLEDGE, HYPOTHECATION OR TRANSFER OF THE SECURITIES REPRESENTED BY THIS CERTIFICATE IS SUBJECT TO, AND IN CERTAIN CASES PROHIBITED BY, THE TERMS AND CONDITIONS OF A CERTAIN SIXTH AMENDED AND RESTATED RIGHT OF FIRST REFUSAL AND CO-SALE AGREEMENT BY AND AMONG THE STOCKHOLDER, THE CORPORATION AND CERTAIN OTHER HOLDERS OF STOCK OF THE
17
CORPORATION. COPIES OF SUCH AGREEMENT MAY BE OBTAINED UPON WRITTEN REQUEST TO THE SECRETARY OF THE CORPORATION.”
(iii)“THE SHARES EVIDENCED HEREBY ARE SUBJECT TO A SIXTH AMENDED AND RESTATED VOTING AGREEMENT, AS MAY BE AMENDED FROM TIME TO TIME, (A COPY OF WHICH MAY BE OBTAINED UPON WRITTEN REQUEST FROM THE COMPANY), AND BY ACCEPTING ANY INTEREST IN SUCH SHARES THE PERSON ACCEPTING SUCH INTEREST SHALL BE DEEMED TO AGREE TO AND SHALL BECOME BOUND BY ALL THE PROVISIONS OF THAT SIXTH AMENDED AND RESTATED VOTING AGREEMENT, INCLUDING CERTAIN RESTRICTIONS ON TRANSFER AND OWNERSHIP SET FORTH THEREIN.”
(iv)Any legend required by the securities laws of any state to the extent such laws are applicable to the Consideration Shares represented by the certificate, instrument, or book entry so legended.
Section 3.19Brokers. No broker, finder, or investment banker is entitled to any brokerage, finders, or other fee or commission in connection with the transactions contemplated by this Agreement or any other Transaction Document based upon arrangements made by or on behalf of Sellers.
Section 3.20Privacy and Security.
(a)For the last three (3) years, the Sellers have been in material compliance with all Laws applicable to the operation of the Business related to the collection, use, storage, disclosure, and protection, transfer, sharing, or other processing, of Personal Data (collectively, “Information Security and Data Privacy Laws”).
(b)There are no pending, and to Sellers’ Knowledge, threatened Actions against the Sellers alleging either (i) a violation of any Person’s privacy, personal or confidential rights under the Sellers’ information security or data privacy practices or any Information Security and Data Privacy Laws, or (ii) breach of any information security, including, but not limited to, a network intrusion involving Personal Data or data breach of the Systems (as defined below). The Systems are in good working condition to effectively perform all computing, information technology and data processing operations necessary for the operation of the Business and are free from any vulnerabilities or defects that would reasonably be expected to result in an actual or potential partial or complete loss of control of any assets of the Sellers.
(c)The Sellers have established and maintained administrative, technical and physical safeguards required by the Information Security and Data Privacy Laws which are designed to safeguard the security, confidentiality and integrity of the computer, information technology and data processing systems and services used by the Sellers in the operation of the Business to process Personal Data (“Systems”). The Systems are sufficient for the current needs of the Sellers in the operation of the Business, including as to capacity, scalability and ability to process current historical peak volumes in a timely manner. The Systems are in good working condition to effectively perform all computing, information technology and data processing operations necessary for the operation of the Business, and are free from any vulnerabilities or defects that would reasonably be expected to result in an actual or potential partial or complete loss of control of any assets of the Sellers.
(d)For the past three (3) years, (i) there has been no material unauthorized access to the Systems or any networks, computers, computer software, firmware, middleware, servers, workstations, routers, hubs, switches, data communications lines and other information technology equipment or systems of the Sellers in the operation of the Business (“IT Assets”), (ii) there has been no unauthorized access to or processing of any Personal Data processed by or on behalf of the Sellers in the operation of the Business, and (iii) the IT Assets and the information and data processed or stored by the Sellers in connection with the operation of its Business have not been corrupted or compromised.
18
Section 3.21Affiliate Transactions. Except as set forth on Section 3.21 of the Disclosure Schedules, neither Sellers nor any of their Affiliates have any Contract, arrangement or understanding with the Business or with respect to the Purchased Assets, the Assumed Liabilities or the Business, other than (a) Contracts entered into in the ordinary course of business on arm’s-length terms, and (b) Contracts that will be terminated at or prior to the Closing without further liability to Buyer or the Business; and (iii) no Affiliate of Sellers has any right, title or interest in any property or asset used in or necessary for the conduct of the Business.
Article IV
REPRESENTATIONS AND WARRANTIES OF BUYER
REPRESENTATIONS AND WARRANTIES OF BUYER
Buyer represents and warrants to Sellers that the statements contained in this ARTICLE IV are true and correct as of the date hereof.
Section 4.01Organization and Authority of Buyer. Buyer is a limited liability company duly organized, validly existing, and in good standing under the Laws of the State of Delaware. Buyer has full corporate power and authority to enter into this Agreement and the other Transaction Documents to which Buyer is a party, to carry out its obligations hereunder and thereunder, and to consummate the transactions contemplated hereby and thereby. The execution and delivery by Buyer of this Agreement and any other Transaction Document to which Buyer is a party, the performance by Buyer of its obligations hereunder and thereunder, and the consummation by Buyer of the transactions contemplated hereby and thereby have been duly authorized by all requisite corporate action on the part of Buyer. This Agreement and the Transaction Documents constitute legal, valid, and binding obligations of Buyer enforceable against Buyer in accordance with their respective terms.
Section 4.02No Conflicts; Consents.
The execution, delivery, and performance by Buyer of this Agreement and the other Transaction Documents to which it is a party, and the consummation of the transactions contemplated hereby and thereby, do not and will not: (a) violate or conflict with any provision of the certificate of incorporation, by-laws, or other organizational documents of Buyer; (b) violate or conflict with any provision of any Law or Governmental Order applicable to Buyer; or (c) require the consent, notice, declaration, or filing with or other action by any Person or require any permit, license, or Governmental Order.
Section 4.03Brokers. No broker, finder, or investment banker is entitled to any brokerage, finders, or other fee or commission in connection with the transactions contemplated by this Agreement or any other Transaction Document based upon arrangements made by or on behalf of Buyer.
Section 4.04Legal Proceedings. There are no Actions pending or, to Buyer’s knowledge, threatened against or by Buyer that challenge or seek to prevent, enjoin, or otherwise delay the transactions contemplated by this Agreement. No event has occurred or circumstances exist that may give rise to, or serve as a basis for, any such Action.
Article V
REPRESENTATIONS AND WARRANTIES OF BUYER PARENT
REPRESENTATIONS AND WARRANTIES OF BUYER PARENT
Buyer Parent represents and warrants to Sellers that the statements contained in this Article V are true and correct as of the date hereof.
Section 5.01Organization and Authority of Buyer Parent. Buyer Parent is a corporation duly organized, validly existing, and in good standing under the Laws of the State of Delaware. Buyer Parent has full corporate power and authority to enter into this Agreement and the other Transaction Documents to which Buyer Parent is a party, to carry out its obligations hereunder and thereunder, and to consummate the transactions contemplated hereby and thereby. The execution and delivery by Buyer Parent of this Agreement and any other Transaction Document to which Buyer Parent is a party, the performance by Buyer Parent of its obligations hereunder and thereunder, and the consummation by Buyer Parent of the transactions contemplated hereby and thereby have been duly authorized by all
19
requisite corporate action on the part of Buyer Parent. This Agreement and the Transaction Documents constitute legal, valid, and binding obligations of Buyer Parent enforceable against Buyer Parent in accordance with their respective terms.
Section 5.02No Conflicts; Consents. The execution, delivery, and performance by Buyer Parent of this Agreement and the other Transaction Documents to which it is a party, and the consummation of the transactions contemplated hereby and thereby, do not and will not: (a) violate or conflict with any provision of the certificate of incorporation, by-laws, or other organizational documents of Buyer Parent; (b) violate or conflict with any provision of any Law or Governmental Order applicable to Buyer Parent; or (c) require the consent, notice, declaration, or filing with or other action by any Person or require any permit, license, or Governmental Order.
Section 5.03Valid Issuance of Consideration Shares. The Consideration Shares, when issued and delivered in accordance with the terms and for the consideration set forth in this Agreement, will be validly issued, fully paid and nonassessable and free of restrictions on transfer other than restrictions on transfer under this Agreement, Buyer Parent’s bylaws, Buyer Parent’s Right of First Refusal and Co-Sale Agreement, Buyer Parent’s Voting Agreement, applicable state and federal securities laws, and liens or encumbrances created by or imposed by Sellers.
Section 5.04Brokers. No broker, finder, or investment banker is entitled to any brokerage, finders, or other fee or commission in connection with the transactions contemplated by this Agreement or any other Transaction Document based upon arrangements made by or on behalf of Buyer Parent.
Section 5.05Legal Proceedings. There are no Actions pending or, to Buyer Parent’s knowledge, threatened against or by Buyer Parent that challenge or seek to prevent, enjoin, or otherwise delay the transactions contemplated by this Agreement. No event has occurred or circumstances exist that may give rise to, or serve as a basis for, any such Action.
Article VI
COVENANTS
COVENANTS
Section 6.01Confidentiality. From and after the Closing, each party shall, and shall cause its Affiliates to, hold, and shall use its reasonable best efforts to cause it’s or their respective directors, officers, employees, consultants, counsel, accountants, and other agents (“Representatives”) to hold, in confidence any and all information, whether written or oral, concerning the other party and/or their respective business, except to the extent that it can show that such information: (a) is generally available to and known by the public through no fault of disclosing party, any of its Affiliates, or their respective Representatives; or (b) is lawfully acquired by the disclosing party, any of its Affiliates, or their respective Representatives from and after the Closing from sources which are not prohibited from disclosing such information by a legal, contractual, or fiduciary obligation. If any party or any of its Affiliates or their respective Representatives are compelled to disclose any information by Governmental Order or Law, the disclosing party shall promptly notify the other party in writing and shall disclose only that portion of such information which is legally required to be disclosed, provided that the disclosing party shall use commercially reasonable efforts to obtain as promptly as possible an appropriate protective order or other reasonable assurance that confidential treatment will be accorded such information.
Section 6.02Books and Records.
(a)In order to facilitate the resolution of any claims made by or against or incurred by Buyer after the Closing, or for any other reasonable purpose, for a period of five (5) years following the Closing, Sellers shall:
(i)retain the books and records (including personnel files) of Sellers which relate to the Business and its operations for periods prior to the Closing; and
(ii)upon reasonable notice, afford Buyer Parent’s Representatives reasonable access (including the right to make, at Buyer's expense, photocopies), during normal business hours, to such books and records.
20
(b)Sellers shall not be obligated to provide Buyer with access to any books or records (including personnel files) pursuant to this Section 6.02 where such access would violate any Law.
(c)Notwithstanding the transfer and sale to the Buyer under Section 1.01 of this Agreement, Buyer agrees to grant to Sellers’ auditors reasonable access, during normal business hours and upon reasonable prior written notice, to any books and records of the Business included in the Purchased Assets covering the period up to and as of the date of this Agreement as the auditors may reasonably require in connection with their FY 2025 audit of the Sellers.
Section 6.03Public Announcements. Unless otherwise required by applicable Law, no party to this Agreement shall make any public announcements in respect of this Agreement or the transactions contemplated hereby without the prior written consent of the other party (which consent shall not be unreasonably withheld or delayed), and the parties shall cooperate as to the timing and contents of any such announcement.
Section 6.04Bulk Sales Laws. The parties hereby waive compliance with the provisions of any bulk sales, bulk transfer, or similar Laws of any jurisdiction that may otherwise be applicable with respect to the sale of any or all of the Purchased Assets to Buyer.
Section 6.05Wrong Pocket. To the extent that, following the Closing, Sellers or any of their Affiliates receives any payment or other amount that properly belongs to Buyer (including, without limitation, any accounts receivable or other amounts in respect of services performed by or on behalf of Buyer after the Closing), Sellers shall, within ten (10) Business Days after receipt thereof, remit (or cause to be remitted) such payment or amount to Buyer. Conversely, to the extent that Buyer or any of its Affiliates receives any payment or other amount that properly belongs to Sellers (including, without limitation, any accounts receivable or other amounts in respect of services performed by or on behalf of Sellers prior to the Closing), Buyer shall, within ten (10) Business Days after receipt thereof, remit (or cause to be remitted) such payment or amount to Sellers. Each of Buyer and Sellers agrees to endorse and deliver to the other party any checks or other instruments of payment that it may receive representing any such payments promptly after its receipt thereof.
Section 6.06Transfer Taxes. All sales, use, registration, and other such Taxes and fees (including any penalties and interest) incurred in connection with this Agreement and the other Transaction Documents, if any, shall be borne and paid by Sellers when due. Sellers shall, at its own expense, timely file any Tax Return or other document with respect to such Taxes or fees (and Buyer and Buyer Parent shall cooperate with respect thereto as necessary).
Section 6.07Shared Contracts. For a period of six months following the Closing, upon the Sellers’ or the Buyer’s reasonable request, with respect to any Shared Contract, the Sellers and the Buyer shall use, and shall cause their respective Affiliates to use, commercially reasonable efforts to cause the counterparty to such Shared Contract to enter into a new contract with the Buyer or Buyer Parent, as applicable, on terms substantially similar in all material respects to those contained in such Shared Contract, in order for the Business to receive the applicable claims, rights and benefits and bear the applicable burdens and obligations under such Shared Contract (each such new contract, a “New Contract”), it being understood that a New Contract will not necessarily contain the same terms as the Shared Contract to which it relates. Until such time as such a New Contract is executed, the Buyer and the Sellers shall use commercially reasonable efforts to secure an alternative arrangement reasonably satisfactory to both parties under which the Business would, in compliance with applicable Law, obtain the benefits and bear the burdens associated with the applicable Shared Contract such that the Business would be placed in a substantially similar position as if such a New Contract had been executed or such claims, rights, benefits, burdens and obligations had been so assigned.
Section 6.08Exclusivity; ROFR. For a period of three (3) years following the Closing Date, the Sellers grant to Buyer Parent: (i) exclusive Distribution Rights with respect to all IM8 products (including any related products of IM8 sold through an Affiliate of IM8, including IM8 US) in [***] (collectively, the “Exclusive Distribution Geographies”); and (ii) the First Right of Refusal with respect to any Distribution Rights relating to IM8’s products (including any related products sold through any
21
Affiliate of IM8, including IM8 US) for any geography outside of the Exclusive Distribution Geographies, subject to Buyer’s Parent’s satisfaction of the Expansion Criteria. For a period of three (3) years following the Closing Date, the Sellers grant to Buyer Parent Exclusive Parcel Shipping Rights for all IM8 products (including any related products of IM8 sold through an Affiliate of IM8, including IM8 US), subject to Buyer’s Parent’s satisfaction of the Expansion Criteria for any location that is not currently contemplated in the IM8 Agreement. For a period of three (3) years following the Closing Date, the Sellers shall not, and shall cause their Affiliates not to, directly or indirectly: (i) grant, license, or otherwise authorize any third party to perform any Distribution Rights or Parcel Shipping Rights in violation of this Section 6.08; (ii) perform or engage in any Distribution Rights in the Exclusive Distribution Geographies or Parcel Shipping Rights in violation of this Section 6.08; or (iii) solicit, encourage, or assist any third party in performing such rights. Any breach of this Section 6.08 shall constitute a material breach of this Agreement. For the avoidance of doubt, the exclusivity granted in this Section 6.08 shall not restrict the Sellers or their Affiliates from directly performing logistics, fulfillment, warehousing, and distribution services themselves in Hong Kong, as well as arranging for transportation and delivery of Parcels for themselves from Hong Kong to territories within Asia Pacific.
Section 6.09Covenant to Amend IM8 Agreement. If Buyer Parent satisfies the Expansion Criteria with respect to any proposed additional location(s) in accordance with Section 6.08, then the Buyer Parent and IM8 (and the Sellers, as applicable) shall negotiate in good faith and shall execute and deliver (and shall cause their applicable Affiliates to execute and deliver) an amendment to the IM8 Agreement reflecting such expansion to such location(s) on terms consistent with this Agreement.
Section 6.10Branding; Introductions. Following the Closing and so long as the IM8 Agreement (or any successor agreement) is in effect: (i) the parties will work in good faith on co-branding and joint marketing announcements on a quarterly basis with respect to the Exclusive Distribution Geographies; (ii) the parties will utilize Buyer Parent’s Order Management System (known as [***]) for all connectivity, order routing, and account maintenance relating to the operation of the IM8’s business for all expansion opportunities that are not currently contemplated in the IM8 Agreement, and IM8 will pay a $[***] per order fee, and (iii) the Sellers will introduce Buyer Parent to known co-manufacturers that may be interested in Buyer Parent’s services.
Section 6.11Further Assurances. Following the Closing, each of the parties hereto shall, and shall cause their respective Affiliates to, execute and deliver such additional documents, instruments, conveyances, and assurances and take such further actions as may be reasonably required to carry out the provisions hereof and give effect to the transactions contemplated by this Agreement and the other Transaction Documents, including reasonable cooperation and assistance (without charge) in the transfer of the Purchased Assets to Buyer (including access, know-how, information, software, processes, and other assets to the extent reasonably requested by Buyer).
Article VII
INDEMNIFICATION
INDEMNIFICATION
Section 7.01Survival. Subject to the limitations and other provisions of this Agreement, the representations and warranties contained herein shall survive the Closing and shall remain in full force and effect until the date that is eighteen (18) months from the Closing Date. Notwithstanding the foregoing, the representations and warranties in Section 3.01, Section 3.02, Section 3.07, Section 3.08, Section 3.10, Section 3.17, and Section 3.17 (collectively, the “Fundamental Representations”), shall survive for a period equal to the applicable statute of limitations plus six (6) months. All covenants of the parties contained herein shall survive the Closing indefinitely or for the period explicitly specified therein. Notwithstanding the foregoing, any claims asserted in good faith with reasonable specificity (to the extent known at such time) and in writing by notice from the non-breaching party to the breaching party prior to the expiration date of the applicable survival period shall not thereafter be barred by the expiration.
Section 7.02Indemnification by Sellers. Subject to the other terms and conditions of this ARTICLE VII, from and after Closing, Sellers shall jointly and severally indemnify and defend each of Buyer, Buyer Parent and their respective Affiliates and each of their respective Representatives (collectively, the “Buyer Indemnitees”) against, and shall hold each of them harmless from and against,
22
any and all Losses, incurred or sustained by, or imposed upon, the Buyer Indemnitees based upon, arising out of, or with respect to:
(a)any inaccuracy in or breach of any of the representations or warranties of Sellers contained in this Agreement, any other Transaction Document, or any schedule, certificate, or exhibit related thereto;
(b)any breach or non-fulfillment of any covenant, agreement, or obligation to be performed by Sellers pursuant to this Agreement, any other Transaction Document, or any schedule, certificate, or exhibit related thereto;
(c)any Excluded Asset or any Excluded Liability; or
(d)any Third-Party Claim based upon, resulting from, or arising out of the business, operations, properties, assets, or obligations of Sellers or any of their Affiliates (other than the Purchased Assets or Assumed Liabilities) conducted, existing, or arising on or prior to the Closing Date.
Section 7.03Indemnification by Buyer. Subject to the other terms and conditions of this ARTICLE VII, from and after Closing, Buyer shall indemnify and defend each of Sellers and their Affiliates and their respective Representatives (collectively, the “Sellers Indemnitees”) against, and shall hold each of them harmless from and against any and all Losses incurred or sustained by, or imposed upon, the Sellers Indemnitees based upon, arising out of, or with respect to:
(a)Any inaccuracy in or breach of any of the representations or warranties of Buyer contained in this Agreement, any other Transaction Document, or any schedule, certificate, or exhibit related thereto;
(b)any breach or non-fulfillment of any covenant, agreement, or obligation to be performed by Buyer pursuant to this Agreement, any other Transaction Document, or any schedule, certificate, or exhibit related thereto; or
(c)any Assumed Liability.
Section 7.04Certain Limitations. The indemnification provided for in Section 7.02 and Section 7.03 shall be subject to the following limitations:
(a)Except in the cases of fraud, intentional misrepresentation, or any breach of Fundamental Representations, the aggregate amount of all Losses for which an Indemnifying Party shall be liable pursuant to Section 7.02(a) or Section 7.03(a) shall not exceed $[***] (the “General Cap”).
(b)Except in the cases of fraud or intentional misrepresentation, or claims arising under Sections 7.02(c) and (d), the aggregate amount of all Losses for which an Indemnifying Party shall be liable pursuant to Section 7.02 or Section 7.03 shall not exceed an amount equal to the Purchase Price (the “Purchase Price Cap”).
(c)Basket. No claim for indemnification shall be made unless and until the aggregate amount of all Losses incurred by the Buyer Indemnitees exceeds $[***] (the “Basket”), in which case the Buyer Indemnitees shall be entitled to indemnification for all such Losses from the first dollar.
Section 7.05Indemnification Payments; Offset; Transfer Restrictions.
(a)Except as set forth herein, any amounts payable by Sellers to any Buyer Indemnitee under this Article VII shall be satisfied against the Consideration Shares first; provided, that such amounts may, in lieu thereof, be satisfied in cash if and only to the extent the
23
Seller’s Parent and Buyer’s Parent mutually agree in writing. Any indemnification payment satisfied by Consideration Shares shall be made, at Buyer’s election, either by (i) the return and cancellation of Consideration Shares previously issued to Sellers, with the number of such Consideration Shares determined by dividing the amount of such Losses by the Buyer Parent Share Value of a share of Common Stock as of the date of issuance of the applicable Consideration Shares, or (ii) offset against the Contingent Tranche Value associated with a specific Tranche that would otherwise become issuable to Sellers pursuant to Section 1.04 after the date of determination of such Losses, with the particular Contingent Tranche Value (and corresponding issuance of Contingent Shares) to be reduced designated by Buyer in its sole discretion. For purposes of clause (i), the return and cancellation of Consideration Shares shall be applied on a first-in, first-out basis, such that the earliest issued Consideration Shares shall be deemed to be returned and cancelled first, followed in chronological order by subsequently issued Consideration Shares. In the event Buyer elects satisfaction pursuant to clause (i), Sellers shall promptly (and in any event within ten (10) Business Days following notice from Buyer) deliver to Buyer Parent duly executed stock powers and such other instruments of transfer as Buyer Parent may reasonably request in order to effectuate the transfer and cancellation of such shares, free and clear of all Encumbrances (other than restrictions under applicable securities Laws); provided, that if Sellers fail to execute and deliver such stock powers and other instruments in a timely manner, Sellers hereby irrevocably appoint Buyer Parent and any officer of Buyer Parent, acting severally, as Sellers’ attorney-in-fact, coupled with an interest and irrevocable, to execute and deliver any such stock powers or instruments and to update Buyer Parent’s books and records to reflect such transfer and cancellation without further action by Sellers. To the extent Sellers have transferred or disposed of any such Consideration Shares and are unable to deliver them for cancellation, Sellers shall instead satisfy the Loss in cash in an amount equal to such Loss.
(b)Any Losses arising out of or related to any Excluded Liability, Excluded Asset, or fraud may be satisfied in accordance with Section 7.05(a).
(c)Until the longest survival period set forth in Section 7.01 has expired, Sellers shall not sell, transfer, assign, pledge, hypothecate or otherwise dispose of (including by operation of law) any Consideration Shares, except (i) with the prior written consent of Buyer Parent, or (ii) to the extent necessary to satisfy any obligations under this Article VII. Any attempted transfer in violation of this section shall be null and void.
Section 7.06Indemnification Procedures. Whenever any claim shall arise for indemnification hereunder, the party entitled to indemnification (the “Indemnified Party”) shall promptly provide written notice of such claim to the other party (the “Indemnifying Party”). Such notice by the Indemnified Party shall: (a) describe the claim in reasonable detail; (b) include copies of all material written evidence thereof; and (c) indicate the estimated amount, if reasonably practicable, of the Losses that has been or may be sustained by the Indemnified Party. In connection with any claim giving rise to indemnity hereunder resulting from or arising out of any action, suit or proceeding by a Person who is not a party to this Agreement (a “Third-Party Claim”), the Indemnifying Party, at its sole cost and expense and upon written notice to the Indemnified Party, may assume the defense of any such Third-Party Claim with counsel reasonably satisfactory to the Indemnified Party. The Indemnified Party shall be entitled to participate in the defense of any such Third-Party Claim, with its counsel and at its own cost and expense, subject to the Indemnifying Party’s right to control the defense thereof. If the Indemnifying Party does not assume the defense of any such Third-Party Claim, the Indemnified Party may, but shall not be obligated to, defend against such Third-Party Claim at the Indemnifying Party’s expense and in such manner as it may deem appropriate, including settling such Third-Party Claim, after giving notice of it to the Indemnifying Party, on such terms as the Indemnified Party may deem appropriate and no action taken by the Indemnified Party in accordance with such defense and settlement shall relieve the Indemnifying Party of its indemnification obligations herein provided with respect to any damages resulting therefrom. The Sellers and the Buyer shall cooperate with each other in all reasonable respects in connection with the defense of any claim, including: (i) making available records relating to such claim; and (ii) furnishing, without expense (other than reimbursement of actual out-of-pocket expenses) to the defending party, management employees of the non-defending party as may be reasonably necessary for the preparation of the defense of such claim. The Indemnifying Party shall not settle any Third-Party
24
Claim without the Indemnified Party’s prior written consent (which consent shall not be unreasonably withheld, conditioned or delayed).
Section 7.07Exclusive Remedies. Except as set forth in Section 8.10 and Section 8.11 hereto, the parties acknowledge and agree that from and after Closing their sole and exclusive remedy with respect to any and all claims (other than claims of fraud against a party hereto committing fraud) for any breach of any representation, warranty, covenant, agreement or obligation set forth herein or otherwise relating to the subject matter of this Agreement, shall be pursuant to the indemnification provisions set forth in this Article VII. Nothing in this Section 7.07 shall limit any Person’s right to seek and obtain any equitable relief under Section 8.10 or Section 8.11 to which any Person shall be entitled or to pursue a claim of fraud against a party hereto committing fraud.
Article VIII
MISCELLANEOUS
MISCELLANEOUS
Section 8.01Expenses. All costs and expenses incurred in connection with this Agreement and the transactions contemplated hereby shall be paid by the party incurring such costs and expenses.
Section 8.02Notices. All notices, claims, demands, and other communications hereunder shall be in writing and shall be deemed to have been given: (a) when delivered by hand (with written confirmation of receipt); (b) when received by the addressee if sent by a nationally recognized overnight courier (receipt requested); (c) on the date sent by email of a PDF document (with confirmation of transmission) if sent during normal business hours of the recipient, and on the next business day if sent after normal business hours of the recipient, or (d) on the third day after the date mailed, by certified or registered mail, return receipt requested, postage prepaid. Such communications must be sent to the respective parties at the following addresses (or at such other address for a party as shall be specified in a notice given in accordance with this Section 8.02):
If to Sellers: | Prenetics Global Limited Unit 703-706, K11 Atelier 728 King's Road Quarry Bay, Hong Kong. Attention: [***] Email: [***] | ||||
If to Buyer or Buyer Parent: | [***] [***] [***] Attention: [***] Email: [***] | ||||
with a copy to: | [***] [***] [***] Attention: [***] Email: [***] | ||||
Section 8.03Interpretation; Headings. This Agreement shall be construed without regard to any presumption or rule requiring construction or interpretation against the party drafting an instrument or causing any instrument to be drafted. The headings in this Agreement are for reference only and shall not affect the interpretation of this Agreement. The definitions of the terms herein shall apply equally to the singular and plural forms of the terms defined. Whenever the context may require, any pronoun shall include the corresponding masculine, feminine and neuter forms. The words “include”, “includes” and “including” shall be deemed to be followed by the phrase “without limitation”. Except as set forth in
25
Exhibit A, References to the “knowledge” of a natural person shall mean the actual knowledge of such person and references to the “knowledge” of any entity shall mean the actual knowledge of the officers, directors, managers, managing members and/or partners of such entity, as applicable, after reasonable inquiry of their direct reports responsible for the applicable subject matters and any relevant books and records.
Section 8.04Severability. If any term or provision of this Agreement is invalid, illegal, or unenforceable in any jurisdiction, such invalidity, illegality, or unenforceability shall not affect any other term or provision of this Agreement.
Section 8.05Entire Agreement. This Agreement (including any exhibits and schedules thereto) and the other Transaction Documents (including any exhibits and schedules thereto) constitute the sole and entire agreement of the parties to this Agreement with respect to the subject matter contained herein and therein, and supersede all prior and contemporaneous understandings and agreements, both written and oral, with respect to such subject matter. In the event of any inconsistency between the statements in the body of this Agreement and those in the other Transaction Documents, the Exhibits, and the Disclosure Schedules (other than an exception expressly set forth as such in the Disclosure Schedules), the statements in the body of this Agreement will control.
Section 8.06Successors and Assigns. This Agreement shall be binding upon and shall inure to the benefit of the parties hereto and their respective successors and permitted assigns. Neither party may assign its rights or obligations hereunder without the prior written consent of the other party, which consent shall not be unreasonably withheld or delayed. Any purported assignment in violation of this Section shall be null and void. No assignment shall relieve the assigning party of any of its obligations hereunder. Any transferee of any Consideration Shares shall become a party to the Buyer Parent’s Right of First Refusal and Co-Sale Agreement and Buyer Parent’s Voting Agreement. Upon any transfer of the Consideration Shares, the transferee shall, as a condition to such transfer, execute a joinder to this Agreement for purposes of (i) the first sentence of Section 1.04(b), and (ii) Article VII, whereby it agrees to be bound by the terms of such sections as if an original party hereto.
Section 8.07Amendment and Modification; Waiver. This Agreement may only be amended, modified, or supplemented by an agreement in writing signed by each party hereto. No waiver by any party of any of the provisions hereof shall be effective unless explicitly set forth in writing and signed by the party so waiving. No failure to exercise, or delay in exercising, any right or remedy arising from this Agreement shall operate or be construed as a waiver thereof; nor shall any single or partial exercise of any right or remedy hereunder preclude any other or further exercise thereof or the exercise of any other right or remedy.
Section 8.08Governing Law; Submission to Jurisdiction; Waiver of Jury Trial.
(a)All matters arising out of or relating to this Agreement shall be governed by and construed in accordance with the internal laws of the State of Delaware without giving effect to any choice or conflict of law provision or rule (whether of the State of Delaware or any other jurisdiction). Any legal suit, action, proceeding, or dispute arising out of or related to this Agreement, the other Transaction Documents, or the transactions contemplated hereby or thereby may be instituted in the federal courts of the United States of America or the courts of the State of Delaware in each case located in the city of Wilmington and county of New Castle, and each party irrevocably submits to the exclusive jurisdiction of such courts in any such suit, action, proceeding, or dispute.
(b)EACH PARTY ACKNOWLEDGES AND AGREES THAT ANY CONTROVERSY WHICH MAY ARISE UNDER THIS AGREEMENT OR THE OTHER TRANSACTION DOCUMENTS IS LIKELY TO INVOLVE COMPLICATED AND DIFFICULT ISSUES AND, THEREFORE, EACH PARTY IRREVOCABLY AND UNCONDITIONALLY WAIVES, TO THE FULLEST EXTENT PERMITTED BY APPLICABLE LAW, ANY RIGHT IT MAY HAVE TO A TRIAL BY JURY IN ANY LEGAL ACTION, PROCEEDING, CAUSE OF ACTION, OR COUNTERCLAIM ARISING OUT OF OR RELATING TO THIS AGREEMENT, INCLUDING ANY EXHIBITS AND SCHEDULES
26
ATTACHED TO THIS AGREEMENT, THE OTHER TRANSACTION DOCUMENTS, OR THE TRANSACTIONS CONTEMPLATED HEREBY OR THEREBY. EACH PARTY CERTIFIES AND ACKNOWLEDGES THAT: (I) NO REPRESENTATIVE OF THE OTHER PARTY HAS REPRESENTED, EXPRESSLY OR OTHERWISE, THAT THE OTHER PARTY WOULD NOT SEEK TO ENFORCE THE FOREGOING WAIVER IN THE EVENT OF A LEGAL ACTION; (II) EACH PARTY HAS CONSIDERED THE IMPLICATIONS OF THIS WAIVER; (III) EACH PARTY MAKES THIS WAIVER KNOWINGLY AND VOLUNTARILY; AND (IV) EACH PARTY HAS BEEN INDUCED TO ENTER INTO THIS AGREEMENT BY, AMONG OTHER THINGS, THE MUTUAL WAIVERS AND CERTIFICATIONS IN THIS SECTION.
Section 8.09Counterparts. This Agreement may be executed in counterparts, each of which shall be deemed an original, but all of which together shall be deemed to be one and the same agreement. A signed copy of this Agreement delivered by email or other means of electronic transmission shall be deemed to have the same legal effect as delivery of an original signed copy of this Agreement.
Section 8.10Specific Performance. The parties agree that irreparable damage would occur if any provision of this Agreement were not performed in accordance with the terms hereof (including if the parties fail to take such actions as are required to consummate the transactions contemplated hereby) or were otherwise breached. Each party hereto shall be entitled to an injunction or injunctions to prevent breaches or threatened breaches of this Agreement or to enforce specifically the performance of the terms and provisions hereof without proof of actual damages, in addition to any other remedy to which it is entitled at law or in equity. No party will oppose the granting of an injunction, specific performance, or other equitable relief when expressly available pursuant to the terms of this Agreement on the basis that the other party has an adequate remedy at law or that an award of specific performance is not an appropriate remedy for any reason at law or equity. No other party or other Person shall be required to obtain, furnish, or post any bond or similar instrument in connection with or as a condition to obtaining any remedy referred to in this Section 8.10, and each party hereto irrevocably waives any right it may have to require the obtaining, furnishing, or posting of any such bond or similar instrument. The right of specific enforcement is an integral part of the transactions contemplated hereby and without that right, neither party would have entered into this Agreement.
Section 8.11No Recourse. This Agreement may only be enforced against, and any claim, action, suit or other legal proceeding based upon, arising out of, or related to this Agreement, or the negotiation, execution or performance of this Agreement, may only be brought against the entities that are expressly named as parties hereto and then only with respect to the specific obligations set forth herein with respect to such party. No past, present or future director, officer, employee, incorporator, manager, member, partner, stockholder, Affiliate, agent, attorney or other Representatives of any party hereto or of any Affiliate of any party hereto, or any of their successors or permitted assigns (collectively, the “Non-Party Affiliates”), shall have any liability for any obligations or liabilities of any party hereto under this Agreement or for any claim, action, suit or other legal proceeding based on, in respect of or by reason of the transactions contemplated hereby. This Section 8.11 is intended for the benefit of and shall be enforceable by each of the Non-Party Affiliates. Notwithstanding the foregoing, nothing herein is intended or shall be construed as eliminating or otherwise limiting Buyer and/or Buyer Parent from seeking a court order or other equitable relief including but not limited to specific performance (as set forth in Section 8.10 above) in order to enforce the provisions of this agreement.
[Remainder of page intentionally left blank. Signatures follow.]
27
The parties hereto have caused this Agreement to be executed as of the date first written above by their duly authorized representatives.
[***]
By:
Name:
Title:
[***]
By:
Name:
Title:
Prenetics Global Limited
By:
Name:
Title:
Europa Sports Partners, LLC
By:
Name:
Title:
Hubmatrix Partners, LLC
By:
Name:
Title:
The parties hereto have caused this Agreement to be executed as of the date first written above by their duly authorized representatives.
IM8 Global Limited
By:
Name:
Title:
The following is a brief description of the omitted schedules, exhibits, and attachments:
Omitted Disclosure Schedules:
•Section 1.01(b) —Assigned Contracts
•Section 1.01(h) — Assigned Real Property Leases
•Section 1.01(i) — Insurance Benefits
•Section 1.02(b) — Shared Contracts
•Section 2.02(a)(vii) — Required Consents
•Section 3.02 — Conflicts or Consents
•Section 3.05 — Certain Changes, Events, and Conditions
•Section 3.06(b) — Material Customers of the Business
•Section 3.09 — Tangible Personal Property
•Section 3.10(a) — Real Property Leases
•Section 3.10(b) — Exceptions to Real Property Leases
•Section 3.10(j) — Real Property Liens and Indebtedness
•Section 3.11(a) — Intellectual Property Assets
•Section 3.11(b) — Intellectual Property Agreements
•Section 3.12 — Employee Benefit Plans
•Section 3.13(a) — Employees, Independent Contractors and Consultants
•Section 3.14 — Legal Proceedings
•Section 3.21 — Affiliate Transactions
Omitted Exhibits:
•Exhibit B — Form of Bill of Sale, Assignment and Assumption Agreement
•Exhibit C — Form of Intellectual Property Assignment
•Exhibit D-1 — Form of [***] Landlord Consent
•Exhibit D-2 — Form of [***] Landlord Consent
•Exhibit E — Form of Employee Leasing Agreement
•Exhibit F — Form of Referral Agreement.
•Exhibit G — Form of Joinder to Buyer Parent's Right of First Refusal and Co-Sale Agreement
Exhibit H — Form of Adoption Agreement to Buyer Parent's Voting Agreement
EXHIBIT A
DEFINTIONS
“Affiliate” of a Person means any other Person that directly or indirectly, through one or more intermediaries, controls, is controlled by, or is under common control with, such Person. The term “control” (including the terms “controlled by” and “under common control with”) means the possession, directly or indirectly, of the power to direct or cause the direction of the management and policies of a Person, whether through the ownership of voting securities, by contract or otherwise.
“Business Day” means a day other than a Saturday, Sunday or other day on which banks in Atlanta, Georgia are required or authorized by Law to be closed.
(a)“Business IT Systems” means all Software, computer hardware, servers, networks, platforms, peripherals, and similar or related items of automated, computerized, or other information technology (IT) networks and systems (including telecommunications networks and systems for voice, data, and video) owned, leased, licensed, or used (including through cloud-based or other third-party service providers) in the conduct of the Business.
(b)“[***] Landlord Consent” shall mean the consent from the landlord pursuant to the [***] Lease, in substantially the form attached hereto as Exhibit D-1.
(c)“[***] Lease” means that certain Lease Agreement, by and between, [***] and Europa Sports Seller, dated [***], as amended.
(d)“[***] Landlord Consent” shall mean the consent from the landlord pursuant to the [***] Lease, in substantially the form attached hereto as Exhibit D-2.
“[***] Lease” means that certain Lease Agreement, by and between Europa Sports Seller and [***], dated [***], as amended.
“Debt” means any indebtedness or debt-like items of a Person, in respect of borrowed money or evidenced by bonds, notes, debentures, revolving credit facilities or other similar instruments or letters of credit (or reimbursement agreements in respect thereof) or banker’s acceptances or obligations under capital leases, required to be capitalized under GAAP (excluding the effects of ASC 842) or the unpaid balance of the purchase price of any property or assets (including any earn-out, whether or not contingent), or any outstanding checks, drafts or overdrafts, as well as the amount of all indebtedness of others secured by a Lien on any asset of such Person (whether or not such indebtedness is assumed by such Person), or any off-balance sheet liabilities, unpaid settlement obligations, deferred compensation, retention, severance and similar obligations and all accrued or unpaid commissions, profit sharing and bonuses, payable to current or former employees, directors and/or other service providers (including the employer portion of all Taxes, withholdings and Employee benefit plan contributions in connection with any of the foregoing), liabilities with respect to any unfunded or underfunded Employee Plan, accrued paid time off, or any all customer deposits, deferred income, billings in excess of earnings and other deferred revenue, and, to the extent not otherwise included, the amount of any indebtedness of any other Person guaranteed by such Person, and interest expense accrued but unpaid, income Tax obligations, investment banking fees, intercompany debt, and other expenses and any other Liabilities that are not directly related to the continuing operation of the Business, and all penalties, premiums, termination fees or breakage costs due upon or relating to the prepayment of any of such indebtedness, but the term “Debt” does not include Sellers' ordinary course accounts payable of the Business that are not yet overdue.
“Distribution Rights” means the right to provide third-party logistics, fulfillment, warehousing, and distribution services, including inventory management, order fulfillment, shipping, and related logistics services.
“Enforceability Exceptions” shall mean those certain exceptions to enforceability as may be limited by bankruptcy, insolvency, reorganization, moratorium and other similar Laws now or hereafter in effect relating to or affecting creditors’ rights generally or by general equitable principles (regardless of whether such enforceability is considered in a proceeding in equity or at law) and (b) that specific performance may not be available.
“Environmental Laws” shall mean all federal, state, local and foreign Laws in effect as of the Closing Date relating to protection of the environment.
“Exclusive Parcel Shipping Rights” means the exclusive right to arrange and manage the transportation and delivery of Parcels by third party carriers (under broker or freight forwarder authority granted by the Federal Motor Carrier Safety Administration) for IM8’s products (including any related products sold through an Affiliate of IM8) to IM8’s end customers.
“Expansion Criteria” means, with respect to any proposed additional location(s): Buyer Parent has the operational capability to perform the Distribution Rights and Parcel Shipping Rights, as applicable, in such location(s); and (2) Buyer Parent is able to offer rates that are commercially reasonable and not inconsistent with prevailing market rates for Distribution Rights and Parcel Shipping Rights, as applicable, in such location(s), provided, that if Sellers notify Buyer Parent that Buyer Parrent has not satisfied criteria (2), Buyer Parent shall have five (5) business days to submit revised rates to satisfy such criteria.
“First Right of Refusal” means the right, but not the obligation, of Buyer Parent to enter into a contract with Sellers for the Distribution Rights with respect to IM8’s products (including any related products sold through an Affiliate of IM8) for any location outside of the Exclusive Distribution Geographies, on terms no less favorable than those offered to any third party. Prior to accepting any third-party offer for such Distribution Rights, Sellers shall deliver to Buyer Parent written notice of such offer, including a copy of the proposed definitive terms). Buyer Parent shall have [***] days from receipt of such notice to exercise its right by providing written notice of its intent to match the third-party terms. If Buyer Parent does not exercise this right within the [***] day period, Sellers may proceed with the third-party offer for the applicable location; provided that (A) the executed agreement is on terms no more favorable to such third party than those set forth in the notice, and (B) any amendment, renewal, or replacement of such agreement shall again be subject to the First Right of Refusal.
“GAAP” means generally accepted accounting principles in effect in the United States as of the relevant date(s) of application thereof.
“Governmental Order” means any order, writ, judgment, injunction, decree, stipulation, determination, penalty, or award entered by or with any Governmental Authority.
“Hazardous Substances” shall mean any substance or material that is regulated under any Environmental Law as a “toxic substance,” “hazardous substance,” “hazardous waste” or words of similar meaning and regulatory effect, or contains asbestos, petroleum or polychlorinated biphenyls.
“IM8 US” means IM8 (US) LLC.
(i)“IM8 Agreement” means that certain Service Agreement, dated October 15, 2025, by and between Seller Parent, on behalf of IM8 US, and Buyer Parent.
(ii)“Intellectual Property” means any and all rights in, arising out of, or associated with any of the following in any jurisdiction throughout the world: (a) issued patents and patent applications (whether provisional or non-provisional), including divisional, continuations, continuations-in-part, substitutions, reissues, reexaminations, extensions, or restorations of any of the foregoing, and other Governmental Authority-issued indicia of invention ownership (including certificates of invention, petty patents, and patent utility models) (“Patents”); (b) trademarks, service marks, brands, certification marks, logos, trade dress, trade names, and other similar indicia of source or origin, together with the goodwill connected with the use of and symbolized by, and all registrations, applications for registration, and renewals of, any of the foregoing (“Trademarks”) and any associated brand guides related thereto; (c) copyrights and works of authorship, whether or not copyrightable, and all registrations, applications for registration, and renewals of any of the foregoing (“Copyrights”); (d) internet domain names and social media account or user names (including "handles"), whether or not Trademarks, all associated web addresses, URLs, websites and web pages, social media sites and pages, and all content and data thereon or relating thereto, whether or not Copyrights; (e) mask works, and all registrations, applications for registration, and renewals thereof; (f) industrial designs, and all Patents, registrations, applications for registration, and renewals thereof; (g) trade secrets, know-how, inventions (whether or not patentable), discoveries, improvements, technology, business and technical information, databases, data compilations and collections, tools, methods, processes, techniques, and other confidential and proprietary information and all rights therein (“Trade Secrets”); (h) computer programs, operating systems, applications, firmware and other code, including all source code, object code, application programming interfaces, data files, databases, protocols, specifications, and other documentation thereof (“Software”); and (i) all other intellectual or industrial property and proprietary rights.
(iii)“Intellectual Property Agreements” means all licenses, sublicenses, consent to use agreements, settlements, coexistence agreements, covenants not to sue, waivers, releases, permissions and other Contracts, whether written or oral, relating to any Intellectual Property that is used or held for use in the conduct of the Business as currently conducted or proposed to be conducted to which Sellers are a party, beneficiary or otherwise bound.
(iv)“Intellectual Property Assets” means all Intellectual Property that is owned by Sellers and used or held for use in the conduct of the Business as currently conducted or proposed to be conducted, including as set forth on Section 3.11(a) of the Disclosure Schedules, together with all (i) royalties, fees, income, payments, and other proceeds now or hereafter due or payable to Sellers with respect to such Intellectual Property; and (ii) claims and causes of action with respect to such Intellectual Property, whether accruing before, on, or after the date hereof and accruing on or after the date hereof, including all rights to and claims for damages, restitution, and injunctive and other legal or equitable relief for past, present, or future infringement, misappropriation, or other violation thereof.
(v)“Intellectual Property Registrations” means all Intellectual Property Assets that are subject to any issuance, registration, or application by or with any Governmental Authority or authorized private registrar in any jurisdiction, including issued Patents, registered Trademarks, domain names and Copyrights, and pending applications for any of the foregoing.
“Knowledge of Sellers or Sellers’ Knowledge” or any other similar knowledge qualification, means the actual knowledge of [***], [***], and [***] after reasonable inquiry.
“Landlord Consents” shall mean the [***] Landlord Consent, and the [***] Landlord Consent.
“Law” means any statute, law, ordinance, regulation, rule, code, constitution, treaty, common law, other requirement, or rule of law of any Governmental Authority.
“Legal Proceeding” means any judicial, administrative or arbitral actions, investigations, suits, inquiries, hearings or proceedings (public or private) by or before a Governmental Authority.
“Liabilities” means liabilities, obligations, or commitments of any nature whatsoever, whether asserted or unasserted, known or unknown, absolute or contingent, accrued or unaccrued, matured or unmatured, or otherwise.
“Lien” means any lien, encumbrance, pledge, mortgage, deed of trust, security interest, claim, lease, charge, option, right of first refusal, easement, servitude or transfer restriction.
“Loss” means any losses, damages, liabilities, deficiencies, judgments, interest, awards, penalties, fines, costs, or expenses of whatever kind, including reasonable attorneys’ fees.
“Material Adverse Effect” means any event, occurrence, fact, condition or change that is materially adverse to (a) the business, results of operations, financial condition or assets of the Business, taken as a whole; or (b) the ability of Sellers to consummate the transactions contemplated hereby; provided, however, that for purposes hereof, any facts, events, occurrences, changes or effects directly or indirectly attributable to, resulting from, relating to or arising out of the following (by themselves or when aggregated with any other, changes or effects) shall not be deemed to be, constitute, or be taken into account when determining whether there has or may, would or could have occurred a Material Adverse Effect: (i) general changes in the financial or securities markets (including changes in the credit, debt, securities and capital markets) or general economic or political conditions in the country or region in which Sellers or any of their subsidiaries do business; (ii) changes, conditions or effects that generally affect the industries in which Sellers or any of their subsidiaries principally operate; (iii) changes in applicable Laws or generally accepted accounting principles or other applicable accounting principles or mandatory changes in the regulatory accounting requirements applicable to any industry in which Sellers and their subsidiaries principally operate; or (iv) conditions caused by acts of “God,” terrorism, war (whether or not declared), natural disaster or any outbreak or continuation of an epidemic or pandemic (including, without limitation, COVID-19) or the worsening thereof, including the effects of any Governmental Authority or other third-party responses thereto; provided, that any event, occurrence, fact, condition or change referred to in clauses (i) through (iv) may be taken into account in determining whether a Material Adverse Effect has occurred to the extent such event, occurrence, fact, condition or change has a disproportionate impact on the Business, taken as a whole, relative to other participants in the industries in which the Business operates.
“Parcel(s)” means the merchandise, small packages, or property that IM8 or any of its Affiliates tenders to Buyer Parent to arrange delivery transportation for through a third-party carrier.
“Permit” means any permit, franchise, authorization, license or other approval issued or granted by any Governmental Authority relating primarily to the Business.
“Permitted Encumbrances” means (a) liens for Taxes not yet due and payable or being contested in good faith by appropriate procedures, provided adequate reserves have been established with respect thereto; (b) mechanics’, carriers’, workmen’s, repairmen’s or other like liens arising or incurred in the ordinary course of business for amounts which are not due and payable and as would not in the aggregate
materially adversely affect the value of, or materially adversely interfere with the use of, the property subject thereto; (c) easements, rights of way, zoning ordinances and other similar encumbrances affecting real property; (d) other than with respect to owned real property, liens arising under original purchase price conditional sales contracts and equipment leases with third parties entered into in the ordinary course of business; and (e) other imperfections of title or encumbrances, if any, that have not had, and would not have, a Material Adverse Effect.
“Person” means an individual, corporation, partnership, joint venture, limited liability company, governmental authority, unincorporated organization, trust, association or other entity.
“Personal Data” means all data relating to one or more individual(s) that is personally identifying (i.e., data that identifies an individual or, in combination with any other information or data available to the Sellers in the operation of the Business is capable of identifying an individual).
(vi)“Receiving Seller” shall mean any Seller, IM8, or any of their respective Affiliates who receive Consideration Shares in accordance with the terms of this Agreement.
“[***] Agreement” means that certain Warehouse and Fulfillment Agreement, dated [***], by and between Hubmatrix Seller and [***], as amended.
“[***] Contingency” means the satisfaction of the following condition: [***] pays to Buyer Parent an aggregate amount of at least Three Million Dollars ($[***]) in fees during the twelve (12) month period from and after the Closing Date (the “[***] Minimum Payment Threshold”). For the avoidance of doubt, payments made by [***] shall be counted toward the [***] Minimum Payment Threshold only when actually received by Buyer Parent in cash. Notwithstanding the foregoing, if the [***] Agreement is properly terminated by [***] for cause beyond applicable notice and cure periods in accordance with sections 9, 10 and 11 of Exhibit C (Service Level Agreement) of the Amendment #1 dated [***] to the [***] Agreement during the twelve (12) month period from and after the Closing Date, then the [***] Contingency shall be deemed satisfied at such point when the termination is effective (the “[***] Breach Termination”).
“[***] Contingency Failure Date” means, if the [***] Contingency is not satisfied, the first date upon which the [***] Contingency is no longer able to be satisfied, which shall be determined on or after such date by Buyer Parent in its sole discretion. For example, and without limiting the previous sentence, the [***] Contingency Failure Date may be, if the [***] Minimum Payment Threshold is not satisfied during the twelve-month period from and after the Closing Date, the last day of such twelve-month period.
“Tax” or “Taxes” means all federal, state, local, foreign and other income, gross receipts, sales, use, production ad valorem, transfer, franchise, registration, profits, license, lease, service, service use, withholding, payroll, employment, unemployment, estimated, excise, escheat or unclaimed property, severance, environmental, stamp, occupation, premium, property (real or personal), real property gains, windfall profits, customs, duties or other taxes, fees, assessments, or charges of any kind whatsoever, together with any interest, additions or penalties with respect thereto and any interest in respect of such additions or penalties.
“Transaction Documents” means this Agreement, the Bill of Sale, the Assignment and Assumption Agreement, the TSA, the Employee Leasing Agreement, the Distribution Agreement, and the other agreements, instruments and documents to be delivered at Closing.
Exhibit 4.29
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. Certain schedules have been excluded from this exhibit because they are both (i) not material and (ii) not otherwise disclosed in the exhibit or our annual report on Form 20-F for the fiscal year ended December 31, 2025. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
PRENETICS GLOBAL LIMITED AS THE VENDOR AND IMAGE FRAME INVESTMENT (HK) LIMITED AS THE PURCHASER | ||
SHARE PURCHASE AGREEMENT | ||
CONTENTS | |||||
| Clause | Page | ||||
Agreed form documents
1. Amended Articles
2. Shareholders' Agreement
3. Share Transfer Form
THIS AGREEMENT is dated on 13 February 2026
BETWEEN:
(1)PRENETICS GLOBAL LIMITED, a company incorporated in the Cayman Islands, whose registered office is at Unit 703-706, K11 Atelier King's Road, 728 King's Road, Quarry Bay, Hong Kong and whose shares are traded on NASDAQ with ticker PRE, having its principal executive office at 703-706, K11 Atelier King's Road, 728 Kings Road, Hong Kong (the "Vendor"); and
(2)IMAGE FRAME INVESTMENT (HK) LIMITED, a company incorporated under the laws of Hong Kong, whose registered office is at 29/F., Three Pacific Place, No. 1 Queen's Road East, Wanchai, Hong Kong (the "Purchaser").
The Vendor and the Purchaser are collectively referred to as the "Parties" and each individually as a "Party".
RECITALS:
(A)Insighta Holdings Limited (the "Company") is an exempted company limited by shares and incorporated in the Cayman Islands, further details of which are set out in Schedule 1.
(B)The Vendor and the Purchaser entered into a share purchase agreement dated 14 October 2024 (the "2024 SPA"), pursuant to which the Vendor sold, and the Purchaser acquired, 300,000 Class A Shares, which were reclassified as Class C Shares. Completion of that transaction occurred on the same date.
(C)Subject to the terms and conditions of this Agreement, the Vendor has agreed to sell, and the Purchaser has agreed to purchase, the Sale Shares, all of which are legally and beneficially owned by the Vendor as at the date of this Agreement.
GENERAL TERMS
1.INTERPRETATION
1.1Definitions
In this Agreement, unless the contrary intention appears or the context requires otherwise:
"2024 Announcement 7 Reporting" has the meaning given to it in Clause 3.1(c)(i).
"2024 SPA" has the meaning given to it in Recital (B).
"2024 Transaction" has the meaning given to it in Clause 3.1(c)(ii).
"2026 Announcement 7 Reporting" has the meaning given to it in Clause 5.1.
"Acceptable Evidence of Payment" means receipt of Tax payment (完税证明 including a 中华人民共和国税收缴款书) issued by the relevant Tax Authority in the PRC, evidencing that the final assessed Tax amount has been paid by or on behalf of the Vendor.
"Accounts Date" means 31 December 2024.
"Action" means legal proceedings of any nature, including any litigation, arbitration, hearing, claim, demand, grievance or investigation (in each case, whether civil, criminal, regulatory or otherwise).
"Affiliate" of any person means:
(a)in relation to a person who is an individual, that individual's parents, any spouse and child and any person which is directly or indirectly controlled by that individual and/or that individual's parents, spouse and child (acting singly or together); and
(b)in relation to a person that is not an individual, any other person that directly or indirectly controls, is controlled by or is under common control with the specified person; and
(c)in relation to a trust, includes any trustee or beneficiary (actual or potential) of the trust.
"Allen Chan" means Dr. Kwan Chee Chan.
"Amended Articles" means the amended and restated Articles in the agreed form and to the satisfaction of the Purchaser.
"AMR" means the Administration for Market Regulation.
"Announcement 37" means Announcement of the PRC State Administration of Taxation on Matters Concerning Withholding at Source of Enterprise Income Tax of Non-Resident Enterprises (《关于非居民企业所得税源泉扣缴有关问题的公告》 [国家税务总局公告2017 年第37 号]), promulgated on 17 October 2017, as may be amended or supplemented from time to time by implementing regulations.
"Announcement 7" means the Announcement of the PRC State Administration of Taxation on Several Issues concerning the Enterprise Income Tax on the Indirect Transfers of Properties by Non-Resident Enterprises (《关于非居民企业间接转让财产 企业所得税若干问题的公告》国家税务总局公告2015 年第7 号), promulgated on 3 February 2015, as may be amended or supplemented from time to time by implementing regulations.
"Anti-Bribery Laws" means Laws relating to anti-bribery or anti-corruption (governmental or commercial) which apply to the Vendor, the Group Companies or any employees, directors or agents of the Group Companies from time to time, including Laws that prohibit the payment, offer, promise, or authorisation of the payment or transfer of anything of value (including gifts or entertainment), directly or indirectly, to any government official or commercial entity to obtain an illegitimate business advantage, including the U.S. Foreign Corrupt Practices Act (15 U.S.C. § 78 dd 1 et seq.), the Bribery Act of 2010, any rules and regulations thereunder, any similar laws or regulations in any other jurisdiction (including Hong Kong and the PRC) and all national and international laws enacted to implement the OECD Convention on Combating Bribery of Foreign Officials in International Business Transactions.
"Anti-Money Laundering Laws" means (a) the USA PATRIOT Act of 2001 (Pub. L. No. 107-56), (b) the U.S. Money Laundering Control Act of 1986, (c) the Anti-Money Laundering Law of the PRC, (d) the Hong Kong Anti-Money Laundering and Counter-Terrorist Financing Ordinance, and (e) any other applicable Law of any relevant jurisdiction (including Hong Kong and the PRC) having the force of law and relating to anti-money laundering, as each such Law may be amended from time to time.
"Articles" means the memorandum and articles of association of the Company adopted by the Company from time to time.
"Audited Accounts" means:
(a)the audited consolidated statement of financial position of the Company as at the Accounts Date;
(b)the audited consolidated statement of profit or loss and other comprehensive income, statement of changes in equity and statement of cash flows of the Company for each of
the financial years from 17 March 2023, being the date of incorporation of the Company, to the Accounts Date,
along with any notes, reports, statements or documents included in or annexed or attached to them prepared by the Company and audited by the Auditors.
"Auditor" means Deloitte Touche Tohmatsu Limited.
"Business Day" means a day (other than a Saturday, Sunday or a public holiday in Hong Kong or the PRC, and a day on which a typhoon signal number 8 or a black rainstorm warning is hoisted in Hong Kong at any time) on which commercial banks are open for business in Hong Kong or the PRC.
"Class A Shares" means Class A ordinary shares with par value of US$0.0001 each in the share capital of the Company, each having the rights, benefits and privileges as set out in the Articles, each a "Class A Share".
"Class C Shares" means Class C ordinary shares with par value of US$0.0001 each in the share capital of the Company, each having the rights, benefits and privileges as set out in the Articles, each a "Class C Share".
"Companies Ordinance" means the Companies Ordinance (Chapter 622 of the Laws of Hong Kong).
"Company" has the meaning given to it in Recital (A).
"Completion" means completion of the sale and purchase of the Sale Shares in accordance with this Agreement.
"Completion Amount" means the Purchase Price less the Holdback Amount.
"Completion Date" has the meaning given to it in Clause 4.1.
"Conditions" means the conditions set forth in Clause 3.1.
"Confidential Information" means all information (regardless of its form) disclosed to a Party (or to its Affiliates or Representatives) under or in connection with this Agreement as well as the subject matter or the terms of this Agreement or the other Transaction Documents and any claim or potential claim hereunder and thereunder. Confidential Information shall also include the negotiations relating to this Agreement and the other Transaction Documents, but shall not include information which:
(a)is generally known to the public at the time of disclosure, or becomes generally known to the public other than through a breach of this Agreement or an obligation of confidence owed to the discloser or any Affiliate of the discloser;
(b)was already known to the receiver at the time of that disclosure (unless that knowledge arose from a breach of an obligation of confidentiality);
(c)the receiver acquires from a source other than the discloser (or any Affiliate or Representative of the discloser), where that source is entitled to disclose it; or
(d)is independently created or developed by a Party (or any of its Affiliates or its Representatives) without use of or reference to any Confidential Information.
"control" means the power to direct the management or policies of a person, whether through the ownership (directly or indirectly) of more than fifty per cent (50%) of the legal or beneficial interests or the voting power of such person, through the power to appoint a majority of the members of the board of directors or similar governing body of such person, through contractual arrangements (including through the VIE Documents or the VIE Restructuring Documents (as applicable)) or the power to secure that the affairs, the general management and/or policies of such person are conducted in accordance with the wishes of that person by virtue of any powers conferred by constitutional or corporate documents or any other document, agreement or arrangement or otherwise, and references to "controls", "controlled" or "controlling" shall be construed accordingly.
"Data Protection Laws" has the meaning given to it in Paragraph 22.1 of Schedule 2.
"Data Room" means the virtual data room titled "[***]" hosted by Google at [***] containing documents and information relating to the Group and made available to the Purchaser and its Representatives on 12 December 2025.
"Dennis Lo" means Dr. Yuk Ming Dennis Lo.
"Deyi Health IT" means Deyi Health Information Technology (Shenzhen) Co., Ltd. (得易健康信息科技(深圳)有限责任公司), a company incorporated in the PRC and de-registered with effect from 13 December 2023.
"Deyi Health Medical" means Deyi Health Medical Device (Shenzhen) Co., Ltd. (得易健康医疗器械(深圳)有限责任公司), a company incorporated in the PRC and de-registered with effect from 13 December 2023.
"Director Service Agreements" means, collectively:
(a)the director service agreement dated 20 July 2023 entered into between the Company and [***]; and
(b)the director service agreement dated 20 July 2023 entered into between the Company and [***].
"Disclosed" means fairly disclosed with sufficient detail and particularity to enable a reasonable purchaser experienced in transactions similar to those contemplated hereunder to be able to identify the nature and scope of the matter disclosed.
"Disclosure Letter" means the disclosure letter to be delivered by the Vendor to the Purchaser on the date of, and having the same date as, this Agreement, disclosing information constituting exceptions to the Vendor's Warranties.
"Dispute" has the meaning given to it in Clause 16(b).
"Due Diligence" means the due diligence carried out by the Purchaser and its Representatives in connection with, among others, the assets, liabilities, business, affairs, operations, financial condition, prospects, records and legal matters relating to the Group Companies.
"Due Diligence Responses" means the responses made by or on behalf of the Company and/or its Representatives from time to time in connection with the Due Diligence, including, without limitation:
(a)any and all e-mail correspondences between the Company and/or its Representatives and the Purchaser and/or its Representatives;
(b)any and all documents, notes, information and/or agreements provided by or on behalf of the Company and/or its Representatives to the Purchaser and/or its Representatives; and
(c)the responses made by or on behalf of the Company and/or its Representatives by email:
(i)on [***] and [***], including the responses to the Top-Up LDD Requests prepared by [***];
(ii)on [***], [***], [***], [***] and [***], including the response to supplementary questions to the Top-Up LDD Requests prepared by [***]; and
(iii)on [***], [***] and [***] regarding the Due Diligence questions on intellectual property prepared by [***],
(iv)all of which have been included in the Data Room.
"Employees" has the meaning given to it in Paragraph 15.1 of Schedule 2.
"Encumbrance" means any mortgage, lien, charge, pledge, option, restriction, third party right or interest, assignment, hypothecation, deed of trust, security interest, title retention or any other arrangement the effect of which is the creation of security, or any other interest, equity or other right of any person (including any right to acquire, option, right of first refusal or right of pre-emption), or any arrangement to create any of the same, and "Encumber" shall be construed accordingly.
"Equity Pledges" means, collectively:
(a)the equity pledge agreement dated 16 November 2023 entered into between the Shenzhen WFOE, [***] and the Shenzhen VIE Entity; and
(b)the equity pledge agreement dated 16 November 2023 entered into between the Shenzhen WFOE, [***] and the Shenzhen VIE Entity,
pursuant to which each of [***] and [***] (as applicable) pledges their respective equity interests in the Shenzhen VIE Entity to the Shenzhen WFOE to secure the full performance by the Shenzhen VIE Entity and [***] and [***] (as applicable) of their respective obligations under the VIE Documents.
"Existing Articles" means the third amended and restated memorandum and articles of association of the Company adopted by the Company on 14 October 2024.
"Existing Shareholders' Agreement" means the shareholders' agreement dated 14 October 2024 entered into between the Founders, the Company, the Vendor and the Purchaser.
"Existing Shareholders' Waiver" has the meaning given to it in Clause 4.2(a)(iv).
"Founders" means, collectively, Allen Chan and Dennis Lo, and each a "Founder".
"Fundamental Warranties" means the representations and warranties contained in Paragraphs 1 (Capacity and Authority), 2 (Sale Shares), 3 (Group Companies), 4 (Information) and 18 (Insolvency) of Schedule 2, and each a "Fundamental Warranty".
"Fundamental Warranty Claim" means any claim made by the Purchaser for breach of any Fundamental Warranty.
"Government Authority" means any governmental, semi-governmental, administrative, fiscal, judicial or quasi-judicial body, department, commission, authority, tribunal, agency or entity in any jurisdiction.
"Group" means the Company, the Subsidiaries and their respective subsidiaries (if any), and any one of them, a "Group Company" and collectively, the "Group Companies".
"Group Intellectual Property" means all the Intellectual Property owned by any Group Company or to which any Group Company has the legal or contractual entitlement or claim of ownership.
"Guangzhou VIE Entity" means Insighta Bio-technology (Guangzhou) Co., Ltd., a company incorporated under the laws of the PRC, the particulars of which are set out in Schedule 1.
"HKIAC" has the meaning given to it in Clause 16(b).
"Holdback Amount" means [***].
"Hong Kong" means the Hong Kong Special Administrative Region of the PRC.
"Insighta HK" means Insighta Limited (明察康健有限公司), a company incorporated under the laws of Hong Kong, the particulars of which are set out in Schedule 1.
"Intellectual Property" means all industrial and intellectual property rights, whether registered or not, including pending applications for registration of such rights and the right to apply for registration or extension of such rights including patents, petty patents, utility models, design patents, designs, copyright (including moral rights and neighbouring rights), database rights, rights in integrated circuits and other sui generis rights, trade marks, trading names, company names, service marks, logos, the get-up of products and packaging, geographical indications and appellations and other signs used in trade, internet domain names, social media user names, rights in know-how (including information comprised in formulae, algorithms, techniques, specifications, computation models, software and manuals), trade secrets, right to use and protection of the confidentiality of Confidential Information and any rights of the same or similar effect or nature as any of the foregoing anywhere in the world.
"IP Licence Agreement" means the licence agreement dated [***] entered into between [***] and Insighta HK.
"IP Licences In" has the meaning given to it in Paragraph 12.7of Schedule 2.
"Last Management Accounts Date" means 31 December 2023.
"Law" means all civil and common law, statute, subordinate legislation, treaty, regulation, rule, directive, decision, by-law, ordinance, circular, code, order, notice, demand, decree, injunction, resolution or judgment of any Government Authority.
"Leased Properties" means the properties leased by a Group Company, the list of which is set out in Schedule 4.
"Licensed Field of Use" has the meaning given to it in the IP Licence Agreement.
"Long Stop Date" means the date falling 60 days after the date of this Agreement, or such other date as the Parties may agree in writing.
"Management Accounts" means:
(c)the unaudited consolidated profit and loss accounts of the Company in respect of the periods starting on (i) 17 March 2023 and ending on the Last Management Accounts Date; and (ii) 1 January 2024 and ending on the Management Accounts Date; and
(d)the unaudited consolidated balance sheets of the Company as at (i) the Last Management Accounts Date; and (ii) the Management Accounts Date.
"Management Accounts Date" means 30 September 2025.
"Material Contract" has the meaning given to it in Paragraph 14.2 of Schedule 2.
"Notice" shall have the meaning given to it in Clause 13.1.
"Permit" means:
(e)a permit, licence, consent, approval, certificate, qualification, specification, registration or other authorisation; or
(f)a filing of a notification, report or assessment,
in each case necessary for or material to (i) the effective operation of the business of any Group Company (including the research and development or any clinical trial of any product or technology by or on behalf of any Group Company), or (ii) any Group Company's ownership, possession, occupation or use of an asset or Intellectual Property.
"PRC" means the People's Republic of China (for the purposes of this Agreement only, excluding Hong Kong, Macau Special Administrative Region and Taiwan region).
"PRC Tax and AMR Filings" have the meaning given to it in Clause 8.3.
"Purchase Price" means [***]
"Representative" means any person acting on behalf of a Party or an Affiliate of that Party, including any of its employee, agent, officer, director, auditor, adviser, partner, associate, consultant, joint venturer or sub-contractor.
"Sale Shares" means the 700,000 Class A Shares, representing approximately thirty four point ninety four per cent (34.94%) of the total issued shares of the Company (on an as-converted and fully diluted basis) as at the date of this Agreement, which will be reclassified into Class C Shares at Completion.
"Sanctions" mean the export controls, economic, financial or trade controls or sanctions laws, regulations, embargoes or restrictive measures, administered, enacted or enforced by any Sanctions Authority from time to time.
"Sanctions Authority" means:
(a)the United Nations;
(b)the United States;
(c)the European Union and each of its member states;
(d)the United Kingdom;
(e)the PRC; or
(f)any other jurisdiction which the Vendor or any Group Company operates, or to which it is subject,
and the respective governmental institutions, departments, committees, agencies, or offices of any of the foregoing, including, without limitation, the U.S. Department of State, the U.S. Department of Commerce's Bureau of Industry and Security, the U.S. Department of the Treasury's Office of Foreign Assets Control, the Council of the EU, the European Parliament, the European Commission (including the competent authorities of any present or future member state of the European Union), HM Treasury of the United Kingdom, the United Kingdom Department for Business and Trade, the Export Control Joint Unit, the PRC Ministry of Commerce, the PRC Ministry of Foreign Affairs, and/or any other applicable government or regulatory body.
"Sanctioned List" means any list of persons who are the subject of Sanctions published, or public announcement of Sanctions designations made, by any of the Sanctions Authorities, each as amended, supplemented or substituted from time to time.
"Sanctioned Jurisdiction" means any country, territory or region that is the subject or target of comprehensive, country-wide or territory-wide Sanctions from time to time (currently, Cuba, Iran, North Korea the Crimea region, and the non-government controlled areas of Ukraine in the oblasts of Donetsk, Luhansk, Kherson, and Zaporizhzhia and, until 1 July 2025, Syria).
"Sanctioned Person(s)" means any person that is: (i) listed on any Sanctioned List; (ii) located, organized or ordinarily resident in a Sanctioned Jurisdiction; (iii) directly or indirectly controlled by any Person covered by, or acting on behalf of, (i) or (ii); (iv) the government of Cuba, Iran, North Korea or Syria or a person owned or controlled by or acting on behalf of such governments; or (v) any other person targeted by Sanctions.
"Shareholders' Agreement" means the amended and restated shareholders' agreement to be entered into between the Founders, the Company and the Purchaser at Completion, in the agreed form.
"Share Transfer Form" means the share transfer form to be entered into between the Vendor and the Purchaser at Completion in the agreed form.
"Shenzhen VIE Entity" means Insighta Industry (Shenzhen) Co., Ltd., a company incorporated under the laws of the PRC, the particulars of which are set out in Schedule 1.
"Shenzhen WFOE" means 明察康健(深圳)科技有限责任公司, a company incorporated under the laws of the PRC, the particulars of which are set out in Schedule 1.
"Subsidiaries" means, collectively, Insighta HK, the Shenzhen WFOE and the VIE Entities, and "Subsidiary" means any of them.
"Surviving Provisions" means Clauses 1 (Interpretation), 11 (Confidentiality and Announcements), 12 (Costs, Expenses and Fees), 13 (Notices), 14 (Assignment), 15 (Miscellaneous), 16 (Governing Law and Dispute Resolutions) and 17 (Counterparts).
"Take2 Group" means Take2 Holdings Limited and/or its Affiliates.
"Taxes" means any and all taxes, duties, withholdings, levies, assessments, imposts, fees and charges (including any capital gains tax, value-added tax or similar tax and any stamp, registration or similar tax), together with any claims, penalties, fines, surcharges and interest arising or resulting therefrom and any additions thereto, imposed, collected or assessed by, or payable to, a Government Authority, and "Tax" shall be construed accordingly.
"Tax Authority" means any Government Authority anywhere in the world exercising a fiscal, revenue, customs or excise function.
"Tax Warranties" means the representations and warranties contained in Paragraph 8 (Tax) of Schedule 2.
"Tax Warranty Claim" means any claim made by the Purchaser for breach of any Tax Warranty.
"Territory" has the meaning given to it in the IP Licence Agreement.
"Third Party Intellectual Property" means all the Intellectual Property, which is licensed to any Group Company, or in respect of which any Group Company is granted an option or other right to obtain a license, pursuant to any of the IP Licences In.
"Third Parties Ordinance" means the Contracts (Rights of Third Parties) Ordinance (Chapter 623 of the Laws of Hong Kong).
"Transaction" means the sale and purchase of the Sale Shares in accordance with the terms and subject to the conditions of this Agreement.
"Transaction Documents" means this Agreement, the Amended Articles, the Disclosure Letter and the Shareholders' Agreement and each of the documents, instruments, certificates and other ancillary documents required to be entered into or delivered by the Parties or their respective Affiliates in accordance with the terms of this Agreement.
"USD" or "US$" means the lawful currency of the United States of America.
"Vendor's Bank Account" means the USD-denominated bank account in the name of the Vendor with the following details, or such other bank account designated by the Vendor by Notice to the Purchaser at least ten (10) Business Days in advance of the Completion Date.
| Beneficiary Bank: | [***] | ||||
Beneficiary Bank Address: | [***] | ||||
| Beneficiary Bank Code: | [***] | ||||
| Beneficiary Bank SWIFT Code: | [***] | ||||
| Beneficiary Account Name: | [***] | ||||
| Beneficiary Account No.: | [***] | ||||
| Account Currency: | [***] | ||||
"Vendor's Warranty" means a representation and warranty given by the Vendor under Clause 9 and Schedule 2, and "Vendor's Warranties" shall mean all of them.
"VIE Documents" means each agreement or contract entered into between the Company or any of its Subsidiaries, on the one hand, and the relevant VIE Entity or the shareholders of the relevant VIE Entity, on the other hand, which enables the Company or the relevant Subsidiary to control the relevant VIE Entity, including the agreements or contracts set out in folder 8 of the Data Room.
"VIE Entities" means, collectively, (a) the Guangzhou VIE Entity and the Shenzhen VIE Entity and (b) any other company which is not directly or indirectly owned by the Company but the Company is deemed to control such company through the VIE Documents, and "VIE Entity" means any one of them.
"VIE Restructuring Documents" means any agreements or contracts which are necessary to: (i) implement, effectuate, perfect and consummate the transfer of equity interests in the Shenzhen VIE Entity held by [***] to [***] (the "Equity Transfer"), and (ii) establish, maintain and evidence the contractual arrangements pursuant to which the Company or the relevant Subsidiary controls and directs the operation and activities of the relevant VIE Entity and enjoys the economic benefits of, and bears the economic risks associated with, the relevant VIE Entity, including but not limited to the following documents:
(g)the consent for the release of Equity Pledges (《同意解除股权质押的证明》);
(h)the application for cancellation of registration of Equity Pledges (《股权出质注销登记中请书》);
(i)the consent letter for the Equity Transfer and other matters (《同意股权转让等事宜的同意函》);
(j)corporate approvals of the Group necessary for the execution and performance of VIE Restructuring Documents (including: (i) shareholder's decision (股东决定) of the Shenzhen WFOE, (ii) executive director's decision (执行董事决定) of the Shenzhen WFOE, (iii) shareholders' decision (股东决定) of the Shenzhen VIE Entity and (iv) executive director's decision (执行董事决定) of the Shenzhen VIE Entity);
(k)the equity transfer agreement in respect of the Equity Transfer (《股权转让协议》);
(l)the amended and restated articles of association of the Shenzhen VIE entity reflecting the change of shareholder as a result of the Equity Transfer;
(m)termination agreement relating to the termination of the VIE Documents (原VIE协议的《终止协议》);
(n)termination agreement relating to the termination of undertaking issued to [***]; and
(o)new VIE agreements (including: (i) call option agreement (《独家购买权协议》), (ii) power of attorney (《授权委托书》), (iii) equity pledge agreement (《股权质押协议》) and (iv) spousal consent letter (《配偶同意函》)).
"Warranty Claim" means any claim made by the Purchaser for breach of any Vendor's Warranty (including, for the avoidance of doubt, a Fundamental Warranty Claim and a Tax Warranty Claim).
1.2Recitals, schedules, etc.
References to this Agreement shall include the Recitals and Schedules which form part of this Agreement for all purposes. The Recitals do not create binding obligations on a Party. References in this Agreement to the "Recitals", "Schedules", "Clauses" and "Paragraphs" are references respectively to the recitals and schedules to and clauses and paragraphs of this Agreement.
1.3References to certain general terms
Unless the contrary intention appears, a reference in this Agreement to:
(a)"subsidiary" is to be construed in accordance with sections 13 to 15 of the Companies Ordinance;
(b)a "person" includes a reference to any individual, firm, company, corporation or other body corporate, government, state or agency of a state or any joint venture, association or partnership, works council or employee representative body (whether or not having separate legal personality);
(c)a statutory provision includes a reference to the statutory provision as modified or re-enacted or both from time to time before the date of this Agreement and any subordinate legislation made under the statutory provision (as so modified or re-enacted) before the date of this Agreement;
(d)a party being liable to another party, or to liability, includes any liability in equity, contract or tort (including negligence) or under the Misrepresentation Ordinance (Chapter 284 of the Laws of Hong Kong);
(e)words in one gender include any other gender;
(f)a document (including this Agreement) includes any variation, amendment or replacement of it;
(g)a document in the "agreed form" is a reference to that document in the form that the Parties have confirmed by exchange of emails on or around the date of this Agreement is to be treated for the purposes of this Agreement as being the "agreed form", with such amendments as the Parties may subsequently confirm prior to Completion;
(h)a person includes a reference to that person's legal personal representatives, successors and permitted assigns;
(i)"Parties" or a "Party", unless the context otherwise requires, includes a reference to the relevant Party's successors and permitted assigns;
(j)the singular includes the plural and vice versa;
(k)times of the day are to that time in Hong Kong and references to a day are to a period of twenty-four (24) hours running from midnight to midnight;
(l)any "matter" shall be deemed to include any fact, matter, event or circumstance (including any omission to act);
(m)any Hong Kong legal term for any action, remedy, method of judicial proceeding, legal document, legal status, court, official or any legal concept or thing shall, in respect of any jurisdiction other than Hong Kong, be deemed to include what most nearly approximates in that jurisdiction to the Hong Kong legal term, and to any Hong Kong statute shall be construed so as to include equivalent or analogous laws of any other jurisdiction; and
(n)"writing" or "written" include any method of reproducing words or text in a legible and non-transitory form and, for the avoidance of doubt, shall include text transmitted by e-mail.
1.4Unless the context otherwise requires, if a period of time dates from a given day or the day of an act or event, it is to be calculated exclusive of that day, when calculating the period of time before which, within which or following which any act is to be done or step taken under this Agreement, the date that is the reference date in calculating that period shall be excluded. If the last day of that period is not a Business Day, the period in question shall end on the next Business Day.
1.5The ejusdem generis principle of construction shall not apply to this Agreement. Accordingly, general words shall not be given a restrictive meaning by reason of their being preceded or followed by words indicating a particular class of acts, matters or things or by examples falling within the general words. Any phrase introduced by the term "other", "include", "including" or other similar expression are not used as, nor are they to be interpreted as words of limitation and when introducing an example do not limit the meaning of the words to which the example relates to that example or examples of a similar kind.
1.6A reference to the Vendor's knowledge is deemed to include the awareness or knowledge, information and belief which [***], [***], [***], [***], [***] and [***], would have if he or she had made all reasonable enquiries of the Founders and/or the management of the Group Companies.
1.7The headings in this Agreement do not affect its interpretation.
2.SALE OF SHARES
2.1Sale and Purchase of Sale Shares
(a)Subject to and in accordance with the terms and conditions of this Agreement, the Vendor agrees to sell and transfer to the Purchaser, and the Purchaser agrees to purchase from the Vendor, the legal and beneficial interest of the Sale Shares and together with any and all rights which now are, or at any time hereafter may become, attached to them, including all rights to any dividend or other distribution declared, made or paid after the date of Completion, free and clear of any Encumbrances.
(b)The Purchaser shall pay to the Vendor the Purchase Price in consideration for the sale and purchase of the Sale Shares, payable in accordance with this Agreement.
(c)Subject to and contingent upon Completion, the Vendor hereby irrevocably and unconditionally waives all pre-emption rights, rights of first refusal, redemption rights and rights of notice and any similar rights over and with respect to any of the Sale Shares and/or the Transaction conferred on it by the Existing Articles, the Existing Shareholders' Agreement or otherwise.
3.CONDITIONS
3.1Condition of Completion
(a)Completion is conditional on the following Conditions continuing to be fulfilled or having been waived in accordance with this Agreement:
(a)(i) the Fundamental Warranties are true and correct, (ii) the other representations, warranties, and covenants of the Vendor under this Agreement are true and correct in all material respects, and (iii) there has been no material breach of this Agreement by the Vendor;
(b)no event, circumstance or change has occurred that, individually or in the aggregate with any other events, circumstances, or changes, has had or could reasonably be expected to have a material adverse effect on the prospects, operations, or financial condition of any member of the Group;
(c)the Vendor has:
(i)completed the Announcement 7 Reporting (as defined under the 2024 SPA) (the "2024 Announcement 7 Reporting"); and
(ii)to the extent that payment of such Tax is required by the relevant Tax Authority in the PRC to be made on or prior to Completion, paid all necessary Tax in connection with the transaction contemplated under the 2024 SPA (the "2024 Transaction") under Announcement 7 and/or Announcement 37 (including any late payment or penalty), and delivered the Acceptable Evidence of Payment in respect of such Tax payment to the Vendor; and
(d)the Vendor has delivered to the Purchaser the VIE Restructuring Documents in such form to the satisfaction of the Purchaser, each duly executed but left undated by the relevant parties thereto.
3.2Vendor's obligations
(a)The Vendor shall use its best endeavours to:
(i)ensure that the Conditions set out in Clauses 3.1(a) and 3.1(b) remain satisfied at all times after the date of this Agreement; and
(ii)achieve or procure satisfaction of the Conditions set out in Clause 3.1(c) as soon as possible after the date of this Agreement and in any event not later than the Completion Date.
(b)If, at any time, the Vendor becomes aware of a fact or circumstance that might prevent a Condition being satisfied, it shall immediately inform the Purchaser.
(c)The Vendor shall inform the Purchaser of the satisfaction of the relevant Condition immediately on becoming aware of the same.
(d)At any time on or before the Long Stop Date, the Purchaser may waive a Condition by notice to the Vendor on any terms it decides.
(e)If a Condition is not satisfied (or waived pursuant to Clause 3.2(d) by the Long Stop Date, the Purchaser may, in its sole discretion, terminate this Agreement (other than the Surviving Provisions) with immediate effect. Each Party's further rights and obligations shall cease immediately on termination, but termination of this Agreement does not affect a Party's accrued rights and obligations at the date of termination or its rights and obligations arising as a result of termination.
4.COMPLETION
4.1Time and place of Completion
Subject to the terms and conditions of this Agreement, Completion shall take place on the date that is within ten (10) Business Days after the fulfilment or waiver of the Conditions (or such other date as may be agreed between the Vendor and the Purchaser from time to time) (such date, the "Completion Date") at such place (or remotely via electronic exchange of documents and signatures) as may be agreed by the Parties.
4.2Vendor's obligations at Completion
(a)At Completion, the Vendor shall deliver, or cause to be delivered, to the Purchaser:
(i)the Share Transfer Form duly executed by it in respect of the Sale Shares in favour of the Purchaser;
(ii)(if not already delivered) a copy (certified to be a true copy by a director or registered office provider or company secretary of the Vendor) of the resolutions of the board of directors of the Vendor or minutes of a duly held meeting of the board of directors of the Vendor approving:
(A)the sale of the Sale Shares by the Vendor to the Purchaser; and
(B)the execution and delivery by the Vendor of the Transaction Documents to which it is a party and any other documents to be delivered by it at Completion, and the performance by it of its obligations thereunder;
(iii)a copy (certified to be a true copy by a director or registered office provider or company secretary of the Company) of the resolutions of the board of directors of the Company or minutes of a duly held meeting of the board of directors of the Company, pursuant to which all directors of the Company approve:
(A)the execution and delivery by the Company of each Transaction Document to which it is a party and the performance by it of its obligations thereunder;
(B)the reclassification of the Sale Shares into Class C Shares at Completion;
(C)the transfer of the Sale Shares, the update of the register of members of the Company to reflect the Purchaser as the registered owner of the Sale Shares as at the Completion Date, the cancellation of the existing share certificate of the Sale Shares in the name of the Vendor and the issuance of new share certificate(s) in the name of the Purchaser with respect to the Sale Shares (including the new share certificate(s) in the name of the Purchaser with respect to the Sale Shares after the reclassification into Class C Shares), in each case in accordance with the terms of this Agreement;
(D)the update of the register of directors of the Company to reflect change of directors at Completion;
(E)the adoption of the Amended Articles; and
(F)the resignation of Danny Sheng Wu Yeung as a director and the Chief Executive Officer and Yin Pang Chang as a director of the Company and appointing such person nominated by each of the Purchaser and Dennis Lo, each as a director of the Company, in each case with effect from Completion.
(iv)a copy (certified to be a true copy by a director or registered office provider or company secretary of the Company) of the unanimous written resolutions of the shareholders of the Company, pursuant to which all the shareholders of the
Company (x) waive all pre-emption rights, rights of first refusal, redemption rights and rights of notice and any similar rights over and with respect to any of the Sale Shares and/or the Transaction conferred on the relevant shareholder under the Existing Articles, the Existing Shareholders' Agreement or otherwise (the "Existing Shareholders' Waiver") and (y) approve:
(A)the transfer of the Sale Shares;
(B)the amendment and restatement of the Existing Shareholders' Agreement, and the execution and delivery by the Company of the Shareholders' Agreement;
(C)the reclassification of the Sale Shares into Class C Shares at Completion;
(D)the adoption of the Amended Articles;
(E)the Existing Shareholders' Waiver; and
(F)the resignation of Danny Sheng Wu Yeung as a director and the Chief Executive Officer and Yin Pan Cheng as director of the Company and appointing such person nominated by each of the Purchaser and Dennis Lo, each as a director of the Company, in each case with effect from Completion.
(v)(if not already executed and delivered) each of the Transaction Documents (other than this Agreement) duly executed and delivered by each party thereto other than the Purchaser;
(vi)a copy (certified to be a true copy by a director or company secretary of the Company) of (A) the updated register of members of the Company reflecting (x) the Purchaser as the registered owner of the Sale Shares as at the Completion Date; and (y) the reclassification of the Sale Shares into Class C Shares at Completion; and (B) the updated register of directors of the Company reflecting change of directors at Completion;
(vii)(if not already delivered) copies of the VIE Restructuring Documents, each duly executed by the relevant parties thereto; and
(viii)(if not already delivered) the evidence to the satisfaction of the Purchaser, which can prove that the 2024 Announcement 7 Reporting has been duly submitted to the relevant Tax Authority in the PRC, and to the extent any Tax is required by the relevant Tax Authority in the PRC to be paid thereon on or prior to Completion, the Acceptable Evidence of Payment in respect of such Tax payment.
(b)At Completion, the Vendor shall deliver, or cause to be delivered, to the Company:
(i)the existing share certificate in respect of the Sale Shares in the name of the Vendor; and
(ii)a resignation letter from each of Danny Sheng Wu Yeung and Yin Pan Cheng, resigning from the board of directors of the Company and in each case confirming no outstanding claims or rights against the Company arising out of his directorship or the termination of his directorship.
4.3Purchaser's obligations at Completion
The Purchaser shall:
(a)approve the written resolutions of the shareholders of the Company under Clause 4.2(a)(iv) and procure the director of the Company nominated by the Purchaser to approve the resolutions of the board of directors of the Company under Clause 4.2(a)(iii);
(b)against compliance by the Vendor with Clause 4.2, deliver, or cause to be delivered, to the Vendor:
(i)the Share Transfer Form duly executed by it in respect of the Sale Shares; and
(ii)(if not already executed and delivered) each of the Transaction Documents to which it is a party (other than this Agreement) duly executed and delivered by the Purchaser; and
(c)against compliance by the Vendor with Clause 4.2, pay, or procure the payment of, an amount in USD equal to the Completion Amount to the Vendor by electronic transfer of immediately available funds to the Vendor's Bank Account, and deliver to the Vendor the form MT103 (or similar evidence of irrevocable payment) evidencing such payment and the provision of such evidence to the Vendor shall be deemed to satisfy the Purchaser's payment obligations under this Clause 4.3(c). In the case of any telegraphic transfers by the Purchaser of the Completion Amount or the Holdback Amount under this Agreement, any bank charges resulting therefrom shall be borne by the Purchaser.
4.4Default
(a)If Completion does not take place on the Completion Date because either the Vendor or the Purchaser (the "Defaulting Party") fails to comply with any of its respective obligations under (with respect to the Vendor) Clause 4.2 and (with respect to the Purchaser) Clause 4.3 (whether such failure by the Defaulting Party amounts to a repudiatory breach or not), the other Party (the "Non-Defaulting Party") may by Notice to the Defaulting Party:
(i)proceed to Completion to the extent reasonably practicable;
(ii)postpone Completion to such date as the Non-Defaulting Party may specify (being a date not later than the Long Stop Date); or
(iii)terminate this Agreement.
(b)If the Non-Defaulting Party postpones Completion to another date in accordance with Clause 4.4(a)(ii), the provisions of this Agreement apply as if that other date is the Completion Date.
(c)If the Non-Defaulting Party terminates this Agreement pursuant to Clause 4.4(a)(iii), each Party's further rights and obligations shall cease immediately on termination (excluding those under the Surviving Provisions), but termination does not affect a Party's accrued rights and obligations at the date of termination.
5.ANNOUNCEMENT 7
5.12026 Announcement 7 Reporting
5.1The Vendor shall prepare and submit (or procure to submit) the tax reporting in connection with the sale and purchase of the Sale Shares pursuant to this Agreement under Announcement 7 and/or Announcement 37 (the "2026 Announcement 7 Reporting") as soon as practicable and in any case within the statutory period in accordance with applicable Laws.
5.2The Vendor shall deliver to the Purchaser such evidence to the satisfaction of the Purchaser which can prove that the 2026 Announcement 7 Reporting has been duly submitted to the relevant Tax Authority in the PRC within ten (10) Business Days after the Completion Date.
Payment of Tax in respect of the Transaction and/or the 2024 Transaction
5.3If the relevant Tax Authority in the PRC determines or assesses after Completion that, under Announcement 7 and/or Announcement 37, Tax is liable to be paid in connection with the Transaction and/or the 2024 Transaction, the Vendor shall:
(a)prepare and submit the tax return in relation to the Transaction and/or the 2024 Transaction (as the case may be) to the relevant Tax Authority in the PRC;
(b)pay (or procure the payment of) the final assessed Tax amount under Announcement 7 and/or Announcement 37 in connection with the Transaction and/or the 2024 Transaction (as the case may be), in each case within the time period prescribed by applicable Laws and instructions from the relevant Tax Authority in the PRC; and
(c)deliver to the Purchaser the Acceptable Evidence of Payment in respect of the relevant Tax payment, in each case within five (5) Business Days after such payment.
5.4The Vendor shall, at the request of the Purchaser, provide the Purchaser with such assistance as the Purchaser may reasonably require in connection with the 2024 Announcement 7 Reporting, the 2026 Announcement 7 Reporting and/or any matters in connection with any withholding obligations of the Purchaser under the Announcement 7 and/or Announcement 37.
5.5The Vendor shall indemnify the Purchaser against the losses suffered directly or indirectly by the Purchaser and/or any Group Company as a result of the failure by the Vendor to submit any tax return or pay (or procure the payment of) the final assessed Tax amount in connection with the Transaction and/or the 2024 Transaction (as the case may be) under Announcement 7 and/or Announcement 37 within the time period prescribed by applicable Laws and instructions from the relevant Tax Authority in the PRC.
6.RELEASE OF HOLDBACK AMOUNT
6.1Subject to the occurrence of all of the following events, within ten (10) Business Days following the occurrence of last such event, the Purchaser shall pay (or procure the payment of) the Holdback Amount to the Vendor by electronic transfer of immediately available funds to the Vendor's Bank Account:
(a) the Purchaser having received evidence satisfactory to the Purchaser that:
(i)the 2026 Announcement 7 Reporting in accordance with Clause 5.1 above has been duly submitted by the Vendor to the relevant Tax Authority in accordance with applicable Laws; and
(ii)either (x) the relevant Tax Authority has confirmed that no Tax is payable in connection with each of the submissions made in the 2026 Announcement 7 Reporting and the 2024 Announcement 7 Reporting, or (y) all Tax assessed by the relevant Tax Authority in connection with each of the submissions made in the 2026 Announcement 7 Reporting and the 2024 Announcement 7 Reporting has been paid in full in compliance with applicable Laws (including any late payment fee or penalty) and the Purchaser has received Acceptable Evidence of Payment for such Tax with respect to such submission; and
(b)the Purchaser having received evidence satisfactory to the Purchaser that all necessary PRC Tax and AMR Filings have been completed by the Vendor in accordance with Clause 8.3 below.
6.2For the avoidance of doubt, the Purchaser shall have no obligation to pay the Holdback Amount if any part of Clauses 6.1(a) and 6.1(b) is not complied with.
7.INDEMNITIES
7.1Tax Indemnity
(a)The Vendor covenants with the Purchaser to pay to the Purchaser an amount equal to [***] of any Tax Liability of the Group arising as a consequence of or by reference to any Tax Event which occurred on or before Completion for the purposes of any Tax or arising in respect of or by reference to any income, profits or gains to the extent that such
income, profits or gains were earned, accrued or received on or before Completion or in respect of a period ending on or before Completion.
(b)For the purposes of this Clause 7.1:
(i)"Tax Liability" of the Group shall mean liabilities or any increase in the liabilities of the Group to make actual payments of Tax; and
(ii)"Tax Event" shall mean any transaction, event, circumstance, action or omission, including any change in the residence of any person for the purposes of any Tax, which has any Tax implication.
7.2Other Indemnity
The Vendor shall indemnify the Purchaser against any losses suffered directly or indirectly by the Purchaser and/or any Group Company as a result of: (a) the failure by the Vendor to complete (or procure the completion of) the PRC Tax and AMR Filings pursuant to this Agreement and the 2024 SPA; and (b) for so long as the VIE Documents remain in place, any non-compliance by [***] of any provision under the VIE Documents, any action or omission of [***] as a holder of equity interests in the Shenzhen VIE Entity or otherwise as a result of [***] remaining as a party to any of the VIE Documents or as a holder of equity interests in the Shenzhen VIE Entity.
8.OTHER POST-COMPLETION UNDERTAKINGS
8.1As soon as possible and in any event within three (3) Business Days after the Completion Date, the Vendor shall deliver, or cause to be delivered, to the Purchaser:
(a)the original new share certificate(s) in the name of the Purchaser with respect to the Sale Shares (including the original new share certificate(s) in the name of the Purchaser with respect to the Sale Shares after the reclassification into Class C Shares); and
(b)documentary evidence which shows that the relevant filings have been made by the Company with the Cayman Islands Registrar of Companies for the updated register of directors and the adoption of the Amended Articles.
8.2The Purchaser shall provide reasonable assistance to the Vendor as necessary in relation to the filings and disclosures relating to the Group or this transaction as required to be made by the Vendor under U.S. securities laws and NASDAQ rules. Furthermore, the Purchaser shall use reasonable endeavours to procure the Company to allow the Vendor's auditors access to the Group's books, records, and facilities as reasonably necessary to enable the Vendor's auditors to complete their audit of the Vendor’s financial statements for the fiscal years ended December 31, 2025, and 2026 and to prepare standalone audited and pro forma financial statements as required under U.S. securities laws and NASDAQ rules.
8.3As soon as possible after the Completion Date and in any case within the period required under the applicable Laws, the Vendor shall complete, and shall procure the relevant Group Company to complete, all filings, registrations or recordings required to: (a) implement, effectuate, perfect and consummate the Equity Transfer, including but not limited to the completion of the relevant tax declarations and the registration of the Equity Transfer with the competent AMR; and (b) the registration and perfection of the pledge over 100% equity interests in the Shenzhen VIE Entity granted by [***] in favour of the Shenzhen WFOE with the competent AMR (the "PRC Tax and AMR Filings"). The Vendor shall deliver to the Purchaser such evidence to the satisfaction of the Purchaser which can prove that all necessary PRC Tax and AMR Filings have been completed. For so long as [***] remains as a party to any VIE Document or a holder of equity interests in the Shenzhen VIE Entity, the Vendor shall procure that [***] strictly complies with, observe and perform all obligations under each of the VIE Documents and [***] shall act in accordance with the instructions of the Company and the Shenzhen WFOE.
8.4Within fifteen (15) Business Days after the Completion Date, the Vendor shall deliver, or cause to be delivered, to the Purchaser the certificate of incumbency of the Company (which shall include
the certification on the Company's good standing) issued by the registered office provider of the Company within one month of the Completion Date.
9.REPRESENTATIONS AND WARRANTIES GIVEN BY THE VENDOR
9.1The Vendor represents and warrants to the Purchaser that each of the statements set out in Schedule 2 is true, accurate and not misleading as at (i) the date of this Agreement and (ii) the Completion Date, in each case, by reference to the facts and circumstances then subsisting.
9.2The Vendor's Warranties are qualified by the facts and circumstances Disclosed in this Agreement, the Disclosure Letter or in any of the documents annexed to the Disclosure Letter. No other knowledge of the Purchaser relating to any Group Company (actual, constructive or imputed) prevents or limits a claim made by the Purchaser for breach of Clause 9.1 above. The Vendor shall not invoke the Purchaser's knowledge (actual, constructive or imputed) of a fact or circumstance which might make a Vendor's Warranty untrue, inaccurate or misleading as a defence to a claim for breach of Clause 9.1 above or to reduce any amount recoverable.
9.3The Vendor's liability for any Warranty Claim shall be limited or excluded, as the case may be, as set out in Schedule 3.
9.4Each Vendor's Warranty is to be construed independently and (except where this Agreement provides otherwise) is not limited by a provision of this Agreement or another Vendor's Warranty.
10.REPRESENTATIONS AND WARRANTIES GIVEN BY THE PURCHASER
The Purchaser represents and warrants to the Vendor that each of the statements set out under this Clause 10 is true, accurate and not misleading as at (i) the date of this Agreement and (ii) the Completion Date, in each case, by reference to the facts and circumstances then subsisting:
(a)Capacity and authority
(i)It is duly incorporated, validly existing and in good standing under the laws of its jurisdiction of incorporation, and has all necessary corporate power and authority to enter into this Agreement and each other Transaction Document to which it is a party, to carry out its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby.
(ii)No petition has been presented, no order has been made, or resolution passed for the winding up of it or for the appointment of a liquidator, provisional liquidator, receiver, administrator or manager of it. No liquidator, provisional liquidator, receiver, administrator or manager has been appointed in relation to it, nor any notice given of the appointment of any such person, over the whole or part of its business or assets.
(iii)This Agreement has been (and each Transaction Document upon execution and delivery will be) duly executed and delivered by it and constitutes (and each Transaction Document upon execution and delivery will constitute) legal, valid and binding obligations of it, enforceable against it in accordance with its (and their) respective terms.
(iv)The execution, delivery and performance by it of this Agreement and/or each of the Transaction Documents to which it is a party and the consummation of the transactions contemplated in the Transaction Documents will not result in a breach of, or constitute a default under (as applicable):
(A)any provision of its articles of association or equivalent constitutional documents;
(B)any contract, agreement or instrument to which it is a party or by which it is bound; and
(C)a breach of any order, judgment or decree of any court or Government Authority to which it is a party or by which it is bound or submits,
in each case, where such breach or default (as the case may be) would or would be reasonably likely to have a material adverse effect on its ability to perform its obligations under this Agreement and/or any of the Transaction Documents to which it is a party.
11.CONFIDENTIALITY AND ANNOUNCEMENTS
11.1Confidential Information
(a)Subject to Clause 11.1(b), each Party undertakes to the other Party that it shall (and shall procure that its Affiliates shall):
(i)not, without the prior written consent of the other Party, use or disclose to any unauthorised person Confidential Information it has or acquires; and
(ii)make every effort to prevent the unauthorised use or disclosure of Confidential Information.
(b)The confidentiality obligations under Clause 11.1(a) do not apply to:
(i)to the Representatives of a Party whose function requires it to have the relevant Confidential Information, provided that they are subject to obligations to keep such information confidential on the same or equivalent confidentiality terms as Clause 11.1(a);
(ii)any information that is required to be disclosed by applicable Law or any requirement of any competent Government Authority or pursuant to applicable rules or regulations of any relevant regulatory, administrative or supervisory body (including any relevant stock exchange or securities council), whether or not it has the force of law; provided that, to the extent legally permissible and so far as reasonably practicable, the disclosure shall be made after consultation with the other Party and after taking into account the other Party's reasonable requirements as to its timing, content and manner of making or despatch; and
(iii)the disclosure of information by any Party to its Affiliate, its or any of its Affiliate's consultants, auditors, advisors, employees, officers or directors so long as such persons are subject to obligations to keep such information confidential on the same or equivalent confidentiality terms as Clause 11.1(a).
11.2Announcements or releases
(a)A Party may not make press or other announcements, communications, circulars or releases relating to this Agreement and the matters referred to in this Agreement and the other Transaction Documents without the prior approval of the other Party to the form and manner of the announcement or release unless and to the extent that disclosure is required or requested to be made by a Party by applicable Law, or any requirement of any competent Government Authority or pursuant to applicable rules or regulations of any relevant regulatory, administrative or supervisory body (including any relevant stock exchange or securities council), whether or not it has the force of law.
(b)To the extent that any announcement or release is so required or requested to be made by any Party in accordance with Clause 11.2(a), the Party making the announcement or release must, as far as reasonably practicable and legally permissible, consult with the other Party as to the form, content and timing of any such announcement or release.
(c)Without the prior written consent of the Purchaser, the Vendor shall, and shall cause its Affiliates not to: (i) use in advertising, publicity, announcements, or otherwise the name of "[***]", "[***]", "[***]", "[***]", "[***]", "[***]", "[***]", or any Affiliate of [***], either alone or in combination thereof, the associated devices and logos of the above brands, or any company name, trade name, trademark, service mark, domain name, device, design, symbol or any abbreviation, contraction or simulation thereof owned or used by the Purchaser or any of its Affiliates; or (ii) represent, directly or indirectly, that
any product or services provided by the Vendor and/or its Affiliates, the Company and/or its Affiliates has been approved or endorsed by the Purchaser or any of its Affiliates.
12.COSTS, EXPENSES AND FEES
12.1Costs, expenses and fees
(a)Subject to Clause 12.1(b), except where this Agreement or other Transaction Documents provide otherwise, each Party shall pay its own costs, expenses and fees (including any fees of any legal or other professional advisors) in connection with the negotiation, preparation, execution, completion and performance of this Agreement and each document referred to hereunder.
(b)The Vendor shall bear any and all costs, expenses and fees incurred by the Purchaser in connection with the negotiation, preparation, execution, completion and performance of this Agreement and each document referred to hereunder (including any costs, expenses and fees in connection with conducting Due Diligence and any fees of any legal or other professional advisors) up to an aggregate amount of [***].
12.2Taxes
The Vendor shall bear and be responsible for all applicable Taxes (if any) payable under applicable Law in connection with this Agreement and/or the sale of Sale Shares from the Vendor to the Purchaser, and shall indemnify and hold harmless the Purchaser and its Affiliates from and against any loss, liability, costs or expenses suffered or incurred (directly or indirectly) by the Purchaser or its Affiliates which arises out of or in connection with any failure by the Vendor to comply with its obligations under this Clause 12.2.
13.NOTICES
13.1Address for delivery of notices
(a)Any notice or other communication given or made under this Agreement (a "Notice") shall be:
(i)in writing;
(ii)unless otherwise specified, in English; and
(iii)delivered personally or sent by courier by an internationally recognised courier company or by electronic mail (e-mail), to the Party due to receive the Notice at its address or e-mail address set out in Clause 13.1(b) or to such other address or e-mail address as notified by one Party to the other Party in accordance with Clause 13.3.
(b)The address or e-mail address referred to in Clause 13.1(a) is:
(i)if to the Vendor:
Address: Unit 703-706, K11 Atelier King's Road, 728 King's Road, Quarry Bay, Hong Kong
E-mail address: [***]
[***]
Attention: [***]
[***]
(ii)if to the Purchaser:
Address: [***]
Attention: [***]
with a copy to:
c/o [***]
Attention: [***]
E-mail address: [***]
[***]
13.2Delivery of Notices
In the absence of evidence of earlier receipt, a Notice shall be deemed to have been duly given or made:
(a)if sent by personal delivery, upon delivery at the address of the relevant Party;
(b)if sent by courier, two (2) Business Days (if posted within Hong Kong) or five (5) Business Days (if posted outside of Hong Kong) after the date of posting; and
(c)if sent by e-mail:
(i)before 5:00 p.m. on a Business Day, at the time when the e-mail was sent (as recorded on the device from which the sender sent the email); or
(ii)after 5:00 p.m. on a Business Day, at 9:00 a.m. on the next Business Day following the day when the e-mail was sent (as recorded on the device from which the sender sent the email),
(iii)in each case, unless the sender receives an automated message that the email has not been delivered.
13.3Changes to addresses
A Party may notify the other Party of a change to its relevant address or e-mail address for the purposes of this Clause 13, provided that such notification shall only be effective on:
(a)the date specified in the notification as the date on which the change is to take place; or
(b)if no date is specified or the date specified is earlier than the date on which a notice is deemed under Clause 13.2 to have been duly given, the date on which such notice is so deemed to have been duly given.
14.ASSIGNMENT
(a)A Party must not assign, transfer, dispose of or otherwise part with any of its rights under this Agreement without the prior written consent of the other Party. Any such assignment without such prior written consent shall be null and void.
(b)This Agreement shall be binding upon, inure to the benefit of, and be enforceable by, the Parties and their respective heirs, successors and permitted assigns.
15.MISCELLANEOUS
15.1Discretion in exercising rights
A Party may exercise a right or remedy or give or refuse its consent in any way it considers appropriate (including by imposing conditions), unless this Agreement expressly states otherwise.
15.2Failure to exercise rights
Except as otherwise set out in this Agreement or expressly agreed by the Parties in writing, any partial exercise, failure to exercise, or delay in exercising, a right or remedy provided under this Agreement or by Law does not impair or operate as a waiver or prevent or restrict any further or other exercise of that or any other right or remedy in accordance with this Agreement. No single or partial exercise of a right or remedy provided by this Agreement or by Law prevents further exercise of the right or remedy or the exercise of another right or remedy.
15.3No liability for loss
Except as otherwise set out in this Agreement, a Party is not liable for loss caused by the exercise or attempted exercise of, failure to exercise, or delay in exercising a right or remedy that is available to it under this Agreement.
15.4Approvals and consents
By giving its approval or consent, a Party does not make or give any warranty or representation as to any circumstance relating to the subject matter of the consent or approval.
15.5Remedies cumulative
The rights and remedies provided in this Agreement are cumulative and in addition to other rights and remedies given by Law independently of this Agreement.
15.6Variation and waiver
(a)This Agreement may only be varied if it is in writing and signed by or on behalf of the Vendor and the Purchaser. For the avoidance of doubt, "varied" shall include any amendment, supplement, deletion or replacement howsoever effected.
(b)Unless expressly agreed, no variation or waiver shall constitute a general variation or waiver of any provisions of this Agreement, nor shall it affect any rights, obligations or liabilities under this Agreement that have already accrued up to the date of variation or waiver, and the rights and obligations of the Parties under this Agreement shall remain in full force, except and only to the extent that they are so varied or waived.
15.7Further steps
Each Party agrees, at its own expense but subject to Clause 12, to take all actions, execute or procure to be done and executed all such further acts, deeds, things and documents as may be necessary, by Law or as may be necessary or reasonably requested by the Vendor or the Purchaser, to give full effect to the terms of the Transaction Documents.
15.8Time of the essence
Except as otherwise set out in this Agreement, time is of the essence of this Agreement in respect of any date or period determined under this Agreement.
15.9Entire agreement
(a)This Agreement, together with other Transaction Documents, represents the entire understanding, and constitutes the entire agreements, of the Parties about its subject matter and supersedes all previous agreements between the Parties (whether written or oral), understandings and negotiations on that subject matter.
(b)Nothing in this Clause 15.9 shall have the effect of limiting any liability arising from fraud, wilful misconduct or wilful concealment.
15.10Severability
If any provision or of any part of a provision of this Agreement is invalid or void, unenforceable or illegal in any jurisdiction, such provision shall apply with whatever deletion or modification is necessary so that the provision is legal, valid and enforceable and gives effect to the commercial intention of the Parties. The remainder of this Agreement remains in full force and effect and the validity or enforceability of that provision in any other jurisdiction shall not be affected. This Clause 15.10 has no effect if the severance alters the basic nature of this Agreement.
15.11Third Parties
Except as expressly provided in this Agreement, a person who is not a party to this Agreement has no right under the Third Parties Ordinance to enforce any term of this Agreement but this does not affect any right or remedy of a third party which exists or is available apart from the Third Parties Ordinance.
16.GOVERNING LAW AND DISPUTE RESOLUTIONS
(a)The construction, validity and performance of this Agreement and all non-contractual obligations (if any) arising from or connected with this Agreement shall be governed by the laws in force in Hong Kong.
(b)The Parties agree to negotiate in good faith to resolve any dispute, controversy, difference or claim arising out of or in connection with this Agreement or its subject matter (including a dispute regarding the existence, validity, formation, effect, interpretation, performance or termination of this Agreement) or any dispute regarding non-contractual obligations arising out of this Agreement (a "Dispute"). In the event the negotiations between the Parties cannot resolve any such Dispute within thirty (30) days, such Dispute shall be referred to and finally settled by arbitration administered by the Hong Kong International Arbitration Centre (the "HKIAC") for arbitration in Hong Kong.
(c)The arbitration shall be conducted in accordance with the HKIAC Administered Arbitration Rules in force at the time of the initiation of the arbitration, which rules are deemed to be incorporated by reference into this Clause 16 save to the extent that they are inconsistent with the express terms of this Agreement.
(d)The seat or legal place of arbitration shall be Hong Kong, and the arbitral proceedings shall be conducted in English.
(e)The arbitration tribunal shall consist of three (3) arbitrators. The claimant(s) and the respondent(s) shall each nominate one (1) arbitrator, and the two (2) arbitrators thus appointed shall nominate the third arbitrator who shall be the presiding arbitrator; if the claimant(s) or the respondent(s) fails to appoint the arbitrator within fourteen (14) days of a request to do so from the other Party, or if the two (2) arbitrators fail to nominate the third arbitrator within fourteen (14) days after the appointment of the second arbitrator, the appointment shall be made, upon request of a Party, by the HKIAC in accordance with the HKIAC Administered Arbitration Rules.
(f)Any award of the arbitral tribunal shall be final and binding upon the Parties from the day it is made, and the Parties undertake to carry out the award without delay.
(g)The Parties shall have the right to seek interim injunctive relief or other interim relief from a court of competent jurisdiction, both before or after the arbitral tribunal has been appointed, at any time until the arbitral tribunal has made its final award.
17.COUNTERPARTS
This Agreement may be signed in any number of counterparts, all of which taken together shall constitute one and the same instrument. Facsimile signatures, electronic signatures or signatures sent by email attachment shall be valid and binding to the same extent as original signatures.
Schedule 1
GROUP COMPANIES
GROUP COMPANIES
THE COMPANY | |||||||||||
Company name | : | Insighta Holdings Limited | |||||||||
Company number | : | 398503 | |||||||||
Date of incorporation | : | 17 March 2023 | |||||||||
Place of incorporation | : | Cayman Islands | |||||||||
Address of registered office | : | The Office of Vistra (Cayman) Limited, P. O. Box 31119 Grand Pavilion, Hibiscus Way, 802 West Bay Road, Grand Cayman, KY1 - 1205 Cayman Islands | |||||||||
Authorised share capital | : | US$50,000.00 divided into 150,000,000 Class A Shares, 250,000,000 Class B ordinary shares and 100,000,000 Class C Shares each of a nominal or par value of US$0.0001 | |||||||||
Issued share capital | : | US$200.30 divided into 1,053,000 Class A Shares, and 650,000 Class B ordinary shares and 300,000 Class C Shares each of a nominal or par value of US$0.0001 | |||||||||
Directors | : | [***] [***] [***] [***] [***] [***] | |||||||||
Shareholders (immediately before Completion) | : | Shareholder | Number of Shares | ||||||||
[***] | [***] Class A Shares | ||||||||||
Prenetics Global Limited | 700,000 Class A Shares | ||||||||||
[***] | [***] Class A Shares | ||||||||||
[***] | [***] Class A Shares | ||||||||||
[***] | [***] Class B ordinary shares | ||||||||||
[***] | [***] Class C Shares | ||||||||||
INSIGHTA HK | |||||||||||
Company name | : | Insighta Limited (明察康健有限公司) | |||||||||
Business registration number | : | 74748355 | |||||||||
Date of incorporation | : | 5 January 2023 | |||||||||
Place of incorporation | : | Hong Kong | |||||||||
Address of registered office | : | 21/F, Edinburgh Tower, The Landmark, 15 Queen's Road, Central, Hong Kong | |||||||||
Share capital | : | HKD[***] divided into [***] ordinary shares | |||||||||
Directors | : | [***] [***] | |||||||||
Shareholder(s) | : | Shareholder | Number of Shares | ||||||||
[***] | [***] ordinary shares | ||||||||||
SHENZHEN WFOE | |||||||||||
Company name | : | 明察康健(深圳)科技有限责任公司 | |||||||||
Unified social credit code | : | [***] | |||||||||
Date of incorporation | : | 4 September 2023 | |||||||||
Place of incorporation | : | PRC | |||||||||
Address of registered office | : | 214E, Building 543, Bagualing Industrial Zone, Bagua Second Road, Hualin Community, Yuanling Street, Futian District, Shenzhen (深圳市福田区园岭街道华林社区八卦二路八卦岭工业区543栋214E) | |||||||||
Registered capital (and Paid-up Capital) | : | RMB40,000,000 (RMB [***] paid up) | |||||||||
Director(s) and Supervisor(s) | : | [***] (Executive Director and General Manager) [***] (Supervisor) | |||||||||
Legal representative | : | [***] | |||||||||
Shareholder(s) | : | Shareholder | Paid-up Capital | ||||||||
[***] | RMB [***] | ||||||||||
GUANGZHOU VIE ENTITY | |||||||||||
Company name | : | Insighta Bio-technology (Guangzhou) Co., Ltd. | |||||||||
Unified social credit code | : | [***] | |||||||||
Date of incorporation | : | 15 November 2022 | |||||||||
Place of incorporation | : | PRC | |||||||||
Address of registered office | : | Unit C301, 3/F, Building 4, No.9, Luoxuan Fourth Road, International Biology Island, Huangpu District, Guangzhou (广州市黄埔区国际生物岛螺旋四路9号4栋第三层C301单元) | |||||||||
Registered capital (and Paid-up Capital) | : | RMB 20,000,000 (RMB [***] paid up) | |||||||||
Director(s) and Supervisor(s) | : | [***] (Executive Director and Manager) [***] (Supervisor) | |||||||||
Legal representative | : | [***] | |||||||||
Shareholder(s) | : | Shareholder | Paid-up Capital | ||||||||
[***] | RMB [***] | ||||||||||
SHENZHEN VIE ENTITY | |||||||||||
Company name | : | Insighta Industry (Shenzhen) Co., Ltd. | |||||||||
Unified social credit code | : | [***] | |||||||||
Date of incorporation | : | 19 August 2020 | |||||||||
Place of incorporation | : | PRC | |||||||||
Address of registered office | : | Unit 5H71A, Xinlikang Building, 3044 Xinghai Avenue, Nanshan Street, Qianhai Shenzhen-Hong Kong Cooperation Zone, Shenzhen(深圳市前海深港合作区南山街道兴海大道 3044 号信利康大厦 5H71A) | |||||||||
Registered capital (and Paid-up Capital) | : | RMB 100,000 ([***] paid up) | |||||||||
Director(s) and Supervisor(s) | : | [***] (Executive Director and Manager) [***] (Supervisor) | |||||||||
Legal representative | : | [***] | |||||||||
Shareholder(s) | Shareholder | Paid-up Capital | |||||||||
[***] ([***]%) [***] ([***]%) | RMB [***] RMB [***] | ||||||||||
Schedule 2
VENDOR'S WARRANTIES
VENDOR'S WARRANTIES
Capacity and Authority
1.1The Vendor is duly incorporated, validly existing and in good standing under the laws of its jurisdiction of incorporation, and has all necessary corporate power and authority to enter into this Agreement and each other Transaction Document to which it is a party, to carry out its obligations hereunder and thereunder and to consummate the transactions contemplated hereby and thereby.
1.2No petition has been presented, no order has been made, or resolution passed for the winding up of the Vendor or for the appointment of a liquidator, provisional liquidator, receiver, administrator or manager of the Vendor. No liquidator, provisional liquidator, receiver, administrator or manager has been appointed in relation to the Vendor, nor any notice given of the appointment of any such person, over the whole or part of the business or assets of the Vendor.
1.3This Agreement has been (and each Transaction Document upon execution and delivery will be) duly executed and delivered by the Vendor and constitutes (and each Transaction Document upon execution and delivery will constitute) legal, valid and binding obligations of the Vendor, enforceable against the same in accordance with its (and their) respective terms.
1.4The Vendor has the right, power and authority, and has taken all action necessary, to execute, deliver and exercise its respective rights, and perform its respective obligations, under this Agreement and/or each Transaction Document to which it is a party to be executed at or before Completion.
1.5The execution, delivery and performance by the Vendor of this Agreement and/or each of the Transaction Documents to which it is a party and the consummation of the transactions contemplated in the Transaction Documents will not result in a breach of, or constitute a default under (as applicable):
1.5.1any provision of its memorandum or articles of association or equivalent constitutional documents;
1.5.2any contract, agreement or instrument to which it is a party or by which it is bound; and
1.5.3a breach of any order, judgment or decree of any court or Government Authority to which it is a party or by which it is bound or submits,
in each case, where such breach or default (as the case may be) would or would be reasonably likely to have a material adverse effect on its ability to perform its obligations under this Agreement and/or any of the Transaction Documents to which it is a party.
1.Sale Shares
1.1The Vendor is the sole legal and beneficial owner of the Sale Shares. At Completion, the Vendor shall have transferred and delivered to the Purchaser full legal and beneficial ownership of the Sale Shares, free and clear of all Encumbrances.
1.2The Sale Shares have been properly allotted and issued and are fully paid or credited as fully paid.
1.3Other than in respect of this Agreement and the Existing Shareholders' Agreement, (a) there is no Encumbrance, and there is no agreement, arrangement or obligation to create or give an Encumbrance, in relation to any of the shares or unissued shares in the capital of the Company; and (b) no person has claimed to be entitled to an Encumbrance in relation to any of the Sale Shares.
1.4Other than in connection with this Agreement and the Director Service Agreements, there is no agreement, arrangement or obligation requiring the creation, allotment, issue, transfer, redemption or repayment of, or the grant to a person of the right (conditional or not) to require the allotment, issue, transfer, redemption or repayment of, a share in the capital of the Company (including an option or right of pre-emption or conversion).
1.5To the Vendor's knowledge, other than the 2024 Announcement 7 Reporting and 2026 Announcement 7 Reporting as set out in Clauses 3.1(c) and 5, no third party and regulatory consents or notifications, including the approval of or notification to any Government Authority, Tax Authority, court, arbitral tribunal, regulatory body or administrative body, are or will be required or desirable for the transfer of the Sale Shares or any interest in or part thereof to the Purchaser.
2.Group Companies
2.1The corporate particulars of the Group Companies as set out in Schedule 1 are true, accurate and not misleading.
2.2Each Group Company is duly incorporated, validly existing and in good standing under the laws of its jurisdiction of incorporation. Each Group Company has the right, power and authority to conduct its business as conducted as at the date of this Agreement.
2.3Save for the Subsidiaries, no Group Company has any other subsidiary or any interest in or has agreed to acquire any interest in, any other person.
2.4There is no Encumbrance, and there is no agreement, arrangement or obligation to create or give an Encumbrance, in relation to any of the shares or equity interests of each Group Company, and no person has claimed to be entitled to an Encumbrance in relation to any of those shares or equity interests.
2.5Each of the Group Companies is operating and has always operated its business in all respects in accordance with its memorandum and articles of association or equivalent constitutional document at the relevant time.
2.6Each register, minute book and other book which each of the Group Companies is required by Law to keep has been properly kept and contains a true, accurate and not misleading record of the matters which it is required by applicable Laws to record in all material respects.
2.7Except as disclosed in the Disclosure Letter, all returns, particulars, resolutions and other documents required to be delivered or registered by the Group Companies to or with any Government Authority have been properly prepared and delivered in all material respects.
2.8Except as disclosed in the Disclosure Letter, no Group Company has given a power of attorney or other authority by which a person may enter into an agreement, arrangement or obligation on such Group Company's behalf (other than an authority for a director, other officer or employee to enter into an agreement in the usual course of that person's duties).
2.9Each allotted and issued share in the capital of each Group Company has been properly allotted and issued and is fully paid or credited as fully paid. Each shareholder of the Group Companies established in the PRC has fully, validly and punctually paid-up its capital contribution of the registered capital of the relevant Group Company in accordance with the articles of association of the relevant Group Company and applicable Laws in the PRC and such registered capital has been verified as being fully, validly and punctually paid-up by a duly qualified accountant registered in the PRC.
3.Information
3.1All information contained in the Transaction Documents (including any annex to the Disclosure Letter), the Due Diligence Responses and in the Data Room is true, accurate and not misleading in all material respects, and there is no matter which renders any such information untrue, inaccurate or misleading in any material respect.
3.2To the Vendor's knowledge, all information about the Sale Shares and the Group which might be material for disclosure to a purchaser of the Sale Shares has been Disclosed to the Purchaser.
4.Effect of Sale
4.1Neither the execution nor the performance of this Agreement and/or each of the Transaction Documents will result in any Group Company losing the benefit of an asset, grant, subsidy, right or privilege which it enjoys on the date of this Agreement.
4.2Neither the execution nor the performance of this Agreement and/or each of the Transaction Documents will (a) conflict with, result in a breach of, give rise to an event of default under, require the consent of a person under, enable a person to terminate, or relieve a person from an obligation under any agreement or arrangement to which any Group Company is a party or any legal or administrative requirement by which any Group Company is bound, (b) result in the breach of, or create on behalf of any person the right to terminate or modify, any Group Company's rights in any Group Intellectual Property or any Third Party Intellectual Property, or (c) cause the revocation, suspension, cancellation, variation or non-renewal or any Permit.
5.Audited Accounts, Management Accounts and Accounting Records
5.1The Audited Accounts have been prepared and audited with all due care and attention and on a basis consistent with applicable Law, policies, standards, principles and practices generally accepted in the relevant jurisdictions.
5.2The Audited Accounts show a true and fair view of (i) the assets, liabilities (actual, contingent or otherwise), financial position and state of affair of the Company and the Group Companies (as the case may be) as at the Accounts Date, and (ii) the profits and losses of the Company and the Group Companies for the financial year ended on the Accounts Date.
5.3The Audited Accounts fully disclose and provide adequately for all bad and doubtful debts, all liabilities (actual, contingent or otherwise) and all financial commitments existing at the Accounts Date.
5.4The Management Accounts have been properly prepared with all due care and attention and on a basis consistent with applicable Law, policies, standards, principles and practices generally accepted in the relevant jurisdictions.
5.5The Management Accounts fairly present and do not misstate the assets and liabilities of each of the Group Companies as at the Last Management Accounts Date and Management Accounts Date (as applicable) and the profits and losses of each of the Group Companies for the period concerned.
5.6The Management Accounts fully disclose and provide adequately for all bad and doubtful debts, all liabilities (actual, contingent or otherwise) and all financial commitments existing at the Last Management Accounts Date and the Management Accounts Date (as applicable).
5.7The Audited Accounts and Management Accounts are each not affected by any unusual, exceptional or nonrecurring items.
5.8As at the Last Management Accounts Date and the Management Accounts Date, no material liability, obligation or commitment of any nature whatsoever (asserted or unasserted, known or unknown, absolute or contingent, accrued or unaccrued, matured or unmatured or otherwise) existed in relation to any Group Company which was not required to, or permitted not to, be included in the Management Accounts in accordance with applicable accounting policies.
5.9Each Group Company's accounting records are up-to-date, in its possession or under its control and are properly completed in accordance with applicable Laws, policies, standards, principles and practices generally accepted in the relevant jurisdictions.
6.VIE Documents
6.1True and complete copies of the VIE Documents have been provided or made available to the Purchaser on or before the date of this Agreement, which are set out in folder 8 of the Data Room.
6.2(i) Each party to the VIE Documents and (ii) (in the case of the VIE Restructuring Documents) any Group Company and [***] (as applicable) has the right, power and authority, and has taken all action necessary, to enter into and perform its/his respective obligations under the VIE Documents and the VIE Restructuring Documents (as applicable). Each party's obligations under the VIE Documents and the VIE Restructuring Documents (as applicable) are valid, legal and binding, enforceable in accordance with their terms.
6.3(i) Each party to the VIE Documents and (ii) (in the case of the VIE Restructuring Documents) any Group Company and [***] (as applicable) is not in breach of any of the provisions of the
relevant VIE Document. There has not been any dispute relating to the performance of any VIE Document or VIE Restructuring Documents. No fact or circumstance exists which might give rise to a dispute relating to the performance of any VIE Document or VIE Restructuring Document or invalidate or give rise to a ground for termination of any VIE Document or VIE Restructuring Document.
6.4Except as disclosed in the Disclosure Letter, all filings, registrations or recordings required to perfect the pledge over the equity interests in the Shenzhen VIE Entity granted by [***] and [***], each in favour of the Shenzhen WFOE, have been made pursuant to applicable Law.
6.5The Company possesses, through the relevant VIE Documents and the VIE Restructuring Documents (as applicable), the power to control and direct or cause the direction of the operation and activities of the relevant VIE Entity. The Company is entitled, through the relevant VIE Documents and the VIE Restructuring Documents (as applicable), to consolidate the financial results of the relevant VIE Entity in its financial statements.
6.6There is not, and has not been, any investigation or enquiry by any Government Authority in any jurisdiction with respect to any VIE Document or any VIE Restructuring Documents (as applicable).
7.Tax
7.1Each Group Company is and has at all times been subject to or assessed on Taxes in its place of incorporation only. Except as disclosed in the Disclosure Letter, each Group Company is not liable to pay and has at no time incurred any liability to Tax chargeable under the Laws of any jurisdiction other than its place of incorporation, or has otherwise paid all Tax which it has become liable to pay and is not liable to pay a penalty, surcharge, fine or interest in connection with Tax.
7.2Each Group Company has made appropriate deductions and withholdings in respect, or on account, of any Tax from any payments made by it, which it is obliged to make under applicable Laws and has accounted to the Tax Authority for all amounts so deducted or withheld.
7.3Each Group Company has received all necessary approvals with respect to Taxes, including the application of preferential Tax provisions, exemptions, and benefits, and such approvals are in full force and effect under the Laws of the jurisdictions where each Group Company operates.
7.4Except as disclosed in the Disclosure Letter, each Group Company has within applicable time limits made all such returns, provided all such information and maintained all such records in relation to Tax as are required to be made or provided or maintained by it as required by applicable Law and none of such returns is disputed by a Tax Authority concerned.
7.5No Group Company is, in relation to its business, involved in a dispute with any Tax Authority. No Tax Authority has stated that the Tax Authority has investigated or indicated that it intends to investigate the Tax affairs of any Group Company.
7.6Each Group Company has timely withheld, or collected from each payment made to each of its employees, the full amount of all Taxes required to be withheld or collected therefrom, and has timely paid the same to the competent Tax Authority in accordance with applicable Laws.
7.7The Audited Accounts reserve or provide for, in accordance with applicable Law, policies, standards, principles and practices generally accepted in the relevant jurisdictions:
7.7.1all Taxes liable to be assessed on the Company and each Group Company, or for which they are or may become accountable, since the date of incorporation of the Company or the relevant Group Company (as the case may be) (whether or not the Company or the relevant Group Company has or may have a right of reimbursement against another person); and
7.7.2all contingent or deferred liabilities to Tax since the date of incorporation of the Company or the relevant Group Company (as the case may be).
7.8No Group Company has participated in any transaction, scheme or arrangement of which the sole or main purpose or effect is the avoidance or evasion of a liability to Tax or which could be
reasonably re-characterised or treated as unenforceable or ineffective for Tax purposes for such reasons.
8.Related Party Transactions
8.1No Group Company is or has been a party to or otherwise involved in any contract, transaction, agreement or arrangement (including any contract, transaction, agreement or arrangement with any shareholder of the Company or any of his or its Affiliates) which is not entered into by way of a bargain at arm's length, or any transaction, agreement or arrangement under which it has been or is or may be required to make any payment for any goods, services or facilities provided to it which is in excess of the market value of such goods, services or facilities or under which it has been, or is or may be required to provide goods, services or facilities for a consideration which is less than the market value of such goods, services or facilities and/or in consequence of which it is or will be liable to Tax in respect of an amount deemed for Tax purposes to be income or gains of the Group Company but not actually income or gains of the Group Company.
9.Financial Obligations
9.1Except as disclosed in the Management Accounts, (a) the Group Companies do not have outstanding and have not agreed to create or incur loan capital, borrowings or indebtedness in the nature of borrowings; and (b) none of the Group Companies is party to and is not liable under any guarantee, indemnity or other agreement to secure or incur a financial or other obligation with respect to another person's obligation.
9.2No Group Company is liable to repay an investment or other grant or subsidy made to it by any person or Government Authority.
9.3No fact or circumstance (including the execution and performance of this Agreement) exists which might entitle any person or Government Authority to require repayment of, or refuse an application by any Group Company for, the whole or part of a grant or subsidy.
10.Properties
10.1None of the Group Companies owns any premises, buildings, land or other real property right.
10.2The list of Leased Properties set out in Schedule 4 is, in all respects, a true, correct and complete list of all of the Leased Properties of the Group.
10.3With respect to each contract pertaining to any Leased Property (each a "Lease Agreement"):
10.3.1such Lease Agreement is valid, binding, enforceable and in full force and effect;
10.3.2all rents have been paid in full and there are no rent arrears;
10.3.3no Group Company nor any other party to the Lease Agreement is in breach or default under such Lease Agreement, and no event has occurred or circumstance exists which, with the delivery of notice, the passage of time or both, would constitute a breach or default thereunder or would result in the premature termination of such Lease Agreement; and
10.3.4no member of the Group is currently subleasing, licensing or otherwise granting any person the right to use or occupy such Leased Property or any portion thereof.
10.4Each Leased Property is presently used for the purpose of the business permitted under applicable Laws and the relevant Lease Agreement, and all necessary permissions and consents in respect of use for the purpose of the Leased Property and for any previous use and/or any development at each Leased Property have been obtained.
11.Intellectual Property
11.1Each Group Company, as applicable, owns, has a license or sublicense to, or otherwise possesses or can obtain on commercially reasonable terms, legally enforceable rights to all Intellectual Property (including all Intellectual Property developed by employees or consultants of the Group) necessary for, held for use in or used in, or otherwise is or likely to be material to the business of the Group Companies as currently conducted or as currently proposed to be conducted.
11.2There are no material defects in any of the patents or patent applications included in the Group Intellectual Property or, to the Vendor's knowledge, the Third Party Intellectual Property.
11.3Except as disclosed in the Disclosure Letter, there are no Group Intellectual Property in respect of which any Group Company is registered as the owner or is the applicant for registration.
11.4Except as disclosed in the Disclosure Letter, no Group Company has granted, nor is obliged to grant, any licence or assignment in respect of any Intellectual Property to any third party.
11.5To the Vendor's knowledge, each of the relevant counterparties to all written contracts, agreements or arrangements to which a Group Company is a party owns, has a license, sublicense or permit to, or otherwise possesses or can obtain on commercially reasonable terms, legally enforceable rights to all Intellectual Property necessary for the performance of its respective obligations under any such written contracts, agreements or arrangements.
11.6No party to an agreement relating to the use by any Group Company of any Intellectual Property owned by another person is, or has at any time been, in breach of the agreement and no circumstances exist which would give rise to any breach of any such agreement or to any such agreement being terminated, suspended, varied or revoked without the relevant Group Company's consent (other than termination without cause upon notice in accordance with the terms of the agreement).
11.7Schedule 6 contain true, accurate and not misleading details of all licences of Intellectual Property granted to a Group Company ("IP Licences In") respectively, including, as Part A, all patents and patent applications specified in Schedule 1 of the IP Licence Agreement, covering the Licensed Field of Use in any jurisdiction within the Territory, and, as Part B, all grants, divisions, continuations, continuations-in-part, reissues, re-examinations, extensions and foreign counterparts of all patents and patent applications in Part A within the Territory covering the Licensed Field of Use as of the date of this Agreement. Copies of all IP Licences In (including any variations, amendments, novations, extensions and other documents affecting the terms of any IP Licences In) have been provided to the Purchaser and the parties to such licences have not agreed to vary any of the terms of any such licences. To the best knowledge of the Vendor, all patent applications or patents listed in Part B of Schedule 6 that claim priority from the patents or patent applications mentioned in Part A of Schedule 6, including any continuations, divisionals, continuations-in-part, and foreign equivalents, are and will remain fully supported by, and within the scope of, the disclosure of the relevant priority document as filed, so that the claims of those patents and applications are entitled to the priority date of the priority document under applicable law.
11.8Other than the Group Intellectual Property and the Third Party Intellectual Property, there is no other Intellectual Property necessary for, held for use in or used in, or otherwise material to the conduct of the business of the Group Companies as currently conducted or as currently proposed to be conducted.
11.9Except for the Group Companies, no other person has any right, title or interest in and to the Group Intellectual Property, and no person who was involved in, or who contributed to, the creation or development of any Group Intellectual Property has performed services for the government, a university, a college, or other educational institution or research centre in a manner that would affect any Group Company's rights in such Intellectual Property.
11.10Neither the conduct of the business of the Group as currently conducted or proposed to be conducted nor any product or service marketed or sold (or proposed to be marketed or sold) by the Group violates or will violate any license, or infringes or misappropriates, or will infringe or misappropriate any Intellectual Property rights of any other person. None of the Group Companies has received any claims or communications alleging that any Group Company has violated, infringed or misappropriated, or by conducting its business as currently conducted or proposed to be conducted, would violate, infringe or misappropriate any Intellectual Property rights of any other person.
11.11To the Vendor's knowledge, there is not, and has not at any time been, an infringement, misappropriation, misuse, violation or other unauthorised use of any of the Group Intellectual Property or any of the Third Party Intellectual Property.
11.12The Group Companies have taken all necessary measures (including to impose appropriate confidentiality obligations on parties given access to any confidential information of any Group Company, and to have systematic safeguards) to maintain in confidence all confidential information comprising a part of the Group Intellectual Property or Third Party Intellectual Property, and there has been no unauthorized or illegal use of, access to, or disclosure of any such confidential information.
11.13Each of the IP Licences In is a valid, binding and enforceable agreement of the parties thereto, the performance of which does not violate any applicable Law, and is in full force and effect, and the terms thereof have been complied with by the relevant Group Companies and by all the other parties thereto. No IP Licences In is currently being, or has at any time been, breached by any Group Company or, to the best of the Vendor's knowledge, information and belief, by any other party thereto.
11.14No grounds for rescission, avoidance or repudiation of any IP Licences In, and no notices of violation, default, termination or intention to terminate (whether or not such notice is in writing so long as it is in accordance with the terms of such contracts) have been received in respect of any IP Licences In. No Group Company is in breach of any IP Licences In.
11.15The Group Companies have paid all license and royalty fees required to be paid under the Third Party Intellectual Property and/or pursuant to any IP Licences In.
11.16All renewal and maintenance fees and Taxes due and payable in respect of any Third Party Intellectual Property have been paid in full. Each other action required to maintain and protect any Third Party Intellectual Property has been taken.
11.17No employee, officer, director (whether executive or non-executive), consultant, agent or other representative of any Group Company or any individual who has formerly held one of these positions has any right to claim ownership or any other right in respect of Intellectual Property used in the business of the Group and all necessary assignments, settlements and compromise agreements relating to the transfer or assignment of any of the Group Intellectual Property by any of the aforementioned individuals have been executed and are valid and enforceable. None of the Intellectual Property used by the Group in its businesses has been obtained or is being used by any Group Company in violation of any contractual obligation that is binding on the Group or any of its respective officers, directors or employees.
12.Assets
12.1All assets included in the Management Accounts or acquired by a Group Company since the Management Accounts Date, other than any assets disposed of or realised in the ordinary course of business is (a) legally and beneficially owned solely by the relevant Group Company; (b) where capable of possession, in the possession or under the control of the relevant Group Company, and (c) free from Encumbrances.
12.2Each of the Group Companies owns or has the right to use such asset used in and is necessary for the effective operation of its business.
12.3Each material asset of the Group, which is normal to have maintained by independent or specialist contractors, is in good condition and working order and has been regularly maintained to a good technical standard and in accordance with safety regulations required to be observed in relation to it.
13.Agreements
13.1Except as set out in folder 4. Financial Documents > 001. Provision of information > 01. FDD > 19. RP of the Data Room and as disclosed in the Disclosure Letter, no Group Company is party to or subject to any contract, agreement, arrangement or transaction which:
13.1.1is with a shareholder of the Company or any of his or its Affiliates;
13.1.2is with any entity within the Take2 Group;
13.1.3is not in the ordinary course of its business;
13.1.4restricts such Group Company's freedom to operate the whole or part of its business or to use or exploit any of its assets;
13.1.5cannot be complied with on time or without undue or unusual expenditure of money or effort by such Group Company; or
13.1.6is of a long-term nature that is incapable of termination in accordance with its terms by such Group Company on six (6) months' notice or less.
13.2All of the written contracts, agreements, arrangements or obligations to which a Group Company is a party and which are material to the business of the Group (a "Material Contract") are valid, binding and enforceable on the parties thereto in accordance with their respective terms. No fact or circumstance exists which might invalidate or give rise to a ground for termination, avoidance or repudiation of any Material Contract.
13.3Neither a Group Company (on the one hand) nor any party with whom a Group Company (on the other hand) has entered into a Material Contract is in breach of any Material Contract, and no fact or circumstance exists which might give rise to a breach of this type.
13.4No Group Company is, or has agreed to become, a member of any joint venture, consortium, partnership or other unincorporated association.
14.Employees
14.1The details of all employees to the Group as set out in Schedule 5 (the "Employees") are true, correct and complete.
14.2No Group Company is party to, nor bound by, any collective bargaining contract or work rules or practices with any labour union, labour organisation or works council.
14.3Each Group Company (a) has properly classified all of its workers as independent contractors or employees, or (b) has properly classified, recorded and documented the employment of all of its employees under applicable Law.
14.4To the Vendor's knowledge, no employee or director of any Group Company is in violation of any term of any employment agreement, non-disclosure agreement, statutory non-disclosure obligation, noncompetition agreement, restrictive covenant or other obligation to a former employer of any such employee relating to (a) the right of any such employee to be employed by that Group Company or (b) the use of trade secrets or proprietary information.
14.5Each employment contract with the directors, officers and Employees is legally binding and enforceable in accordance with its respective terms.
14.6No Group Company has:
14.6.1incurred any liability for breach or termination of an employment contract;
14.6.2provided or agreed to provide a gratuitous payment or benefit to a current or former director, officer or employee or to any of their dependants; or
14.6.3made or agreed to make a payment or provided or agreed to provide a benefit to a present or former director, other officer or employee of such Group Company or to any of their dependants in connection with the actual or proposed termination or suspension of employment or variation of an employment contract.
14.7Other than the Director Service Agreements, no Group Company has nor is proposing to introduce a share incentive, share option, profit sharing, bonus or other incentive scheme for any of its directors, other officers or Employees.
14.8None of the Group Companies has given notice of termination to or received notice of resignation from any of its directors (other than [***]) or employees.
15.Pension Schemes
15.1No Group Company has any material liability to make any payment to any pension, contribution, social security, allowance or similar scheme or any insurance arrangement held in relation thereto which is due, but remains unpaid.
15.2Each Group Company is in compliance with the provisions of the pension, contribution, social security, allowance and/or similar schemes in respect of such Group Company and Law.
16.Permits
16.1Each Group Company has obtained all Permits and has complied with the terms and conditions of, each Permit.
16.2Each Permit is in force, unimpeachable and unconditional or subject only to a condition that has been satisfied.
16.3Each action required for the renewal or extension of each Permit has been taken.
17.Insolvency
17.1No Group Company is insolvent under the laws of its jurisdiction of incorporation or unable to pay its debts as they fall due, nor has any Group Company commenced negotiations with one (1) or more of its creditors with a view to rescheduling or restructuring any of its indebtedness.
17.2There are no proceedings in relation to any compromise or arrangement with creditors or any winding-up, bankruptcy or other insolvency proceedings concerning any Group Company.
17.3No steps have been taken to enforce any security over any assets of any Group Company.
17.4No Group Company has received a notice to the effect that an administrator has been or will be appointed in relation to the relevant Group Company. No notice has been given or filed with the court of an intention to appoint an administrator and no petition or application has been presented or order made for the appointment of an administrator in respect of any Group Company.
17.5No receiver or administrative receiver has been appointed, nor any notice given to the relevant Group Company of an intention to appoint, any such person, over the whole or part of any Group Company's business or assets.
17.6No Group Company has proposed or agreed to a composition, compromise, assignment or arrangement with any of its creditors.
17.7No distress, execution, attachment, sequestration or other process has been levied on an asset of a Group Company which remains undischarged.
17.8No action is being taken by a court to dissolve a Group Company and/or strike it off the relevant register.
17.9No Group Company is, in any jurisdiction, subject to or threatened by any other procedures or steps which are analogous to those set out above.
18.Litigation
18.1No Group Company nor, to the Vendor's knowledge, any of the directors, key officers and employees of the Group is, or has since the date of its incorporation been, involved, whether as claimant or defendant or other party, in any claim, Action, suit or litigation, in relation to the business of any Group Company.
18.2No written notification has been received by a Group Company in the two (2) years ending on the date of this Agreement, to the effect that a claim, Action, suit or litigation is pending or threatened
by or against a Group Company or any of the directors, officers and employees of the Group and no matter exists which will or would reasonably be likely to give rise to a claim, Action, suit or litigation involving a Group Company or any of the directors, officers and employees of the Group.
19.Compliance with Law
19.1Each of the Group Companies and its directors, officers and employees has conducted the business of the Group and dealt with the Group's assets in accordance with all applicable Laws in all material respects.
19.2There is not, and has not been, any investigation, enquiry or disciplinary proceeding instigated by or involving any Government Authority concerning or involving a Group Company or any of its directors, officers and employees, and none is pending or threatened. To the Vendor's knowledge, no fact or circumstance exists which might give rise to an investigation, enquiry or proceedings of that type.
20.Anti-Bribery, Anti-Money Laundering and Sanctions
20.1None of the Group Companies, the Vendor nor any of the Vendor's agents, employees, and other persons acting on behalf of the Vendor, nor, to the Vendor's knowledge, any of the Group Company's respective agents, employees, and other persons acting on its behalf, have taken any action that would cause the Group to be in violation of Anti-Bribery laws or Anti-Money Laundering Laws.
20.2No Group Company is subject to any investigation by, or request for information from, law enforcement officials regarding a violation or potential violation of Anti-Bribery laws or Anti-Money Laundering Laws by a Group Company.
20.3To the Vendor's knowledge, there are no facts or circumstances that would lead a reasonable person to believe that there is a likelihood that any of the officers, directors, employees or agents of the Group Companies has made any payment in violation of any Anti-Bribery laws or Anti-Money Laundering Laws on behalf of or for the benefit of a Group Company.
20.4None of the Group Companies, the Vendor or any of their respective directors, officers, agents, employees or affiliates, is a Sanctioned Person or engaged in any activity that is reasonably likely to cause them to be designated as Sanctioned Persons by any Sanctions Authority.
20.5Each Group Company has (a) complied with, and has not engaged in any activity or conduct which has or would violate, applicable Sanctions since the date of its incorporation; (b) not engaged in a transaction or dealing, directly or knowingly indirectly, with or involving a Sanctioned Jurisdiction or Sanctioned Person; (c) implemented and maintained policies and procedures designed to promote and achieve compliance with applicable Sanctions; and (d) not been, nor any of their assets are or have been, subject to any claim, action, suit, proceeding, investigation, injunction or enforcement action (criminal, regulatory, administrative or civil) concerning or relating to any actual or alleged breach of applicable Sanctions, and to the Vendor's knowledge, no such action, claim, suit, proceeding, investigation, injunction or enforcement action is pending, threatened or contemplated.
21.Data Protection
21.1Each Group Company has at all times complied with any and all applicable Laws regulating data protection, data security, privacy, or the recording, monitoring or interception of communications or electronic communications (the "Data Protection Laws").
21.2No Group Company has received:
21.2.1any enforcement, information or equivalent notice under the Data Protection Laws;
21.2.2any written communication from any data protection or privacy authority with competent authority over such Group Company's data processing activities, indicating that it is investigating an allegation that such Group Company is in breach of the Data Protection Laws or that it proposes to take, or is considering taking, enforcement action under the Data Protection Laws; or
21.2.3any written communication from any person complaining about such Group Company's use of information about that person or alleging any breach of any of the Data Protection Laws.
21.3No Group Company has compensated or been required to compensate any individual under the Data Protection Laws, no claim for such compensation is outstanding and there is no reason to believe that any circumstances exist which might lead to any claim for compensation being made.
21.4There is no personal data breach or other incident involving unauthorised loss, disclosure, corruption, impairment or other prejudice to the security of any personal data or confidential or proprietary information in the possession or under the control of any Group Company that was, is or is likely to be required under the Data Protection Laws to be notified to any data protection or privacy authority and/or to any other external third parties.
22.Insurance
22.1The Disclosure Letter contains a true, accurate and not misleading list of each current insurance and indemnity policy in respect of which a Group Company has an interest (together the "Insurance Policies").
22.2Each of the Insurance Policies is valid and enforceable and is not void or voidable.
22.3The Company has not done anything or omitted to do anything which might (a) make any of the Insurance Policies void or voidable; or (b) prejudice the ability to effect insurance on the same or better terms in the future.
22.4No claim is outstanding under any of the Insurance Policies.
22.5All premiums which are due under the Insurance Policies have been paid.
23.Side agreements and insider agreements
23.1Other than the 2024 SPA, Existing Shareholders' Agreement, the share sale agreement dated 25 June 2023 between Dennis Lo, Allen Chan and the Vendor and the share subscription agreement dated 25 June 2023 between the Company and the Vendor, there is no letter, agreement or arrangement between or among the Vendor, any of the Founders and/or any of the Group Companies in relation to the voting or governance, issuance or disposition of any securities (whether debt or equity) in or sharing of profits of any Group Company or any other matter contemplated by this Agreement or any of the Transaction Documents, and no such letter, agreement or arrangement is contemplated to be entered into between or among the Vendor, any of the Founders and/or any of the Group Companies.
23.2Except as set out in the Disclosure Letter, to the Vendor's knowledge, there is no agreement or arrangement (legally enforceable or not) to which a Group Company is a party and in which:
23.2.1the Vendor;
23.2.2an Affiliate of the Vendor; or
23.2.3a director or officer or former director or officer of the Vendor or an Affiliate of the Vendor,
is interested in any way.
24.Brokerage or commissions
25.No person is entitled to receive a finder's fee, brokerage or commission from the Company in connection with this Agreement.
Schedule 3
LIMITATION OF THE VENDOR'S LIABILITY
LIMITATION OF THE VENDOR'S LIABILITY
1.Limitation on Quantum
1.1The Vendor shall not be liable in respect of a Warranty Claim unless:
1.1.1the amount recoverable from the Vendor in respect of that Warranty Claim exceeds $[***]. For this purpose, Warranty Claims arising out of the same, related or similar subject matter shall be aggregated and form a single Warranty Claim; and
1.1.2the aggregate amount recoverable from the Vendor in respect of that Warranty Claim and any other Warranty Claims exceeds $[***], in which case the Vendor shall be liable for such aggregated amount and not just the excess.
1.2The Vendor's total liability in respect of all Warranty Claims (other than Fundamental Warranty Claims) is limited to an amount equal to [***]% of the aggregate of the Purchase Price, provided that the Vendor's total liability in respect of all Warranty Claims (including the Fundamental Warranty Claims) shall be limited to an amount equal to [***]% of the Purchase Price.
2.Time Limits for bringing claims
2.1The Vendor is not liable for a Fundamental Warranty Claim or a Tax Warranty Claim unless the Purchaser has given the Vendor a Notice of such claim on or before the date that is seven (7) years from the Completion Date.
2.2The Vendor is not liable for a Warranty Claim (other than a Fundamental Warranty Claim and a Tax Warranty Claim) unless the Purchaser has given the Vendor a Notice of such claim on or before the date that is three (3) years from the Completion Date.
3.No Double Recovery
4.The Purchaser may not recover from the Vendor under this Agreement more than once in respect of the same loss suffered.
5.Mitigation
6.Nothing in this Schedule 3 restricts or limits the Purchaser's common law duty to mitigate any loss suffered by it as a result of a breach of a Vendor's Warranty.
7.General
8.Nothing in this Schedule 3 shall have the effect of limiting or restricting any liability of the Vendor in respect of a Warranty Claim arising as a result of any fraud, wilful misconduct or wilful concealment.
Schedule 4
LIST OF LEASED PROPERTIES
LIST OF LEASED PROPERTIES
[***] | ||
Schedule 5
LIST OF EMPLOYEES
LIST OF EMPLOYEES
[***]
Schedule 6
LIST OF INTELLECTUAL PROPERTY
LIST OF INTELLECTUAL PROPERTY
Part A – IP Licences In
All patents and patent applications specified in Schedule 1 of IP Licence Agreement (reproduced below), covering the Licensed Field of Use in any jurisdiction within the Territory, including any grants, divisions, continuations, continuations-in-part, reissues, re-examinations and extensions of all such patents claiming priority therefrom, including any foreign counterparts thereof within the Territory covering the Licensed Field of Use, as well as renewals thereof.
[***]
Part B – Current status of IP Licences In
Status of all patents and patent applications specified in Schedule 1 of IP Licence Agreement, covering the Licensed Field of Use in any jurisdiction within the Territory, including any grants, divisions, continuations, continuations-in-part, reissues, re-examinations and extensions of all such patents claiming priority therefrom, including any foreign counterparts thereof within the Territory covering the Licensed Field of Use, as well as renewals thereof, as of the date of this Agreement:
[***]
IN WITNESS WHEREOF, each of the Parties hereto has caused this Agreement to be executed by their duly authorised representatives on the date first set forth above.
VENDOR | ||||||||
For and on behalf of PRENETICS GLOBAL LIMITED By: ___________________________ Name: Title: | ||||||||
[Signature page to SPA]
PURCHASER | ||
For and on behalf of IMAGE FRAME INVESTMENT (HK) LIMITED By: ___________________________ Name: Title: | ||
[Signature page to SPA]
Exhibit 4.30
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
July 5, 2024
Sheng Wu Yeung
Co-Founder, Chairperson & CEO
Prenetics Global Limited
Unit 703-706
K11 Atelier King’s Road 728 King’s Road
Quarry Bay, Hong Kong
Dear Mr. Yeung:
This letter will confirm our understanding that Prenetics Global Limited (the “Company” or “PRE”) has engaged Roth Capital Partners, LLC (“Roth”) in connection with the matters described below, subject to the terms and conditions set forth in this letter agreement (this “Agreement”).
Section 1. Engagement as Capital Markets Advisor. For a period of twelve (12) months (the “Engagement Period”), Roth will provide PRE with capital markets advisory services (the “Capital Markets Advisory”). These services include, but are not limited to, providing advice to PRE relating to:
1.Developing a short- and long-term capital market strategy;
2.Analyzing PRE, its business, industry, competition, and anticipated capital requirements as they relate to the valuation of PRE;
3.Strategic development of institutional investor relationships;
4.Attendance at relevant sector Roth Conference and/or Roth Annual Conference participation;
5.Meeting with Roth Sales personnel, at Roth offices or remotely, as determined by Roth; and
6.Such other activities as may be mutually agreed to from time to time between PRE and Roth.
Additionally, rules to which we are subject prohibit our giving research coverage for compensation (unless disclosed). Accordingly, we in no way promise or represent to PRE that our research department will cover PRE.
The Parties acknowledge and agree that in consideration of Roth’s agreement to
provide the Services set forth in this Agreement, Roth shall be paid an Advisory Fee, as set forth in Exhibit I.
Section 2. Termination. Either PRE, on the one hand, or Roth on the other hand, may terminate this Agreement upon thirty (30) days written notice to the other, provided, however, that the provisions of Section 2 through Section 9 (including Addendum attached hereto) shall survive termination of this Agreement.
Section 3. Other Services. Provided this Agreement is not terminated by Roth, during the period of twelve (12) months following the termination of this agreement, if the Company decides to (i) use a placement agent other than Roth to pursue any private placement not contemplated hereby, or (ii) pursue any public offering of equity, equity-linked, debt securities or an at-the-market offering (each a “Financing”), then the Company shall offer Roth the right to act as placement agent or lead underwriter and book runner, as applicable, for such Financing, in each case under a separate agreement containing terms and conditions customary for Roth and mutually agreed upon by the Company and Roth.
Section 4. Indemnification. PRE agrees to indemnify Roth and its affiliates as set forth in Addendum A attached hereto.
Section 5. No Limitations. Nothing in this Agreement shall be construed to limit the ability of Roth or its affiliates to (a) trade in the company’s securities or publish research on the Company or any other company, subject to applicable law, or (b) pursue or engage in investment banking, financial advisory or other business relationships with entities that may be engaged in or contemplate engaging in, or acquiring or disposing of, businesses that are similar to or competitive with the business of the Company.
Section 6. Miscellaneous. This Agreement shall be binding on and inure to the benefit of PRE, the Company, Roth, each Indemnified Person (as defined in Exhibit I attached hereto) and their respective successors and assignees. This Agreement sets forth the entire understanding of the Parties relating to the subject matter hereof and supersedes and cancels any prior communications, understandings, and agreements between the Parties. This Agreement may not be amended or modified except in writing and signed. This Agreement shall be governed by and construed in accordance with the laws of the State of New York, without regard to principles of conflicts of law. If any term, provision, covenant or restriction contained in this Agreement, including Addendum A hereto, is held by a court of competent jurisdiction or other authority to be invalid, void, unenforceable or against its regulatory policy, the remainder of the terms, provisions, covenants and restrictions contained in this Agreement shall remain in full force and effect and shall in no way be affected, impaired or invalidated. Roth is an independent contractor, and any duties of Roth arising out of its engagement hereunder shall be owed solely to PRE and the Company or, where applicable, to the Board of Directors or a special committee thereof. Any advice provided to PRE or the Board of Directors (or a special committee thereof) is solely for the benefit of PRE and may not be used, reproduced, disseminated, quoted or referred to, without Roth’s prior written consent.
Section 7. Arbitration. Any dispute, claim or controversy arising out of or relating to this Agreement or the breach, termination, enforcement, interpretation or validity thereof, including the determination of the scope or applicability of this agreement to arbitrate, shall be determined by arbitration in New York, NY, before one arbitrator. The arbitration shall be administered by JAMS pursuant to its Comprehensive Arbitration Rules & Procedures. Judgment on the Award may be entered in any court having jurisdiction. This clause shall not preclude parties from seeking provisional remedies in aid of arbitration from a court of appropriate jurisdiction.
Section 8. Allocation of Fees and Costs. The arbitrator may, in the Award, allocate all or part of the costs of the arbitration, including the fees of the arbitrator and the reasonable attorneys’ fees of the prevailing party.
[REMAINDER OF PAGE LEFT BLANK INTENTIONALLY]
If the foregoing terms meet with your approval, please indicate your acceptance by signing and returning the attached copy of this letter to us.
Very truly yours,
ROTH CAPITAL PARTNERS, LLC
By:
Name: [***]
Title: Executive Chairman
Accepted as of the date first above written:
PRENETICS GLOBAL LIMITED
By:
Name: Sheng Wu Yeung
Title: Co-Founder, Chairperson & CEO
EXHIBIT I
Advisory Fee: | The Company shall pay Roth a “Capital Markets Advisory Fee”. The Capital Markets Advisor Fee shall be paid equivalent to [***] shares of PRE Common Stock (each common share, a “Common Share”) in accordance with the terms and conditions of Prenetics' 2022 Share Incentive Plan. Twelve (12) months after the execution of this Agreement, Prenetics shall issue fully vested restricted shares units, which would be fully vested and readily convertible into [***] Common Shares to Roth at no exercise cost, in accordance with the terms and conditions of Prenetics’ 2022 Share Incentive Plan. In the event that Prenetics is unable to issue fully vested restricted share units to Roth Capital Partners, LLC as described above, Prenetics will issue [***] shares of common stock with registration rights as described below. The shares of Common Shares issued or transferred to Roth in satisfaction of Roth’s Fee shall be free and clear of all liens, encumbrances and other restrictions including without limitation, any restrictions that may arise due to applicable securities laws; provided, however, that if the Company is unable at the time of such issuance or transfer to provide for the issuance or transfer of the Stock without any transfer restrictions that may arise due to applicable securities laws, then the Company shall enter into an agreement with Roth on the date of the consummation of the Agreement, in form and substance reasonably acceptable to Roth, to provide Roth with customary registration rights with respect to the Stock (the “Registration Rights”). Such Registration Rights shall include an agreement that the Company shall as soon as practicable, but no later than fifteen (15) days after the issuance of Roth’s shares, file with the U.S. Securities and Exchange Commission a registration statement for the registration, under the Securities Act of 1933, as amended, of the resale of the Stock (the “Registration Statement”). The Company shall use its commercially reasonable efforts to cause the same to become effective and to maintain the effectiveness of such Registration Statement, and a current prospectus relating thereto, until the earlier of (i) the date on which Roth ceases to hold any of such Stock or (ii) such Stock may be freely tradable under United States federal securities laws by anyone who is not an affiliate (as such term is defined in Rule 144 under the Securities Act of 1933, as amended (or any successor rule)) of the Company. | ||||
ADDENDUM A
Indemnification Provisions
Prenetics Global Limited (collectively, “PRE”) agree to indemnify and hold harmless Roth and its affiliates (as defined in Rule 405 under the Securities Act of 1933, as amended) and their respective directors, officers, employees, agents and controlling persons (Roth and each such person being an “Indemnified Party”) from and against all losses, claims, damages and liabilities (or actions, including shareholder actions, in respect thereof), joint or several, to which such Indemnified Party may become subject under any applicable federal or state law, or otherwise, which are related to or result from the performance by Roth of the services contemplated by or the engagement of Roth pursuant to this Agreement and will promptly reimburse any Indemnified Party for all reasonable expenses (including reasonable counsel fees and expenses) as they are incurred in connection with the investigation of, preparation for or defense arising from any threatened or pending claim, whether or not such Indemnified Party is a party and whether or not such claim, action or proceeding is initiated or brought by PRE. PRE will not be liable to any Indemnified Party under the foregoing indemnification and reimbursement provisions (i) for any settlement by an Indemnified Party effected without its prior written consent (not to be unreasonably withheld); or (ii) to the extent that any loss, claim, damage or liability is found in a final, non-appealable judgment by a court of competent jurisdiction to have resulted primarily from the Indemnified Party willful misconduct or gross negligence. PRE also agrees that no Indemnified Party shall have any liability (whether direct or indirect, in contract or tort or otherwise) to PRE or its security holders or creditors related to or arising out of the engagement of Roth pursuant to, or the performance by Roth of the services contemplated by, this Agreement except to the extent that any loss, claim, damage or liability is found in a final, non-appealable judgment by a court of competent jurisdiction to have resulted primarily from Roth’s willful misconduct or gross negligence.
Promptly after receipt by an Indemnified Party of notice of any intention or threat to commence an action, suit or proceeding or notice of the commencement of any action, suit or proceeding, such Indemnified Party will, if a claim in respect thereof is to be made against PRE pursuant hereto, promptly notify PRE in writing of the same. Any failure or delay by an Indemnified Party to give the notice referred to in this paragraph shall not affect such Indemnified Party’s right to be indemnified hereunder, except to the extent that such failure or delay causes actual material harm to PRE, or materially prejudices its ability to defend such action, suit or proceeding on behalf of such Indemnified Party. In case any such action is brought against any Indemnified Party and such Indemnified Party notifies PRE of the commencement thereof, PRE may elect to assume the defense thereof, with counsel reasonably satisfactory to such Indemnified Party, and an Indemnified Party may employ counsel to participate in the defense of any such action provided, that the employment of such counsel shall be at the Indemnified Party’s own expense, unless (i) the employment of such counsel has been authorized in writing by PRE, (ii) the Indemnified Party has reasonably concluded (based upon advice of counsel to the Indemnified Party) that there are legal defenses available to the Indemnification Party that are not available to PRE, or that there exists a conflict or potential conflict of interest (based upon advice of counsel to the Indemnified Party) between the Indemnified Party and PRE that makes it impossible or inadvisable for counsel to PRE to conduct the defense of both parties (in which case PRE will not have the right to direct the defense of such action on behalf of the Indemnified Party), or (iii) PRE
has not in fact employed counsel reasonably satisfactory to the Indemnified Party to assume the defense of such action within a reasonable time after receiving notice of the action, suit or proceeding, in each of which cases the reasonable fees, disbursements and other charges of such counsel will be at the expense of PRE; provided, further, that in no event shall PRE be required to pay fees and expenses for more than one firm of attorneys (and local counsel) representing Indemnified Parties.
If the indemnification provided for in this Agreement is for any reason held unenforceable by an Indemnified Party, PRE agrees to contribute to the losses, claims, damages and liabilities for which such indemnification is held unenforceable (i) in such proportion as is appropriate to reflect the relative benefits to PRE, on the one hand, and Roth on the other hand or, (ii) if (but only if) the allocation provided for in clause (i) is for any reason unenforceable, in such proportion as is appropriate to reflect not only the relative benefits referred to in clause (i) but also the relative fault of PRE, on the one hand and Roth, on the other hand, as well as any other relevant equitable considerations. PRE agrees that for the purposes of this paragraph the relative benefits to PRE and Roth shall be deemed to be in the same proportion that the total value received or contemplated to be received by PRE bear to the fees paid or to be paid to Roth under this Agreement. Notwithstanding the foregoing, PRE expressly agrees that Roth shall not be required to contribute any amount in excess of the amount by which fees paid to Roth hereunder (excluding reimbursable expenses), exceeds the amount of any damages which Roth has otherwise been required to pay.
PRE agrees that without Roth’s prior written consent, which shall not be unreasonably withheld, it will not settle, compromise or consent to the entry of any judgment in any pending or threatened claim, action or proceeding in respect of which indemnification could be sought under the indemnification provisions of this Agreement (whether or not Roth or any other Indemnified Party is an actual or potential party to such claim, action or proceeding), unless such settlement, compromise or consent includes an unconditional release of each Indemnified Party from all liability arising out of such claim, action or proceeding.
In the event that an Indemnified Party is requested or required to appear as a witness in any action brought by or on behalf of or against PRE in which such Indemnified Party is not named as a defendant, PRE agrees to promptly reimburse Roth on a monthly basis for all expenses incurred by it in connection with such Indemnified Party’s appearing and preparing to appear as such a witness, including, without limitation, the reasonable fees and disbursements of its legal counsel.
If multiple claims are brought with respect to at least one of which indemnification is permitted under applicable law and provided for under this Agreement, PRE agrees that any judgment or arbitration award shall be conclusively deemed to be based on claims as to which indemnification is permitted and provided for, except to the extent the judgment or arbitration award expressly states that it, or any portion thereof, is based solely on a claim as to which indemnification is not available.
Exhibit 4.31
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
INVESTOR RELATIONS CONSULTING AGREEMENT
THIS CONSULTING AGREEMENT (“Agreement”) is made this 25th day of April 2024 by and between Prenetics Global Limited (hereinafter referred to as the “Company” or “PRE”) and MZHCI, LLC, a MZ Group Company (hereinafter referred collectively as the “Consultant” or “MZHCI”).
EXPLANATORY STATEMENT
The Consultant affirms that it has successfully demonstrated financial and public relations consulting expertise, and possesses valuable knowledge, and experience in the areas of business finance and corporate investor/public relations. The Company believes that the Consultant’s knowledge, expertise, and experience would benefit the Company, and the Company desires to retain the Consultant to perform consulting services for the Company under this Agreement.
NOW, THEREFORE, in consideration of their mutual Agreements and covenants contained herein, and for other valuable consideration, the receipt and sufficiency of which is hereby acknowledged, and in further consideration of the affixation by the parties of their respective signatures below, the parties agree as follows:
Consulting Services
1.1 MZHCI agrees that commencing on the 25th day of April 2024 (the “Effective Date”), the Consultant will reasonably be available during regular business hours to advise, counsel, and inform designated officers and employees of the Company as it relates to financial markets and exchanges, competitors, business acquisitions and other aspects of or concerning the Company’s business about which MZHCI has knowledge or expertise.
1.2 MZHCI shall render the services set out in section 1 below (“Services”) to the Company as an independent contractor, and not as an employee. All Services rendered by MZHCI on behalf of the Company shall be performed to the best of MZHCI’s ability, in accordance with all applicable laws and regulations, including SEC and Nasdaq regulations, rules, and guidance, in concert with the overall business plan of the Company, and the goals and objectives of the Company’s management and Board of Directors, including articulating PRE’s investment story and highlights; building and maintaining relationships with supporters of the stock, including institutional investors and sell-side analysts; increasing the Company participation in investment conferences; achieving a fair market value for the Company’s stock; and significantly increasing the Company’s exposure in the financial market.
I. Scope of Services, Programs and Deliverables
MZHCI will develop, implement, and maintain an ongoing stock market support system for PRE with the general objective of expanding awareness in PRE among stockbrokers, analysts, small-cap portfolio/fund managers, market makers, and the appropriate financial & trade publications.
INVESTOR RELATIONS
A. Complete IR Audit (including full review of the investor and shareholder database, IR website and all
public documentation)
B. Understand the financials and all operating metrics of PRE in detail, facilitating interactions with new
and current investors.
C. Senior Account Manager and single point of contact for all investors and streamlining of all
communication and IR functionality.
D. Develop and Update FAQ
E. Create 2-Page Corporate Profile
F. PowerPoint Presentation Updates
G. Review and provide suggestions for IR website
H. Quarterly Conference Call Script and Preparation
I. Press Release Input and Dissemination
J. Facilitate incoming and outgoing investor/shareholder calls. Screen all parties before allowing
communication with management.
K. Shareholder Database Management
L. Roadshow Management Coaching
M. Roadshows with Detailed Follow-Up
N. Targeted Sell-Side Research and Financial Media Introductions
O. Retail Investors Engagement Programs
P. Investor Conference Invites
INVESTMENT AWARENESS AND OUTREACH
A.Consultant will make introductions to investors worldwide utilizing a proprietary, robust database:
i. Equity Brokers
ii. Analysts (both generalists and industry specialists)
iii. Portfolio Managers/Institutions
iv. High Net Worth Investors & Family Offices
v. Private Equities/ Hedge Funds / Long Funds
vi. Financial Publication
FINANCIAL MEDIA RELATIONS
Targeted media relations offer an important segment to the corporate story. MZHCI will target media opportunities that highlight PRE’s strategy, growth objectives, board of directors, developments and milestones related to its business. Services include:
A. Targeted media programs B. Strategic counsel
C. Release drafts and media targets
D. Q&A to support significant corporate developments
E. Feedback after interviews
Business & Financial Media – MZHCI will identify the optimal news, corporate, and industry trends that will provide angles in the business/financial media and then actively pursue those opportunities with the appropriate reporter(s).
PUBLIC MARKET INSIGHT
MZHCI will counsel and educate the Company’s senior management on the life cycle of the financial markets and most importantly how the Company is impacted directly and indirectly by different variables. The team at MZHCI leverages its collective expertise on all aspects of strategic financial, corporate, and crisis communications gained through representing over 200 public companies. MZHCI will help the Company set and manage expectations while relaying valuation metrics, perceptions, and methodologies
utilized by investment professionals. This consulting aspect of MZHCI’s business is extremely valuable for management to optimize key opportunities and to avoid pitfalls.
As part of its ongoing commitment and partnership with the Company, MZHCI will educate the Company’s senior management on the importance of establishing conservative expectations and how various corporate actions may be perceived and impact the public market.
ONGOING
A. Respond to all investor requests and calls in a timely manner to facilitate the distribution of corporate information. Focus on educating shareholders, with the premise that an informed investor will become a longer-term investor.
B. Continually update the database to ensure that all press releases are e-mailed to all interested professionals. This includes the input of notes to keep track of all investor correspondence and reminders investors prior to earnings conference calls.
C. Provide consulting services to PRE management on the public markets.
D. Provide progress reports to senior management and evaluate achievements with a summary of activities and a detailed report as requested.
Many of the above items will occur simultaneously but certain items will have chronological priority over others. As PRE grows, MZHCI will recommend changes to the agenda that complement its growth. As the Company continues to execute its strategic plan by winning new customers and expanding its base of business, MZHCI will target an expanded universe of institutional investors. At each stage of growth, the appropriate approach to the market will be incorporated into the agenda for optimal results.
Assuming that management’s efforts are leading ultimately to success and great profitability, the end results of this financial communication and awareness campaign should be:
A. An increase in the number of financial professionals (including brokers, institutions, and analysts) and individual investors well educated and knowledgeable about PRE: including senior management, the company’s products, and its current financial condition & growth opportunities.
B. An increase in the number of articles printed in both trade and financial publications.
B. An increase in the number of articles printed in both trade and financial publications.
C. An increase in the liquidity of the common stock.
D. An increase in PRE’s market capitalization coupled with a broader, more diverse shareholder base.
E. Suitable and better access to the capital markets, which will facilitate future acquisitions and
working capital needs.
II. Term
This agreement shall be effective upon the Effective Date and shall remain effective for a period of nine (9) months (“Initial Term”), unless extended or terminated as follows:
A. Either party may terminate this Agreement at the end of the Initial Term, by providing written notice to the other party no less than 30 days prior to the end of the Initial Term;
B. If the Agreement is not terminated following the Initial Term, it shall automatically renew every one (1) month thereafter unless either party provides written notice of termination to the other party at least 30 days prior to the then current term.
Upon termination of this Agreement for any reason:
A. each party agrees to promptly return to the other party all property, including but not limited to documents, equipment, and materials, as well as all copies of confidential information belonging to the other party, in its possession or under its control;
B. the Company shall pay MZHCI for all Services performed up to the date of termination, provided that such Services were performed in accordance with the terms and conditions of this Agreement. All payments due shall be made within thirty (30) days of the date of termination.
C. MZHCI shall immediately cease to represent the Company in its engagement with investors or any other third parties and shall refrain from making any statements, written or oral, that negatively impacts the Company’s reputation, business relationships, or services; and
D. MZHCI shall provide a final report to the Company detailing the work completed, the status of ongoing projects, and any other information pertinent to the orderly cessation and transition of services. In addition, MZHCI shall provide, at the Company’s request, reasonable transition services and cooperation to the Company, including any information it has gathered pursuant to this Agreement.
III. Compensation
| Cash | $[***] USD per month. The first month's payment is due immediately and all subsequent payments are due within ten (10) days of receipt of a valid invoice from MZHCI. At each twelve (12) anniversary of this Agreement, a 5% COLA (Cost of Living Adjustment) increase will be applied to the cash fee. | ||||
| Equities | The Company will award MZHCI [***] PRE restricted stock units (“RSUs”) by June 30, 2024. The RSUs shall be awarded in accordance with the terms of the RSU award agreement and Company’s 2022 Share Incentive Plan, and will vest immediately. Notwithstanding the terms of the Company’s Share Incentive Plan or the RSU award agreement, the RSUs shall not be forfeitable by the Company. The RSUs shall be deemed fully paid for and earned upon issuance. The Company shall post on EDGAR a Form 6-K, or other acceptable SEC filing, reporting on its entry into an Agreement with MZHCI within three (3) months. Required language: On April 25, 2024, Prenetics Global Limited entered into an investor relations consulting agreement with MZHCI, LLC. | ||||
| Expense Reimbursement | Prior to incurring any expenses, MZHCI shall obtain the Company’s written approval for the approximate amount to be incurred. Only expenses that would ordinarily be incurred by the Company will be billed back on a monthly basis. Applicable reimbursements would include creation, printing, and postage for investor packages, fees for news wire services. Any packages requiring additional photocopying/ printing will be billed back to the Company at cost (with no mark-up). Any extraordinary items, such as broker lunch presentations, air travel, hotel, ground transportation or media campaign PRE, etc. shall be paid by the Company. | ||||
IV. Prior Restriction
MZHCI represents to the Company that it is not subject to, or bound by, any Agreement which sets forth or contains any provision, the existence or enforcement of which would in any way restrict or hinder MZHCI from performing the services on behalf of the Company that MZHCI is herein agreeing to perform.
V. Assignment
This Agreement is personal to MZHCI and may not be assigned in any way by MZHCI without the prior written consent of the Company. Subject to the foregoing, the rights and obligations under this Agreement shall inure to the benefit of, and shall be binding upon, the heirs, legatees, successors, and permitted assigns of MZHCI and upon the successors and assigns of the Company.
VI. Confidentiality
Except as required by law or court order, MZHCI will keep confidential any trade secrets or confidential or proprietary information of the Company which are now known to MZHCI or which hereinafter may become known to MZHCI and MZHCI shall not at any time directly or indirectly disclose or permit to be disclosed any such information to any person, firm, or corporation or other entity, or use the same in any way other than in connection with the business of the Company and in any case only with prior written permission of PRE. For purposes of this Agreement, “trade secrets or confidential or proprietary information” includes information unique to or about the Company including but not limited to its business and that is not known or generally available to the public. It is understood and agreed that MZHCI’s obligations pursuant to this section survive the termination of this Agreement.
VII. Default
1.Except for a claim or controversy arising under Section VII of this Agreement, any claim or controversy arising under any of the provisions of this Agreement shall, at the election of either party hereto, be determined by arbitration in California in accordance with the rules of the American Arbitration Association. The decision of the Arbitrator shall be binding and conclusive upon the parties. Each party shall pay its own costs and expenses in any such arbitration. The prevailing party shall be entitled to reimbursement of all fees incurred, including attorney, filing, travel, and anything associated with the arbitration.
2.In the event MZHCI commits any material breach of any provision of this Agreement, as determined by the Company in good faith, the Company may, by injunctive action, compel MZHCI to comply with, or restrain MZHCI from violating, such provision, and, in addition, and not in the alternative, the Company shall be entitled to declare MZHCI in default hereunder and to immediately terminate this Agreement and any further payments hereunder.
3.Since MZHCI must at all times rely upon the accuracy and completeness of information supplied to it by the Company’s officers, directors, agents, and employees, the Company agrees to indemnify, hold harmless, and defend MZHCI, its officers, agents, and employees at the Company’s expense, against any proceeding or suit which may arise out of and/or be due to any material misrepresentation in such information supplied by the Company to MZHCI (or any material omission by the Company that caused such supplied information to be materially misleading).
4.MZHCI agrees to indemnify, hold harmless and defend the Company, its officers, directors, employees, and agents from and against any and all claims, actions, proceedings, losses, liabilities, costs and expenses (including without limitation reasonable attorney’s fees) incurred by any of them in connection with, as a result of, and or due to any actions or inactions or misstatements by MZHCI, its officers, agents, or employees regarding or on behalf of the Company whether as a result of rendering services under this Agreement or otherwise.
VIII. Severability and Reformation
If any provision of this Agreement is held to be illegal, invalid, or unenforceable under present or future law, invalid or unenforceable provisions were never a part hereof, and the remaining provisions shall remain in full force and shall not be affected by the illegal, invalid, or unenforceable provision, or by its severance; but in any
such event this Agreement shall be construed to give effect to the severed provision to the extent legally permissible.
IX. Notices
Any notices required by this Agreement shall (i) be made in writing and delivered to the party to whom it is addressed by hand delivery, by certified mail, return receipt requested, with adequate postage prepaid, or by courier delivery service (including major overnight delivery companies such as Federal Express and UPS), (ii) be deemed given when received, and (iii) in the case of the Company, be mailed to its principal office Prenetics Global Limited, 703-706, K11 Atelier, 728 King’s Road, Quarry Bay, Hong Kong and in the case of MZHCI, be mailed to [***].
X. Miscellaneous
1.This Agreement may not be amended, except by a written instrument signed and delivered by each of the parties hereto.
2.This Agreement constitutes the entire understanding between the parties hereto with respect to the subject matter hereof, and all other agreements relating to the subject matter hereof are hereby superseded.
3.This Agreement shall be governed by, and construed in accordance with, the laws of the State of California, USA
In Witness Whereof, the parties have executed this Consulting Agreement as of the day and year first above written.
AGREED:
MZHCI, LLC Prenetics Global Limited
By: ________________________________ By: ________________________________
[***] Danny Yeung, Co-Founder & CEO
Date: _______________________________ Date: _______________________________
Exhibit 4.32
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
INVESTOR RELATIONS CONSULTING AGREEMENT
THIS CONSULTING AGREEMENT (“Agreement”) is made as of the date last signed below by and between Prenetics Global Limited (hereinafter referred to as the “Company” or “PRE”) and MZHCI, LLC, a MZ Group Company (hereinafter referred collectively as the “Consultant” or “MZHCI”).
EXPLANATORY STATEMENT
The Consultant has investor relations consulting expertise, and possesses valuable knowledge, and experience in the areas of business finance and corporate investor relations. The Company desires to retain the Consultant to perform consulting services for the Company under this Agreement.
NOW, THEREFORE, in consideration of their mutual Agreements and covenants contained herein, and for other valuable consideration, the receipt and sufficiency of which is hereby acknowledged, and in further consideration of the affixation by the parties of their respective signatures below, the parties agree as follows:
Consulting Services
1.1 MZHCI agrees that commencing on the 12th day of August 2025 (the “Effective Date”), the Consultant will reasonably be available during regular business hours to perform the services set forth in Section I below (the "Services"). The Company shall be responsible for the accuracy and completeness of all data and information provided to MZHCI for purposes of its performance of Services under this Agreement.
1.2 MZHCI shall render services to the Company as an independent contractor, and not as an employee, an agent, distributor or representative of the other. Neither party shall act or present itself, directly or indirectly, as an agent of the other or in any manner assume or create any obligation on behalf of, or in the name of, the other. The Services shall be performed in a manner consistent with generally accepted industry standards, in a professional and workmanlike manner. PRE shall cooperate with Consultant in its performance of Services under this Agreement, including without limitation providing Consultant with reasonable facilities and timely access to data, information and personnel of PRE.
I. Scope of Services, Programs and Deliverables
The following Services shall be performed by MZHCI during the Term:
MZHCI will work to develop a stock market support system for PRE with the general objective of expanding awareness in PRE among the retail investment community, as well as stockbrokers, analysts, small-cap portfolio/fund managers.
INVESTOR RELATIONS
A.Strategic Positioning & Messaging – Reframe the company’s identity from a ‘holding’ company (the old PRE) to a Bitcoin-forward operating model, clearly articulating the rationale & long-term value proposition
B.Define relevant KPIs for IR program - Investor base evolution (crypto-native participation vs. legacy holders), Valuation premium/discount to NAV or BTC-adjusted book value, Sell-side coverage initiation, Engagement metrics (web traffic, X followers, call attendance), Retail inflow trends (from broker reporting or trading data)
C.Complete IR Audit (including full review of the investor and shareholder database, IR website and all public documentation)
D.Develop and Update FAQ – Coaching and answers to the most common questions
E.Develop detailed retail investor engagement strategy
F.PowerPoint Presentation Revamp & Updates
G.Review and provide suggestions for IR website
H.Quarterly Conference Call Script and Preparation
I.Press Release Input and Dissemination
J.Facilitate incoming and outgoing investor/shareholder calls. Screen all parties before allowing communication with management.
K.Shareholder Database Management
L.Roadshow Management Coaching
M.Roadshows with Detailed Follow-Up
N.Targeted Sell-Side Research Introductions
O.Investor Conference Invites
INVESTMENT AWARENESS AND OUTREACH
A.Consultant will use good faith efforts to make introductions to investors worldwide utilizing a proprietary, robust database:
i.Analysts (both generalists and industry specialists)
ii.Portfolio Managers/Institutions
iii.High Net Worth Investors & Family Offices
iv.Financial Publications
FINANCIAL MEDIA
MZHCI will work to coordinate opportunities that position PRE in financial news channels (both paid & earned) to build and enhance the company’s image among stakeholders. MZ will focus on identifying and engaging appropriate media to encourage interest in PRE’s news, achievements and milestones related to its corporate goals. Services include:
A.Media training
B.Identification and outreach to financial media across print, online, broadcast, podcast
C.Identification and financial media positioning around industry developments
D.Creation of financial media facing content
E.FAQ message development to support significant corporate initiatives
INVESTOR RELATIONS WEBSITE DESIGN AND HOSTING
MZ will design, develop and host a company-specific investor relations website for PRE upon request.
Website features include:
A.Hosting infrastructure with Amazon AWS data center hosting in the United States.
B.Fully responsive layout structure with customization of logo, content, map, images and color, following the Brand Identity Manual
C.Top-tier security with https
D.Initial SEO preparation and URL customization
E.GDPR Compliance – MZ is committed to the General Data Protection Regulation and protecting Personally Identifiable Information. In additional to the GDPR compliance, our policies and procedures follow the rigorous controls set out in ISO 27001:2013
F.Automated Feed for regulatory filings and press releases, as needed
G.Easy to manage CMS for full autonomy
H.24x7 support team to provide our clients with an outstanding customer experience
II. Term
This agreement becomes effective upon the Effective Date and shall remain effective for a period of six (6) months (the "Initial Term"), unless terminated earlier as set forth below. Upon expiration of the Initial Term, this agreement may be renewed on a mutually agreed basis with written communication (email or mail). MZHCI may terminate this Agreement and cease services if the Company fails to timely pay the Compensation set forth in Section III below. On any such termination, the Company shall still be obligated to pay the Compensation set forth in Section III through the remainder of the then current Term.
III. Compensation
| Cash | $[***] USD per month MZHCI (at its sole discretion) can elect to receive up to 50% of the monthly cash fee in restricted PRE common stock, which will be due within ten (10) days of each month of service if elected. The contract may be renewed by mutual written agreement (mail or email) at any time during the engagement. The first month's payment is due immediately and all subsequent payments are due within fifteen (15) days of each month of service. In the event MZHCI does not receive payment by the 15th day of each month of service, the Company shall accrue a late charge on the balance outstanding at the lesser of (a) 1 1/2% per month or (b) the highest rate allowed by law, in each case compounded monthly to the extent allowed by law. At each annual anniversary of the Effective Date of this Agreement, a 5% COLA (Cost of Living Adjustment) increase will be applied to the cash fee. | ||||
| Performance-Based Bonuses | The Company will award MZHCI [***] restricted stock units (“RSUs”) by August 31, 2025. The RSUs shall be awarded in accordance with the terms of the RSU award agreement and Company’s 2022 Share Incentive Plan, and will vest immediately. Notwithstanding the terms of the Company’s Share Incentive Plan or the RSU award agreement, the RSUs shall not be forfeitable by the Company. The RSUs shall be deemed fully paid for and earned upon issuance. The Company will issue performance-based bonuses (“Performance Bonuses”) to MZHCI upon completion of the following milestones during the term of the engagement: •30-Day Average Daily Dollar Volume (“Dollar Volume”): oIf during the engagement, the Dollar Volume of PRE equals or exceeds $2,000,000, the Company will pay a bonus of $[***] USD to MZHCI. oIf during the engagement, the Dollar Volume of PRE equals or exceeds $3,000,000, the Company will pay a bonus of $[***] USD to MZHCI. oIf during the engagement, the Dollar Volume of PRE equals or exceeds $5,000,000, the Company will pay a bonus of $[***] USD to MZHCI. oIf during the engagement, the Dollar Volume of PRE equals or exceeds $10,000,000, the Company will pay a bonus of $[***] USD to MZHCI. •New Institutional Shareholders: For each introduction that MZHCI makes resulting in an investor purchasing 2.0% or greater of the total shares outstanding of PRE (“New Investors”), the Company will pay a Performance Bonus of $[***] USD to MZHCI. The New Investors shall be verified via public filings, such as 13F, 13G or 13D filings, or brokerage statements for any non-filing shareholders. The Performance Bonuses for New Investors shall not exceed $[***] in any given twelve (12) month term. •Sell-Side Analyst Coverage: For each introduction that MZHCI makes which results in non-paid, institutional research coverage for PRE (“Research Coverage”), the Company will pay a bonus of $[***] USD to MZHCI. The Performance Bonuses for Research Coverage shall not exceed $[***] in any given twelve (12) month term. For the avoidance of doubt, Dollar Volume shall be calculated by the average daily trading volume over the last thirty (30) days, multiplied by the closing price on the day of the calculation. At the sole discretion of the Company, the Performance Bonuses may be paid in either cash ($ USD), or restricted PRE common stock, which will be calculated using the closing price of common stock on the day each respective milestone is achieved. In all instances, the Performance Bonuses are due to MZHCI within twenty (20) days of achieving each milestone. If Shares are issued for performance-based bonuses once achieved, the Shares shall be deemed earned, fully paid, and non-forfeitable. | ||||
| Expense Reimbursement | Only expenses that would ordinarily be incurred by the Company will be billed back on a monthly basis. Applicable reimbursements would include creation, printing, and postage for investor packages, fees for news wire services. Any packages requiring additional photocopying/ printing will be billed back to the Company at cost (with no mark-up). Any extraordinary items, such as broker lunch presentations, air travel, hotel, ground transportation or media campaigns, etc. shall be paid by the Company. | ||||
IV. Prior Restriction
MZHCI represents to the Company that it is not subject to, or bound by, any Agreement which sets forth or contains any provision, the existence or enforcement of which would in any way restrict or hinder MZHCI from performing the services on behalf of the Company that MZHCI is herein agreeing to perform.
V. Assignment
This Agreement may not be assigned by the Company without the prior written consent of MZHCI. This Agreement may be assigned by MZHCI in the event of a sale of substantially all of the assets of MZHCI. Subject to the foregoing, the rights and obligations under this Agreement shall inure to the benefit of, and shall be binding upon, the heirs, legatees, successors, and permitted assigns.
VI. Confidentiality
Except as required by law or court order, MZHCI will keep confidential any trade secrets or confidential or proprietary information of the Company which hereinafter may become known to MZHCI and MZHCI shall not at any time directly or indirectly disclose or permit to be disclosed any such information to any person, firm, or corporation or other entity, or use the same in any way other than in connection with the business of the Company and in any case only with prior written permission of PRE. For purposes of this Agreement, “trade secrets or confidential or proprietary information” includes information unique to or about the Company including but not limited to its business and that is not known or generally available to the public. It is understood and agreed that MZHCI’s obligations pursuant to this section survive the termination of this Agreement.
VII. Default
1.Except as set forth below in this subparagraph 1, any claim or controversy arising under any of the provisions of this Agreement shall be determined by arbitration in Orange County, California in accordance with the Commercial Arbitration Rules (or International Dispute Resolution Procedures if Company is domiciled outside of the United States) of the American Arbitration Association. The decision of the Arbitrator shall be binding and conclusive upon the parties. Notwithstanding the foregoing in this subparagraph 1, MZHCI may, in its sole discretion, forego arbitration and commence litigation in the event that the claim(s) to be asserted relate to or involve: (a) amounts owing by Company to MZHCI under this Agreement; or (b) the need for equitable or injunctive relief. Each party shall pay its own costs and expenses in any such arbitration. In all cases, this Agreement shall be governed by, and construed in accordance with, the laws of the State of California, USA, without regard to conflict of law principles. Sole and exclusive venue for any arbitration or litigation arising under this Agreement shall lie, as appropriate, in the AAA office or the state and federal courts located in Orange County, California and the Company hereby consents to such jurisdiction. The prevailing party shall be entitled to reimbursement of all fees and costs incurred, including attorney, filing, travel, and anything reasonably associated with the arbitration or litigation.
2.MZHCI warrants that the Services provided by it shall be performed in a professional manner. EXCEPT AS SET FORTH IN THE PRECEDING SENTENCE, MZHCI MAKES NO REPRESENTATION OR WARRANTY, EXPRESS, IMPLIED OR STATUTORY. In the event of a breach by MZHCI of this Agreement, the Company’s sole remedy against MZHCI shall be to re-perform the Services in accordance with the warranty. Notwithstanding the foregoing, in no event shall the liability of MZHCI, whether by reason of breach of contract, tort (including without limitation negligence), statute or otherwise exceed the amount of fees paid by the Company under this Agreement. Further, in no event shall MZHCI have any liability for loss of profits, loss of business, indirect, incidental, consequential, special, punitive, indirect or exemplary damages, even if the Company has been advised of the possibility of such damages. In furtherance and
not in limitation of the foregoing, MZHCI shall not be liable in respect of any decisions made by the Company as a result of the Services.
3.Since MZHCI must at all times rely upon the accuracy and completeness of information supplied to it by the Company’s officers, directors, agents, and employees, the Company agrees to indemnify, reimburse, hold harmless and defend MZHCI, its directors, officers, agents, and employees at the Company’s expense, against any and all losses, claims, damages, obligations, penalties, judgments, awards, liabilities, costs, expenses and disbursements (and any and all actions, suits, proceedings and investigations in respect thereof and any and all legal and other costs, expenses and disbursements in giving testimony or furnishing documents in response to a subpoena or otherwise), including, without limitation, the costs, expenses and disbursements, as and when incurred, of investigating, preparing or defending any such action, suit, proceeding or investigation, directly or indirectly, caused by, relating to, based upon, arising out of or in connection with this Agreement, including which may arise out of and/or be due to any material misrepresentation in such information supplied by the Company to MZHCI (or any material omission by the Company that caused such supplied information to be materially misleading).
4.MZHCI agrees to indemnify, hold harmless and defend the Company, its officers, directors, employees, and agents from and against any and all claims, actions, proceedings, losses, liabilities, costs and expenses (including without limitation reasonable attorney’s fees) incurred by any of them in connection with, as a result of, and or due to any actions or inactions or misstatements by MZHCI, its officers, agents, or employees regarding or on behalf of the Company whether as a result of the gross negligence or intentional misconduct in rendering services under this Agreement or otherwise.
VIII. Severability and Reformation
If any provision of this Agreement is held to be illegal, invalid, or unenforceable under present or future law, invalid or unenforceable provisions were never a part hereof, and the remaining provisions shall remain in full force and shall not be affected by the illegal, invalid, or unenforceable provision, or by its severance; but in any such event this Agreement shall be construed to give effect to the severed provision to the extent legally permissible.
IX. Notices
Any notices required by this Agreement shall (i) be made in writing and delivered to the party to whom it is addressed by hand delivery, by certified mail, return receipt requested, with adequate postage prepaid, or by courier delivery service (including major overnight delivery companies such as Federal Express and UPS), (ii) be deemed given when received, and (iii) in the case of the Company, be mailed to its principal office at Prenetics Global Limited, Unit 703-706, K11 Atelier, 728 King’s Road, Quarry Bay, Hong Kong; and in the case of MZHCI, be mailed to [***].
X. Miscellaneous
1.This Agreement may not be amended, except by a written instrument signed and delivered by each of the parties hereto.
2.This Agreement constitutes the entire understanding between the parties hereto with respect to the subject matter hereof, and all other agreements relating to the subject matter hereof are hereby superseded.
3.This Agreement may be executed in any number of counterparts, each of which shall constitute an original. Signatures delivered via facsimile or electronic transmission shall be binding upon the party so delivering such a signature, regardless of whether originally executed signatures are subsequently delivered.
In Witness Whereof, the parties have executed this Consulting Agreement as of the day and year last written below.
AGREED:
MZHCI, LLC Prenetics Global Limited
By: ________________________________ By: ________________________________
[***] Danny Yeung, Co-Founder & CEO
Date: _______________________________ Date: _______________________________
Exhibit 4.33
Certain information have been excluded from this exhibit because it is both (i) not material and (ii) the type that the registrant treats as private or confidential. The excluded information is indicated by brackets containing asterisks ("[***]"). The registrant will furnish supplementally a copy of any excluded information or schedule to the Securities and Exchange Commission upon request.
AMENDMENT AGREEMENT
This Amendment Agreement (the “Amendment”) is made and entered into as of September 5, 2025 by and between Prenetics Global Limited (the “Company”) and MZHCI, LLC (the
"Consultant").
RECITALS
WHEREAS, the Company and the Consultant are parties to that certain Investor Relations Consulting Agreement, dated August 12, 2025 (the “Consulting Agreement”);
WHEREAS, the Company and the Consultant wish to amend the Performance-Based Bonuses clause under the Section III “Compensation” of the Consulting Agreement as set forth herein;
NOW, THEREFORE, in consideration of the mutual covenants and agreements set forth herein, the parties agree as follows:
1. Amendment to the Performance Based Bonuses Clause
The first paragraph of the Performance-Based Bonuses clause under Section III
“Compensation” in the Consulting Agreement is hereby amended and restated in its entirety
as follows:
“The Company will issue [***] restricted Ordinary Class A shares (“Shares”) to the Consultant as soon as practicable. The Company shall issue the Shares to the Consultant in accordance with the terms set out in the share subscription agreement executed by the parties. The Shares would be subject to a six month of transfer restriction immediately upon issuance of the Shares. Such transfer restriction shall expire by a lapse of time.
The other paragraphs of the Performance-Based Bonuses clause under Section III
“Compensation” in the Consulting Agreement shall remain.
2. Effect of Amendment
Except as expressly amended hereby, all other terms and provisions of the Consulting Agreement shall remain in full force and effect.
3. Governing Law
This Amendment shall be governed by and construed in accordance with the laws of the State of California, USA.
4. Entire Agreement
This Amendment, together with the Consulting Agreement, constitutes the entire understanding between the parties with respect to the subject matter hereof.
IN WITNESS WHEREOF, the parties hereto have executed this Amendment Agreement as
of the date first written above.
Prenetics Global Limited
By: ___________________________
Name: Danny Yeung
Title: Chief Executive Officer
MZHCI, LLC
By: ___________________________
Name: [***]
Title: Chief Executive Officer
Exhibit 8.1
Subsidiaries of Prenetics Global Limited
The following list sets forth the subsidiaries of Prenetics Global Limited:
Subsidiaries | Jurisdiction of Incorporation | |||||||
Prenetics Holding Company Limited | Cayman Islands | |||||||
Prenetics International Limited | British Virgin Islands | |||||||
Prenetics Asia Limited | British Virgin Islands | |||||||
Prenetics Limited | Hong Kong | |||||||
Qianhai Prenetics Technology (Shenzhen) Co., Ltd. 前海皮樂迪科技(深圳) 有限公司 | the PRC | |||||||
Zhuhai Prenetics Technology Co., Ltd. 珠海皮樂迪科技有限公司 | the PRC | |||||||
Prenetics (Malaysia) Sdn. Bhd. | Malaysia | |||||||
Prenetics Pte. Ltd. | Singapore | |||||||
Prenetics Innovation Labs Private Limited | India | |||||||
IM8 UK Holdings Limited (formerly known as "Prenetics EMEA Limited") | England & Wales | |||||||
Oxsed Limited | England & Wales | |||||||
IM8 Global Limited | Cayman Islands | |||||||
Europa Global Holdings Inc. | State of Delaware, USA | |||||||
Europa Partners Holdings, LLC | State of Delaware, USA | |||||||
Europa Sports Investments, LLC | State of Delaware, USA | |||||||
Europa Sports Partners, LLC | State of Delaware, USA | |||||||
Hubmatrix Partners, LLC | State of Delaware, USA | |||||||
IM8 Group Holding Company | Cayman Islands | |||||||
IM8 (US) LLC | State of Delaware, USA | |||||||
IM8 Limited | Hong Kong | |||||||
IM8 Technology Limited | Hong Kong | |||||||
Exhibit 11.1
PRENETICS GLOBAL LIMITED
STATEMENT OF POLICIES
GOVERNING MATERIAL NON-PUBLIC INFORMATION AND
THE PREVENTION OF INSIDER TRADING
(AS ADOPTED BY THE BOARD OF DIRECTORS OF PRENETICS GLOBAL LIMITED ON May 10, 2022, effective upon May 18, 2022)
This Statement of Policies Governing Material Non-Public Information and the Prevention of Insider Trading (this “Statement”) applies to all directors, officers, employees and consultants of Prenetics Global Limited and its subsidiaries and affiliated entities (collectively, the “Company”).
This Statement consists of four sections: section 1 provides an overview; section 2 sets forth the Company’s policies prohibiting insider trading; section 3 outlines the penalties that apply for insider trading; and section 4 provides further explanations on insider trading.
A Certificate of Compliance is also attached to this Statement. Every director, officer, employee and consultant of the Company must review this Statement, and when requested by the Company, must execute and return the Certificate of Compliance attached hereto to the Compliance Officer for the Company (the “Compliance Officer”) within seven (7) days after receiving the request.
Questions regarding this Statement should be directed to the Compliance Officer by e-mail at compliance@prenetics.com.
1.SUMMARY
Preventing insider trading is necessary to comply with United States securities laws and to preserve the reputation and integrity of the Company, as well as that of all persons affiliated with it. “Insider trading” occurs when any person purchases or sells any securities while in possession of inside information relating to the securities. As explained in Section 3 below, “inside information” is information which is considered to be both “material” and “non-public.”
The Company considers strict compliance with the policies set forth in this Statement (collectively, the “Policy”) to be a matter of utmost importance. Violation of the Policy could cause extreme reputational damage and possible legal liability to you and the Company. Knowing or willful violations of the letter or spirit of the Policy will be grounds for immediate dismissal from the Company. Violation of the Policy might expose the violator to severe criminal penalties, as well as civil liability to any person harmed by the violation. The monetary damages flowing from a violation could be multiple times the profit realized by the violator, not to mention the attorney’s fees of the persons harmed.
This Statement applies to all directors, officers, employees and consultants of the Company and extends to all of such persons’ activities within and outside their duties at the Company.
2.POLICIES PROHIBITING INSIDER TRADING
For purposes of this Statement, the terms “purchase” and “sell” of securities exclude the acceptance of restricted share units (“RSUs”) or other share-based awards granted by the Company and the exercise of options or vesting of RSUs or other share-based awards, if applicable, that does not involve the sale of securities. Among
1
other things, the cashless exercise of options does involve the sale of securities and therefore is subject to the policies set forth below. The Policy does not apply to the exercise of a tax withholding right pursuant to which you elect to have the Company withhold ordinary shares subject to an RSU or other award to satisfy tax withholding requirements.
2.1No Trading
No director, officer, employee or consultant of the Company may purchase or sell any ordinary shares or other securities of the Company or enter into a binding security trading plan in compliance with Rule 10b5-1 under the U.S. Securities Exchange Act of 1934, as amended (a “Trading Plan”) while in possession of material non-public information relating to the Company or its ordinary shares or other securities (the “Material Information”).
In the event that the Material Information possessed by you relates to the ordinary shares or other securities of the Company, the above policy will require waiting for at least forty-eight (48) hours after public disclosure of the Material Information by the Company, which forty-eight (48) hours shall include in all events at least one full Trading Day on the stock exchange where the Company’s ordinary shares are listed and traded (the “Stock Exchange”) following such public disclosure. The term “Trading Day” is defined as a day on which the Stock Exchange is open for trading. Except for public holidays in the United States, the Stock Exchange’s regular trading hours are from 9:30 a.m. to 4:00 p.m., New York City time, Monday through Friday.
In addition, no director, officer, employee or consultant of the Company may purchase or sell any securities of the Company or enter into a Trading Plan, without the prior clearance by the Compliance Officer, during any period designated as a “limited trading period” by the Company, regardless of whether such director, officer, employee or consultant possesses any Material Information.
Furthermore, all transactions in the securities of the Company (including without limitation, acquisitions and dispositions of the ordinary shares, the sale of ordinary shares issued upon exercise of options or vesting of RSUs or other share-based awards and the execution of a Trading Plan, but excluding the acceptance of RSUs or other share-based awards granted by the Company and the exercise of options or vesting of RSUs or other share-based awards that does not involve the sale of securities) by directors, officers and key employees designated by the Company from time to time must be pre-approved by the Compliance Officer.
Please see Section 4 below for an explanation of what constitutes Material Information.
2.2Trading During a Trading Window
Assuming none of the “no trading” restrictions set forth in Section 2.1 above applies, no director, officer, employee or consultant of the Company may purchase or sell any securities of the Company or enter into a Trading Plan other than during a Trading Window.
A “Trading Window” is the period in any fiscal quarter of the Company commencing at the close of business on the second Trading Day following the date of the Company’s public disclosure of its financial results for the prior year or quarter, as applicable, and ending on December 31, March 31, June 30 or September 30, as the case may be.
In other words,
a)beginning on January 1 of each year, no director, officer, employee or consultant of the Company may purchase or sell any securities of the Company or enter into a Trading Plan until the close of business on the second Trading Day following the date of the Company’s public disclosure of its financial results for the fiscal year ended on December 31 of the prior year, and
2
b)beginning on April 1, July 1 and October 1 of each year, respectively, no director, officer, employee or consultant of the Company may purchase or sell any securities of the Company or enter into a Trading Plan until the close of business on the second Trading Day following the date of the Company’s public disclosure of its financial results for the fiscal quarter ended on March 31, June 30 and September 30 of that year, respectively.
If the Company’s public disclosure of its financial results for the prior period occurs on a Trading Day more than four hours before the Stock Exchange closes, then such date of disclosure shall be considered the first Trading Day following such public disclosure.
Please note that trading in any securities of the Company during the Trading Window is not a “safe harbor,” and all directors, officers, employees and consultants of the Company should strictly comply with the Policy.
When in doubt, do not trade! Check with the Compliance Officer first.
Notwithstanding the foregoing, sale of securities of the Company pursuant to an existing Trading Plan which was entered into in accordance with the Policy and in compliance with applicable law is not subject to the restrictions on trading in Sections 2.1 and 2.2.
2.3No Tipping
No director, officer, employee or consultant of the Company may directly or indirectly disclose any Material Information to anyone who trades in securities (“Tipping”).
2.4Confidentiality
No director, officer, employee or consultant of the Company may communicate any Material Information to anyone outside the Company under any circumstances unless approved by the Compliance Officer in advance, or to anyone within the Company other than on a need-to-know basis.
2.5No Comment
No director, officer, employee or consultant of the Company may discuss any internal matters or developments of the Company with anyone outside the Company, except as required for the performance of regular corporate duties. Unless you are expressly authorized to the contrary, if you receive any inquiries about the Company or its securities by the financial press, research analysts or others, or any requests for comments or interviews, you are required to decline comment and direct the inquiry or request to the Head of Investor Relations who is responsible for coordinating and overseeing the release of information of the Company to the investing public, analysts and others in compliance with applicable laws and regulations.
2.6Corrective Action
If you become aware that any potential Material Information has been or may have been inadvertently disclosed, you must notify the Compliance Officer immediately so that the Company can determine whether or not corrective action, such as general disclosure to the public, is warranted.
3.PENALTIES FOR INSIDER TRADING
Penalties for trading on or tipping material non-public information can extend significantly beyond any profits made or losses avoided, both for individuals engaging in the unlawful conduct and their employers. The United States Securities and Exchange Commission and the United States Department of Justice have made the civil
3
and criminal prosecution of insider trading violations a top priority. Enforcement remedies available to the government or private plaintiffs under the U.S. federal securities laws include:
●administrative sanctions;
●sanctions by self-regulatory organizations in the securities industry;
●civil injunctions;
●damage awards to private plaintiffs;
●disgorgement of profits gained by the violator;
●civil fines for the violator of up to three times the amount of profit gained or loss avoided by the violator;
●civil fines for the employer or other controlling person of a violator (i.e., where the violator is an employee or other controlled person) of up to the greater of US$1,000,000 or three times the amount of profit gained or loss avoided by the violator;
●criminal fines for individual violators of up to US$5,000,000 (US$25,000,000 for an entity); and
●jail sentences of up to 20 years.
In addition, insider trading could result in serious sanctions by the Company, including immediate dismissal. Insider trading violations are not limited to violations of the U.S. federal securities laws. Other U.S. federal and state civil or criminal laws, such as the laws prohibiting mail and wire fraud and the Racketeer Influenced and Corrupt Organizations Act (RICO), also may be violated upon the occurrence of insider trading.
4.EXPLANATION OF INSIDER TRADING
4.1What Constitutes Insider Trading?
As noted above, “insider trading” refers to the purchase or sale of a security while in possession of “material” “non-public” information relating to the security. “Securities” include not only stocks, bonds, notes and debentures, but also options, warrants and similar instruments. “Purchase” and “sale” are defined broadly under the U.S. federal securities laws. “Purchase” includes not only the actual purchase of a security, but any contract to purchase or otherwise acquire a security. “Sale” includes not only the actual sale of a security, but any contract to sell or otherwise dispose of a security. These definitions extend to a broad range of transactions, including conventional cash-for-stock transactions, the grant and exercise of stock options and acquisitions and exercises of warrants or puts, calls or other options related to a security. It is generally understood that “insider trading” includes the following:
●trading by insiders while in possession of material non-public information;
●trading by persons other than insiders while in possession of material non-public information where the information either was given in breach of an insider’s fiduciary duty to keep it confidential or was misappropriated; and
●communicating or tipping material non-public information to others, including recommending the purchase or sale of a security while in possession of material non-public information.
As noted above, for purposes of this Statement, the terms “purchase” and “sell” of securities exclude the acceptance of options, RSUs or other share-based awards granted by the Company and the exercise of options or vesting of other share-based awards that does not involve the sale of securities. Among other things, the
4
cashless exercise of options (i.e. a transaction in which a person exercises options by using a short-term loan provided by a brokerage firm) does involve the sale of securities and therefore is subject to the Policy.
4.2What Facts are Material?
The materiality of a fact depends upon the circumstances. A fact is considered “material” if there is a substantial likelihood that a reasonable investor would consider it important in making a decision to buy, sell or hold a security or where the fact is likely to have a significant effect on the market price of the security. Material information can be positive or negative and can relate to virtually any aspect of a company’s business or to any type of security, debt or equity.
Examples of material information include (but are not limited to) information concerning:
●dividends;
●corporate earnings or earnings forecasts;
●changes in financial condition or asset value;
●negotiations for the mergers or acquisitions or dispositions of significant subsidiaries or assets;
●significant new contracts or the loss of a significant contract;
●significant new products or services;
●significant marketing plans or changes in such plans;
●capital investment plans or changes in such plans;
●material litigation, administrative action or governmental investigations or inquiries about the Company, any of its affiliated companies, or any of its officers or directors;
●significant borrowings or financings;
●defaults on borrowings;
●new equity or debt offerings;
●significant personnel changes;
●changes in accounting methods and write-offs; and
●any substantial change in industry circumstances or competitive conditions which could significantly affect the Company’s earnings or prospects for expansion.
A good general rule of thumb: when in doubt, do not trade.
4.3What is Non-Public?
Information is “non-public” if it is not available to the general public. In order for information to be considered public, it must be widely disseminated in a manner making it generally available to investors through such media as Dow Jones, Reuters Economic Services, The Wall Street Journal, Bloomberg, Associated Press, PR Newswire or United Press International. Circulation of rumors, even if accurate and reported in the media, does not constitute effective public dissemination.
In addition, even after a public announcement, a reasonable period of time must lapse in order for the market to react to the information. Generally, one should allow approximately forty-eight (48) hours following publication as a reasonable waiting period before such information is deemed to be public.
4.4Who is an Insider?
5
“Insiders” include directors, officers, employees and consultants of a company and anyone else who has material non-public information about a company. Insiders have independent fiduciary duties to their company and its shareholders not to trade on material non-public information relating to the company’s securities. All directors, officers, employees and consultants of the Company are considered insiders with respect to material non-public information about business, activities and securities of the Company. The directors, officers, employees and consultants of the Company may not trade the Company’s securities while in possession of material non-public information relating to the Company or tip (or communicate except on a need-to-know basis) such information to others.
It should be noted that trading by household members of a director, officer, employee or consultant can be the responsibility of such director, officer, employee or consultant under certain circumstances and could give rise to legal and Company-imposed sanctions.
4.5Trading by Persons Other than Insiders
Insiders may be liable for communicating or tipping material non-public information to a third party (a “tippee”), and insider trading violations are not limited to trading or tipping by insiders. Persons other than insiders also can be liable for insider trading, including tippees who trade on material non-public information tipped to them or individuals who trade on material non-public information which has been misappropriated.
Tippees inherit an insider’s duties and are liable for trading on material non-public information tipped to them by an insider. Similarly, just as insiders are liable for the insider trading of their tippees, so are tippees who pass the material non-public information along to others who trade on such information. In other words, a tippee’s liability for insider trading is no different from that of an insider. Tippees can obtain material non-public information by receiving overt tips from others or through, among other things, conversations at social, business, or other gatherings.
4.6Material Non-public Information Regarding Other Companies
This Policy and the guidelines described herein also apply to material non-public information relating to other companies, including the Company’s customers, vendors and suppliers (“Business Partners”), particularly when that information is obtained in the course of employment with, or other services performed by, or on behalf of, the Company. Civil and criminal penalties, and discipline, including termination of employment for cause, may result from trading on material non-public information regarding the Company’s Business Partners. Each individual should treat material non-public information about the Company’s Business Partners with the same care required with respect to information related directly to the Company.
5.REVIEW AND MODIFICATION TO THIS POLICY
This Policy and its implementation shall be reviewed by the Compliance Officer from time to time. All directors, officers, employees and consultants of the Company shall be notified of and required to agree to any modification to this Policy.
6
CERTIFICATION OF COMPLIANCE
TO: Compliance Officer
RE: STATEMENT OF POLICIES OF PRENETICS GLOBAL LIMITED GOVERNING MATERIAL NON-PUBLIC INFORMATION AND THE PREVENTION OF INSIDER TRADING
I have received, reviewed, and understand the policies set forth in the above-referenced Statement of Policies (such policies, as amended from time to time, the “Policy”) and hereby undertake, as a condition to my present and continued employment at or association with Prenetics Global Limited or any of its subsidiaries or affiliated entities, to comply fully with the Policy.
I hereby certify that I have adhered to the Policy during the time period that I have been employed by or associated with Prenetics Global Limited or any of its subsidiaries or affiliated entities.
I hereby undertake to adhere to the Policy in the future.
Signature: __________________________
Name: _____________________________
Passport Number: _____________________________
Title: _______________________________________
Date: _______________________________________
Exhibit 12.1
Certification by the Principal Executive Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Danny Sheng Wu Yeung, certify that:
1. I have reviewed this annual report on Form 20-F of Prenetics Global Limited;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report;
4. The company’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have:
(a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c) Evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation;
(d) Disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and
5. The company’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions):
(a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and
(b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting.
Date: April 30, 2026
/s/ Danny Sheng Wu Yeung
Name: Danny Sheng Wu Yeung
Title: Chief Executive Officer
Exhibit 12.2
Certification by the Principal Financial Officer Pursuant to Section 302 of the Sarbanes-Oxley Act of 2002
I, Lo Hoi Chun (Stephen), certify that:
1. I have reviewed this annual report on Form 20-F of Prenetics Global Limited;
2. Based on my knowledge, this report does not contain any untrue statement of a material fact or omit to state a material fact necessary to make the statements made, in light of the circumstances under which such statements were made, not misleading with respect to the period covered by this report;
3. Based on my knowledge, the financial statements, and other financial information included in this report, fairly present in all material respects the financial condition, results of operations and cash flows of the company as of, and for, the periods presented in this report;
4. The company’s other certifying officer(s) and I are responsible for establishing and maintaining disclosure controls and procedures (as defined in Exchange Act Rules 13a-15(e) and 15d-15(e)) and internal control over financial reporting (as defined in Exchange Act Rules 13a-15(f) and 15d-15(f)) for the company and have:
(a) Designed such disclosure controls and procedures, or caused such disclosure controls and procedures to be designed under our supervision, to ensure that material information relating to the company, including its consolidated subsidiaries, is made known to us by others within those entities, particularly during the period in which this report is being prepared;
(b) Designed such internal control over financial reporting, or caused such internal control over financial reporting to be designed under our supervision, to provide reasonable assurance regarding the reliability of financial reporting and the preparation of financial statements for external purposes in accordance with generally accepted accounting principles;
(c) Evaluated the effectiveness of the company’s disclosure controls and procedures and presented in this report our conclusions about the effectiveness of the disclosure controls and procedures, as of the end of the period covered by this report based on such evaluation;
(d) Disclosed in this report any change in the company’s internal control over financial reporting that occurred during the period covered by the annual report that has materially affected, or is reasonably likely to materially affect, the company’s internal control over financial reporting; and
5. The company’s other certifying officer(s) and I have disclosed, based on our most recent evaluation of internal control over financial reporting, to the company’s auditors and the audit committee of the company’s board of directors (or persons performing the equivalent functions):
(a) All significant deficiencies and material weaknesses in the design or operation of internal control over financial reporting which are reasonably likely to adversely affect the company’s ability to record, process, summarize and report financial information; and
(b) Any fraud, whether or not material, that involves management or other employees who have a significant role in the company’s internal control over financial reporting.
Date: April 30, 2026
/s/ Lo Hoi Chun (Stephen)
Name: Lo Hoi Chun (Stephen)
Title: Chief Financial Officer
Exhibit 13.1
Certification by the Principal Executive Officer Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of Prenetics Global Limited (the “Company”) on Form 20-F for the year ended December 31, 2025 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, Danny Sheng Wu Yeung, Chief Executive Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that to my knowledge:
(1) The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934; and
(2) The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
Date: April 30, 2026
/s/ Danny Sheng Wu Yeung
Name: Danny Sheng Wu Yeung
Title: Chief Executive Officer
Exhibit 13.2
Certification by the Principal Financial Officer Pursuant to Section 906 of the Sarbanes-Oxley Act of 2002
In connection with the Annual Report of Prenetics Global Limited (the “Company”) on Form 20-F for the year ended December 31, 2025 as filed with the Securities and Exchange Commission on the date hereof (the “Report”), I, Lo Hoi Chun (Stephen), Chief Financial Officer of the Company, certify, pursuant to 18 U.S.C. Section 1350, as adopted pursuant to Section 906 of the Sarbanes-Oxley Act of 2002, that to my knowledge:
(1) The Report fully complies with the requirements of Section 13(a) or 15(d) of the Securities Exchange Act of 1934; and
(2) The information contained in the Report fairly presents, in all material respects, the financial condition and results of operations of the Company.
Date: April 30, 2026
/s/ Lo Hoi Chun (Stephen)
Name: Lo Hoi Chun (Stephen)
Title: Chief Financial Officer
Exhibit 15.1
Consent of Independent Registered Public Accounting Firm
We consent to the incorporation in this Registration Statement on Form S-8 filed with the U.S. Securities and Exchange Commission (SEC) of our report dated April 30, 2026, relating to the consolidated financial statements of Prenetics Global Limited appearing in the Annual Report on Form 20-F of Prenetics Global Limited for the years ended December 31, 2025 and 2024, respectively.
/s/ Deloitte Touche Tohmatsu
Hong Kong, People's Republic of China
April 30, 2026
Exhibit 15.2

Consent of Independent Registered Public Accounting Firm
We consent to the incorporation by reference in the registration statements (No. 333-288824) on Form F-3 and registration statement (No. 333-287017) on Form S-8 of our report dated April 30, 2024, except for the effects of discontinued operations of ACT Genomics discussed in note 10, as to which the date is April 30, 2026 with respect to the consolidated financial statements of Prenetics Global Limited.
/s/ KPMG
Hong Kong, China
April 30, 2026

Exhibit 15.3
Date: April 30, 2026
Prenetics Global Limited
Unit 703-706, K11 Atelier
728 King’s Road, Quarry Bay
Hong Kong
Dear Sir/Madam:
We hereby consent to the use of our name and the summary of our opinion under the headings, “Item 3. Key Information—Permissions Required from the PRC Authorities for Our Operations,” “Item 3. Key Information—Risks Related to Doing Business in Hong Kong” and “Item 3. Key Information—D. Risk Factors—Risks Relating to Doing Business in Hong Kong,” included in Prenetics Global Limited’s Annual Report on Form 20-F for the year ended December 31, 2025 (the “Annual Report”), which will be filed with the Securities and Exchange Commission (the “SEC”) in the month of April 2026, and further consent to the incorporation by reference, into Prenetics Global Limited’s registration statements on Form S-8 (File Nos. 333-267956, 333-271552, 333-279019 and 333-287017) pertaining to Prenetics Global Limited’s Share Incentive Plan and the Registration Statements on Form F-3 (File Nos. 333-294765, 333-274762, 333-276538 and 333-288824), of the summary of our opinion under the headings “Item 3. Key Information—Permissions Required from the PRC Authorities for Our Operations,” “Item 3. Key Information—Risks Related to Doing Business in Hong Kong” and “Item 3. Key Information—D. Risk Factors—Risks Relating to Doing Business in Hong Kong” in the Annual Report. We also consent to the filing of this consent letter with the SEC as an exhibit to the Annual Report.
In giving such consent, we do not hereby admit that we come within the category of persons whose consent is required under Section 7 of the Securities Act of 1933, or under the Securities Exchange Act of 1934, in each case, as amended, or the regulations promulgated thereunder.
Yours Sincerely, /s/ DaHui Lawyers DaHui Lawyers | |||||
